
Concept explainers
(a)
Interpretation:
Whether the given set are considered as valid pairs of contributing structures or not.
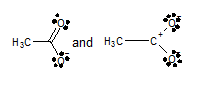
Concept Introduction:
Resonance is the method which explains electron’s delocalization in the molecules. It contrasts and compares more than two Lewis strictures which can signify the specific molecule. When a single molecule couldn’t explain the proper bonding between the two atoms so for explaining the proper bonding between two atoms resonance structure is used in place of Lewis structure.
(b)
Interpretation:
Whether the given set are considered as valid pairs of contributing structures or not.
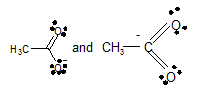
Concept Introduction:
In
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Introduction to General, Organic and Biochemistry
- Problem 3-6 Give each binary compound a systematic name and a common name. (a) FeO (b) Fe2O3arrow_forwardProblem 3-4 Name these binary ionic compounds: (a) MgO ( b) BaI2 (c) KClarrow_forwardProblem 2: Calculate the formal charges to determine the most probable Lewis structure for the thiocyanate ion. [:N=c=s:] [:c=N=S:] [:c=s=N:]arrow_forward
- The strength of a covalent bond depends upon the size of the atoms and the bond order. In general short bonds are strong bonds. For each pair of covalently bonded atoms, choose the one expected to have the higher bond energy. progress (A) N=N (В) N-N (A,B) (C) С-С (D) C=C (С,D) Submit Answer Retry Entire Group No more group attempts remainarrow_forwardWhich of the following contains both ionic and covalent bonds in the same compound? O MgH, NH,+ Bag (PO4)2 O H,O O FeO Submit Answer Try Another Version 6 item attempts remaining ot pt pt pt pt 1 pt 1 ptarrow_forwardProblem 3-7 Name these ionic compounds, each of which contains a polyatomic ion: (a) K2HPO4 (b) Al2(SO4)3 (c) FeCO3arrow_forward
- Practice Exercise: Given the following organic compounds, identify and explain the types of isomers in those samples. 1. CH3 CH3 CH3 C=C H. C=C CHs 2. HH OH || | н он н H-C-C-C-H | | | H CHz H H CHz H 3. CH3CH2-C CHs-C 0-H 0-CH3 4. H. HHHH H CHs H H-C-C-C-C-C-H H-C-C-C-H |||| HH H H H H ČHz H 5. HH CHs H-C-C. H-C-C-Harrow_forwardPROBLEM 1-9 Draw the important resonance forms of the following cations and anions: (a) (b) (c) (e) + CH3O–CH–CH=CH–NH NH₂ + OH H₂C=CH-C=OH H3C-C-CH-CH=CH-CN (d) H H₂N-CH=CH-C-OCH3 H3C-CH-C-CH=CH-CN NH || H₂C=CH-C-CH₂arrow_forwardSome of the following structural formulas are incorrect because they contain one or more atoms that do not have their normal number of covalent bonds. Which structural formulas are incorrect? (Select all that apply.) нн Cl-c=ċ-H H O H-CEC-C=C-H H H H нннн H-o-c-c-N-c-H H. H. =c-o-c- Br H H Submitarrow_forward
- Problem 3-2 Judging from their relative positions in the Periodic Table, which element in each pair has the larger electronegativity? (a) Lithium or potassium (b) Nitrogen or phosphorus (c) Carbon or siliconarrow_forwardQUESTION 5 Which is the most stable resonance structure for the anion ONS¹-? (A) :0-N=S (C) (E) 0 N= 0 0 0 0 0 BCD ‒‒ A (D) :0—N—S: ‒‒ E (B) - 0=N 10: - EN = S:arrow_forwardUse the Ret nces to access impor för this question. The length of a covalent bond depends upon the size of the atoms and the bond order. For each pair of covalently bonded atoms, choose the one expected to have the shortest bond length. (A) C-O (B) С-0 (A,B) (C) C=N (D) C=N (С,D) Submit Answer Try Another Version 2 item attempts remaining Visitedarrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning


