
Study Guide for Campbell Biology
11th Edition
ISBN: 9780134443775
Author: Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Jane B. Reece, Martha R. Taylor, Michael A. Pollock
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 4, Problem 2SYK
Fill in the following table to review the important chemical groups of organic compounds.
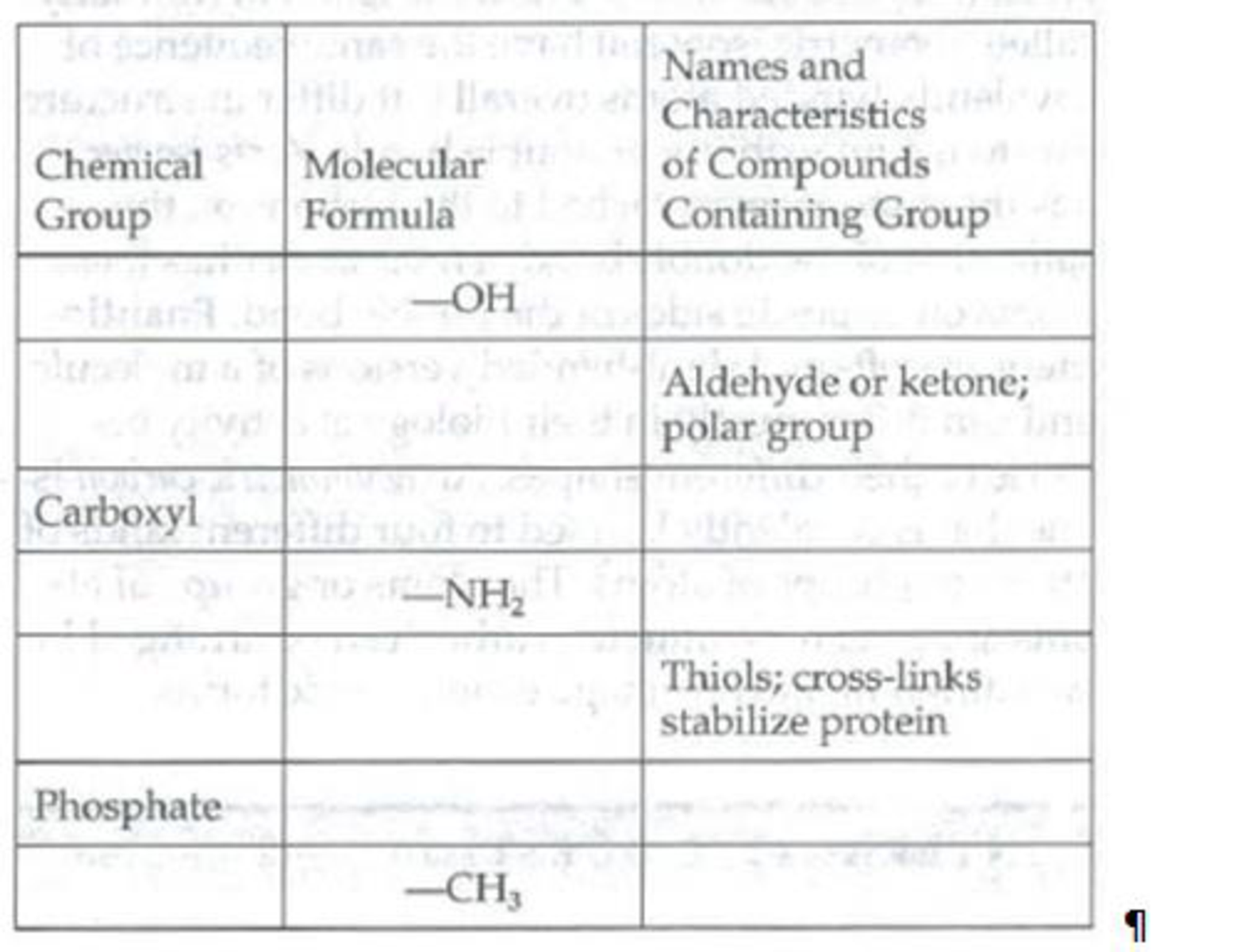
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Choose from the columns the compound that correctly represents the functional groups listed (rows). Structure of compounds are shown below.
In a tabulated form enumerate and draw the functional groups, give examples of biological compound having this functional group and identify its biochemical importance
Functional Groups and Structure
Examples Biological compound and its structure
Biological Importance
Examine the water molecule in figure 2.14 of the textbook . What type of bond occurs between each hydrogen and the oxygen
Chapter 4 Solutions
Study Guide for Campbell Biology
Ch. 4 - How did Millers classic experiment relate to the...Ch. 4 - Define structural isomers, cis-trans isomers, and...Ch. 4 - Practice recognizing the functional groups by...Ch. 4 - Construct a concept map that illustrates your...Ch. 4 - Fill in the following table to review the...Ch. 4 - Carbons valence of four most directly results from...Ch. 4 - Prob. 2TYKCh. 4 - Hydrocarbons are not soluble in water because a....Ch. 4 - Prob. 4TYKCh. 4 - Which of the following is not true of an...
Ch. 4 - Enantiomers are a. molecules that are mirror...Ch. 4 - Which statement is not true about structural...Ch. 4 - Prob. 8TYKCh. 4 - The orbitals of a carbon atom form a(n) a....Ch. 4 - The following ribose molecule contains how many...Ch. 4 - Cis-trans isomers require a. highly polar...Ch. 4 - The chemical group that can cause an organic...Ch. 4 - The chemical group that confers acidic properties...Ch. 4 - Structural isomersCh. 4 - Prob. 2TYKMCh. 4 - can have enantiomersCh. 4 - carboxylic acidCh. 4 - can make cross-link in proteinCh. 4 - hydrophilicCh. 4 - hydrocarbonCh. 4 - amino acidCh. 4 - organic phosphateCh. 4 - aldehydeCh. 4 - amineCh. 4 - ketone
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Compare the polarity of the circled bond in the molecule below. Answer the two questions below: Is this bond polar or non-polar Why is the bond polar or non-polar?arrow_forwardDefine the following terms: a. hydrocarbon b. hydrophilic c. hydrophobic d. functional group e. R grouparrow_forwardMatch the organic compounds with their corresponding examplesarrow_forward
- Distinguish between a nitrogenous base and a base found in inorganic chemistry.arrow_forwardExplain this statement: “All compounds are molecules, but not allmolecules are compounds.” Give an example.arrow_forwardComplete the table below by supplying AT LEAST ONE example of a monomer with its polymer for each macromolecule.arrow_forward
- What are differences between covalent bonds (both polar and nonpolar) and ionic bonds. Recognize examples of compounds with each type of bond.arrow_forwardDraw a “dot & cross” diagram to show the shape of a water molecule (H2O). Bond angles need to be shown. State the shape of a water molecule.arrow_forwardNucleus acids are organic compounds. In addition to hydrogen, what element is an essential component of all organic compounds?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
GCSE Chemistry - Acids and Bases #34; Author: Cognito;https://www.youtube.com/watch?v=vt8fB3MFzLk;License: Standard youtube license