
Concept explainers
Arrange the following pairs of charged particles in order of increasing magnitude of electrostatic attraction (Eel).
(a) i < ii < iii < iv
(b) iv < iii < ii < i
(c) i = iii < ii < iv
(d) ii < i = iii < iv
(e) iv < i = ii < iii
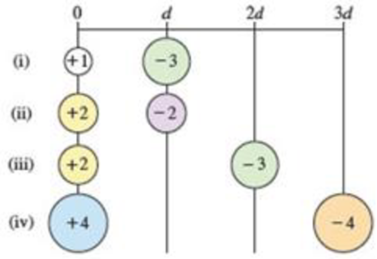
Interpretation:
The electrostatic potential energies between the given charges in the given statement should be compared by using the concept of electrostatic energy.
Concept Introduction:
Energy is the ability to do work or transfer heat where work is the movement of a body using some force. The SI unit of energy is joule (
Electrostatic energy is potential energy which results from the interaction of charged particles. Oppositely charged particles attract each other and particles of like charges repel each other. The magnitude of the resulting electrostatic potential energy is proportional to the product of the two charges (
If the charges
To calculate: Determine the
Answer to Problem 3.1.3SR
The given pairs of charged particles in order of increasing magnitude of electrostatic attraction (
Explanation of Solution
Reason for correct option
The electrostatic energy between two charges is calculated using the formulae:
It is used to compare the magnitudes of the
For charges +1 and −3:
For charges +2 and −2:
For charges +2 and −3:
For charges +4 and −4:
Therefore, the given pairs of charged particles in order of increasing magnitude of electrostatic attraction (
Reasons for incorrect options:
By doing the calculations in which the given values are substituted, it is found that the option (a) is correct. Therefore, the options (b), (c), (d) and (e) are incorrect.
The electrostatic potential energies between the given charges in the given statement are compared by using the concept of electrostatic energy.
Want to see more full solutions like this?
Chapter 3 Solutions
GEN COMBO CHEMISTRY: ATOMS FIRST; ALEKS 360 2S ACCESS CARD CHEMISTRY:ATOMS FIRST
- For each of the following covalent bonds: (a) use the symbols δ+ and δ- to indicate the direction of polarity (if any).(a) C-F; (b) N-Br; (c) B-C; (d) Si-H(b) Rank the following covalent bonds in order of increasing polarity. (i) C-H, O-H, N-H; (ii) C-N, C-O, B-O; (iii) C-P, C-S, C-Narrow_forwardIn(s) + Fe(s) -> FeI,(s) Match the words in the left column to the appropriate blanks in the sentences on the right.arrow_forwardFignm x E OWLv2 | Online teaching and lea x ngagenow.com/ilrn/takeAssignment/takeCovalentActivity.do?locator-assignment-take (Reterences Caiculate uie numoer or posiuve ions and negauve ions contaitieu ii 1.00 m01 Or eaca or me Tollowing compounas. NazO positave ions negative ious b Feo positive 1os egative ions PbS, positive ions negative ions Submit AICI, positive 1ons negative ions earch 米 10 %23 %24arrow_forward
- What is the result of like charges, be they phosphates or magnets, being in close proximity?arrow_forwardBetween Na and Ne, which one has higher first ionization energy and why? Make sure to discuss effective nuclear charge, forces, and energy. (2-3 sentences)arrow_forward(A) if both sentences are true, (B) if both sentences are false, (C) if the first sentence is true but the second is false, and (D) if the first sentence is false but the second is true. 1. Water appears in nature in all three common states of matter. Water is a tasteless, odorless liquid at standard temperature and pressure. 2. The addition of salt in the distilling mixture made the water impure and saline. The addition of 5 drops of the silver nitrate solution into the distillate determines whether the distillate is impure or pure water. 3. The ideal electrical conductivity of totally pure water is 0.055 microSiemens, whereas typical deionized water has an electrical conductivity of 0.1 microSiemens. The freezing point of water at 1 atm is 100degC. 4. From the air, oxygen can slowly diffuse across the water’s surface from the surrounding atmosphere, or be mixed in quickly through aeration, whether natural or man-made. The aeration of water can be caused by wind (creating waves),…arrow_forward
- M Inbox (6)- danasia.... 31 Ascension - Calend... ||| O Advanced Material Predicting the relative lattice energy of binary ionic compounds In each row, pick the compound with the bigger lattice energy. Note: lattice energy is always greater than zero. Which compound has the bigger lattice energy? CaBr₂ Cal₂ O O Cs Cl FH Explanation tab X Esc O Rb₂0 Check Type here to search ! 1 F1 Q F2 Shared drives - Goo... Intrane @ 2 W t F3 # m 3 H F4 $ E 4 & F5 % 5 Rarrow_forwardThe attractive force between two ions in Na20, Fc is 8.64x10-9N. What is the bond length a0 (in nm) given that valence of the charged Na+ is Z1=+1, for O-, is Z2 =-2, the charge of a single electron q is 0.16x10-18 C and the proportionality constant k0=9x109 V.m/C? Note that K=k0(Z1q)(Z2q) and that 1 V.C = 1 J.arrow_forwardWhich of the following is the correct representation of partial charges at the indicated atoms? a. I = δ+; II = δ–; III = δ– b. I = δ–; II = δ–; III = δ– c. I = δ+; II = δ+; III = δ+ d. I = δ–; II = δ+; III = δ– e. I = δ+; II = δ–; III = δ+arrow_forward
- How to Calcula nVellumHMAC=20ff3692a9b8f8628f99c8c00d38dabd#10301 ps Translate EReading list I Review | Constants Periodic Table VWI IICIT IS Tepiesentu vy 4a T O.I IaIly, uie iast prlast Slales UIEIT auueu lu o, "gives 60," which is the term = 60 to create 4x + 8 = 60. in to Part D You know that a number 8 less than 60 is the number 52. So, you could rewrite your answer. from the previous part as, "What number can you multiply by 4 to get 52?" This is just the definition of 52 divided by 4! So, what is the number x you are looking for? Express your answer as an integer. V AZO ? Submit Request Answer Next > Provide Feedback P Pearson ...arrow_forward1.38,Assuming the following species consist of ions, write the valence electron configura- tions and (if appropriate) the Lewis symbols of the cations in (a) UF; (b) WF,; (c) SCI,: (d) CIO,.arrow_forwardCalculate the lattice energy of RbCl using the following data: AH (k) Rb(3) Rb(g) Chig) CI(g) + e Rb(s) + ¾Cl(g} Rb(g) Rb"(g) + e¯ 86 409 2CI(g) CF(g) RÜCI(s) 242 -355 435arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





