
(a)
Interpretation:
The foods in Table
(a)
Explanation of Solution
The Table
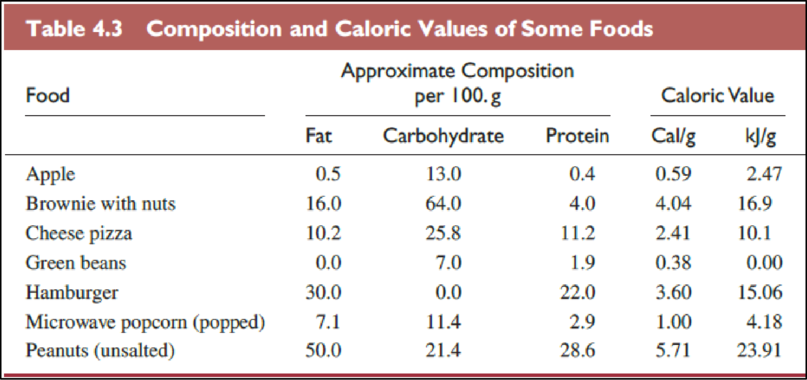
The food items in above table are ranked from low caloric value to high caloric value as follows,
(b)
Interpretation:
The foods in Table
(b)
Explanation of Solution
The Table
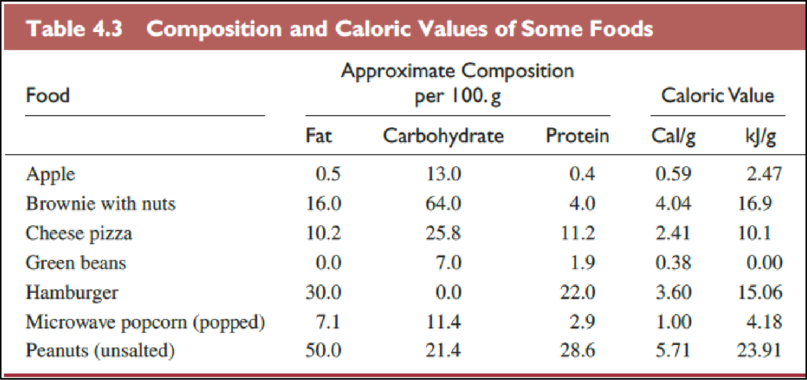
The food items in above table are ranked from low fat content per
(c)
Interpretation:
The foods in Table
(c)
Explanation of Solution
The Table
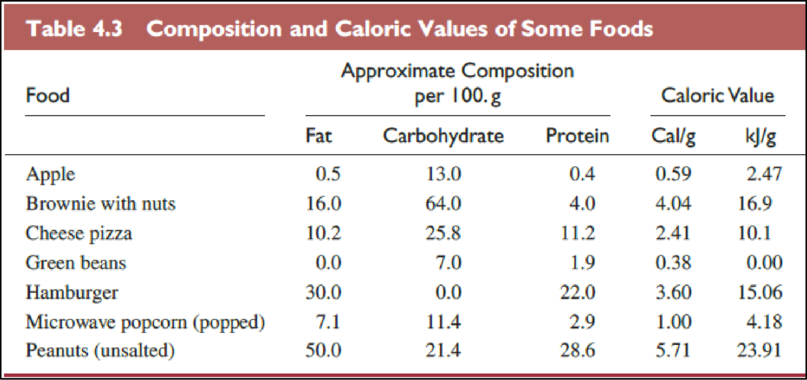
The food items in above table are ranked from most healthful to least one as follows,
Observing sub parts (a) and (b) shows that they matches very well since only 2 items are interchanged. The third list discussed sub part (c) had two high fat foods closer to top of the list, due to fact that protein is important for a healthy diet. Hamburger and peanuts are good protein sources.
Advantage of using caloric value/fat content in food evaluation is that it helps individuals to understand which food helps in weight reduction or weight gain of the individuals. This is achieved by finding alternatives food without consuming more fuel than needed.
Disadvantage of caloric value/fat content in food evaluation is that fats and proteins are important nutritional components of healthy diet. Some amounts of proteins and fats has to be included in daily diet to achieve healthy life style.
Want to see more full solutions like this?
