
Interpretation:
Predict the result of the catalytic hydrogention of natural rubber and will the product obtained show syndiotactic, atactic or isotactic behaviour.
Concept introduction:
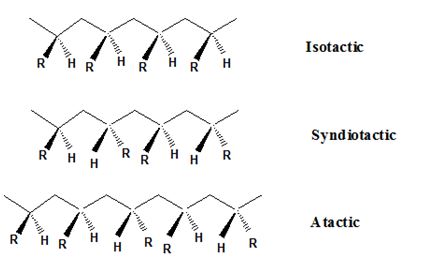
Isotactic, syndiotactic, and atactic are the stereochemical forms. The
Syndiotactic are the macromolecules in which the (-R) groups are arranged in an alternate manner along the long chain of the polymer. Gutta-percha is also an example for Syndiotactic polymer.
In atactic form, the substituents are placed in a random manner along the long chain.
The important point to note here is that the polymer obtained from the chain-growth
The catalytic hydrogenation natural rubber requires the breaking of double bond and the addition of hydrogen. The double bond of
Therefore, it breaks and hydrogen adds to it. When the hydrogen adds, then the double bonds are replaced with the single bonds. The hydrogenation of double bond releases energy therefore, known as an exothermic reaction. The heat released is called the heat of hydrogenation.
The catalyst used for the purpose of hydrogenation can be Ra-Ni (Raney-Nickel), PtO2 (Platinum oxide), Pd-C (Palladium on carbon) etc. They can be used to enhance the
Trending nowThis is a popular solution!

Chapter 31 Solutions
Organic Chemistry
- Vacuum-forming is a term that is sometimes used to describe the polymer conversion process known as ____. Why? Explain and support answer. A) pressure- forming B) Suction- Forming C) Thermo-Forming D) Draw-Formingarrow_forwardGive the structure of the condensation copolymer made from the following monomers: HOOCCH2CH2COOH and H2NCH2CH2CH2CH2CH2CH2NH2arrow_forwardPolymers are often thought to be manufactured in immense chemical plants. Although many are, there is a large group of polymers which are natural. State 2 natural or synthetic polymers and describe how they are utilized in our society (1K, 2C).arrow_forward
- Macromolecules can also be formed from cyclic monomers such as cyclic hydrocarbons, ethers, esters, amides, siloxanes, and sulfur (eight-membered ring). Thus, ring-opening polymerization is of particular interest, since macromolecules of almost any chemical structure can be prepared. Choose two of the above type polymers to explain the mechanism and chemical reaction of polymerization process.arrow_forwardOlefin polymerization is responsible for producing over 100 million tons per year of plastic material. (a) Briefly describe the type of catalysts used in the transition metal catalysed homogeneous polymerization reactions. (b) How are the catalysts activated? (c) Draw the active catalyst, and indicate how the polymer chain is propagated. (d) Suggest a possible chain termination step. (e) Certain single-site catalysts are capable of selectively forming different types of polypropylene. Describe these different types of polymer, and the characteristics of the catalysts that form them.arrow_forwardTires whose sidewalls are made of natural rubber tend to crack and weather rapidly in areas around cities where high levels of ozone and other industrial pollutants are found. Explain.arrow_forward
- In a groundwater 1,1,1 trichloroethane, hexachlorobenzene and 1,1,2 trichloroethane have leached from an underground industrial landfill. Which compound would you expect to move fastest with groundwater? Which would be easiest to remove by air stripping? Which would be best to remove by adsorption?arrow_forwardWhat is the first step in the cationic polymerization of alkenes?arrow_forwardProduct? ASAParrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning


