
(Sample Problems 4.1 and 4.2)
4.1 What two factors cause water to be polar?
Interpretation: Two factors that cause polarity in
Concept introduction: Solution is made by two portions, solute, and solvent. Solute represents substance present in small quantity while solvent represents substance that dissolves solute.
Polarity in molecule is generated if it has unequal distributed electron density between bonds. Polar molecule has partial charges on atoms of the molecule. The general representation of polar molecule is as follows:
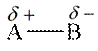
Here, B has high electronegative value than A.
Explanation of Solution
Water molecule has bend shape. In this shape, oxygen present as central atom and two hydrogen atoms are present as side atom. Structure of

Bond
Want to see more full solutions like this?
Chapter 4 Solutions
Principles of General Chemistry
- hen the ionic solute K3PO4is dissolved in water, what can you say about the number of potassium ions (K+)present in the solution compared with the number of phosphate ions (PO43)in the solution?arrow_forward4.5 Lithium fluoride (LiF) is a strong electrolyte. Whatspecies are present in LiF(aq)?arrow_forward2.1. In an acid-base reaction, hydrochloric acid reacts with magnesium hydroxide to give rise to a salt and water. draw a schematic representation of the products, showing the hydrated ions and water molecule orientation.arrow_forward
- 197 Chapter 4 Problems 4.31 Concentrated sulfuric acid (18.3 M) has a density of 1.84 g/mL. (a) How many moles of H2SO, are in each milliliter of solution? (b) What is the mass% of H SO in the solution?arrow_forwardProblems 4.17 Two aqueous solutions of AgNO, and NaCl are mixed. Which of the diagrams (a)-(d) best repre- sents the mixture? For simplicity, water molecules are not shown. (Color codes: Ag* = gray, Cr" = orange, Na* = green, NO5 = blue) == (a) (b) (c) (d)arrow_forward4.4 Discuss the process of sampling and preparation of laboratory sample in quantitative analysis.arrow_forward
- What happens to an ionic compound such as calcium iodide (Cal,) when it dissolves in water? Include a cation and an anion in the explanation. 5.arrow_forward. j. 7 29. If 25.00 ml of 2.333 M FeClg is diluted to 100.00 ml, what is the molarity of iron (III) chloride in the dilute solution? Select one: a. none of these b. 0.7583 M c. 9.332 M d. 0.5833 M e. 0.2614 M f.0.9332 M 30. Some ionic compounds will dissolve in water, other ionic compounds will not dissolve in water. Those ionic compounds which do dissolve in water do so because the attractions þetween each type of ion and seeral water moleculesarrow_forward4.6 The aqueous solutions of three compounds are shown inthe diagrams. Identify each compound as a nonelectrolyte,a weak electrolyte, or a strong electrolyte.arrow_forward
- 4.16 What is the advantage of writing net ionic equations?arrow_forward4.97 Which of the following aqueous solutions would you expect to be the best conductor of electricity at 25°C? Explain your answer.arrow_forwardSuppose you have 50. mL of 0.333 M magnesium acetate. You transfer 5.00 mL of this solution to a 25.0 mL volumetric flask and dilute to the mark (i.e. 25.0 mL). Then you transfer 1.00 mL of this solution to a 50.0 mL volumetric flask and dilute to the mark (i.e. 50.0 mL). 1. What is the concentration of the magnesium acetate in the final solution? 2. What is the concentration of magnesium ion in the final solution? 3. What is the concentration of acetate ion in the final solution?arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