Chapter 4 Solutions
Chemistry: The Molecular Science
- Which is the least expensive source of energy in kilojoules per dollar: a box of breakfast cereal that weighs 32 ounces and costs $4.23, or a liter of isooctane (density, 0.69 19 g/mL) that costs $0.45? Compare the nutritional value of the cereal with the heat produced by combustion of the isooctane under standard conditions. A 1.0-ounce serving of the cereal provides 130 Calories.arrow_forwardHypothetical elements A2 and B2 react according to the following equation, forming the compound AB. A2(aq)+B2(aq)2AB(aq);H=+271kJ/mol If solutions A2(aq) and B2(aq), starting at the same temperature, are mixed in a coffee-cup calorimeter, the reaction that occurs is a exothermic, and the temperature of the resulting solution rises. b endothermic, and the temperature of the resulting solution rises. c endothermic, and the temperature of the resulting solution falls. d exothermic, and the temperature of the resulting solution falls. e exothermic or endothermic, depending on the original and final temperatures.arrow_forward9.30 For the example of shallow water and sandy beaches, which material has a larger heat capacity or specific heat? How does a hot day at the beach provide evidence for your answer?arrow_forward
- A soluble salt, MX2, is added to water in a beaker. The equation for the dissolving of the salt is: MX2(s)M2+(aq)+2X(aq);H0 a Immediately after the salt dissolves, is the solution warmer or colder? b Indicate the direction of heat flow, in or out of the beaker, while the salt dissolves. c After the salt dissolves and the water returns to room temperature, what is the value of q for the system?arrow_forwardSulfur dioxide gas reacts with oxygen, O2(g), to produce SO3(g). This reaction releases 99.0 kJ of heat (at constant pressure) for each mole of sulfur dioxide that reacts. Write the thermochemical equation for the reaction of 2 mol of sulfur dioxide, and then also for the decomposition of 3 mol of sulfur trioxide gas into oxygen gas and sulfur dioxide gas. Do you need any other information to answer either question?arrow_forwardIn a calorimetric experiment, 6.48 g of lithium hydroxide, LiOH, was dissolved in water. The temperature of the calorimeter rose from 25.00C to 36.66C. What is H for the solution process? LiOH(s)Li(aq)+OH(aq) The heat capacity of the calorimeter and its contents is 547 J/C.arrow_forward
- 9.85 The figure below shows a "self-cooling" beverage can. The can is equipped with an outer jacket containing sodium carbonate (Na2CO3), which dissolves in water rapidly and endothermically: Na2CO3(s)2Na+(aq)+CO32(aq) H= 67.7 kJ The user adds water to the outer jacket, and the heat absorbed in the chemical reaction chills the drink. The can contains 200 g of drink, the jacket contains 55 g of Na2CO3, and 100 g of water is to be added. If the initial temperatures of the can and the water are both 32°C on a summer day, what is the coldest temperature that the drink can reach? The can itself has a heat capacity of 40 J/C Assume that the Na2CO3 solution and the drink both have the same heat capacity as pure water, 4.184 J g-l C-l, (HINT: Treat this like a calorimetry problem.)arrow_forwardA piece of unknown solid substance weighs 437.2 g, and requires 8460 J to increase its temperature from 19.3 °C to 68.9 °C. (a) What is the specific heat of the substance? (b) If it is one of the substances found in Table 5.1, what is its likely identity?arrow_forward9.15 Carry out the following conversions of energy units: (a) 14.3 Btu Into calories, (b) 1.4105 cal into joules, (c) 31.6 mJ into Btuarrow_forward
- 9.82 The specific heat of gold is 0.13 J g-1 K-1 and that of copper is 0.39 J g-1 K-1. Suppose that we heat both a 25-g sample of gold and a 25-g sample of copper to 80C and then drop them into identical beakers containing 100 mL of cold water at 10°C. When each beaker reaches thermal equilibrium, which of the following will be true, and why? (You should not need to calculate the actual temperatures here.) (a) Both beakers will be at the same temperature. (b) The beaker with the copper sample in it will be at a higher temperature. (c) The beaker with the gold sample in it will be at a higher temperaturearrow_forwardHydrogen sulfide, H2S, is produced during decomposition of organic matter. When 0.5000 mol H2S burns to produce SO2(g) and H2O(l), 281.0 kJ of heat is released. What is this heat in kilocalories?arrow_forwardConsider the following specific heats of metals. Metal Specific Heat copper 0.385 J/(gC) magnesium 1.02 J/(gC) Mercury 0.138 J/(gC) Silver 0.237 J/(gC) Four 25-g samples, one of each metal, and four insulated containers with identical water volumes, all start out at room temperature. Now suppose you add exactly the same quantity of heat to each metal sample. Then you place the hot metal samples in different containers of water (that all have the same volume of water). Which of the answers below is true? a The water with the copper will be the hottest. b The water with the magnesium will be the hottest. c The water with the mercury will be the hottest. d The water with the silver will be the hottest. e The temperature of the water will be the same in all the cups.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





