
Identify each
represents a lactone-a cyclic ester- and which represents a lactam-a cyclic amide?
a. 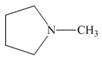 b.
b. 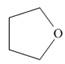 c.
c. 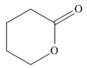 d.
d. 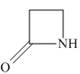
(a)
Interpretation: The functional group present in the given ring and the structure that represent a lactone and a lactam is to be stated.
Concept introduction: The table representing the general formula of different functional groups according to IUPAC convention is given below.
| General formula | Functional group | Functional group name |
| Alcohol | ||
| Aldehyde | ||
| Ketone | ||
| Amine | ||
| Ether | ||
| Ester |
Answer to Problem 3.33P
The functional group present in the given ring is amine. The given structure neither represents a lactone nor a lactam.
Explanation of Solution
The given structure is,
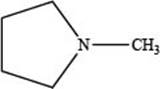
Figure 1
The given structure contains amine as a functional group. In the given structure, nitrogen is bonded to three carbon atoms. Therefore, the given ring is a tertiary amine.
Lactone is a cyclic ester, whereas lactam is a cyclic amide. Thus, the given structure neither represents a lactone nor a lactam.
The functional group present in the given ring is amine. The given structure neither represents a lactone nor a lactam.
(b)
Interpretation: The functional group present in the given ring and the structure that represent a lactone-cyclic ester and a lactam-a cyclic amide is to be stated.
Concept introduction: The table representing the general formula of different functional groups according to IUPAC convention is given below.
| General formula | Functional group | Functional group name |
| Alcohol | ||
| Aldehyde | ||
| Ketone | ||
| Amine | ||
| Ether | ||
| Ester |
Answer to Problem 3.33P
The functional group present in the given ring is ether. The given structure neither represents a lactone nor a lactam.
Explanation of Solution
The given structure is,
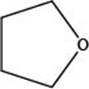
Figure 2
The given structure contains ether as a functional group. The given functional group is in cyclic form. Therefore, the given structure is cyclic ether. Lactone is a cyclic ester, whereas lactam is a cyclic amide. Thus, the given structure neither represents a lactone nor a lactam.
The functional group present in the given ring is ether. The given structure neither represents a lactone nor a lactam.
(c)
Interpretation: The functional group present in the given ring and the structure that represent a lactone-cyclic ester and a lactam-a cyclic amide is to be stated.
Concept introduction: The table representing the general formula of different functional groups according to IUPAC convention is given below.
| General formula | Functional group | Functional group name |
| Alcohol | ||
| Aldehyde | ||
| Ketone | ||
| Amine | ||
| Ether | ||
| Ester |
Answer to Problem 3.33P
The functional group present in the given ring is ester. The given structure represents a lactone-cyclic ester.
Explanation of Solution
The given structure is,
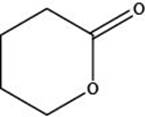
Figure 3
In the given structure, the functional group present is ester. The given functional group is in cyclic form. Therefore, the given structure represents a lactone-cyclic ester.
The functional group present in the given ring is ester. The given structure represents a lactone-cyclic ester.
(d)
Interpretation: The functional group present in the given ring and the structure that represent a lactone-cyclic ester and a lactam-a cyclic amide is to be stated.
Concept introduction: The table representing the general formula of different functional groups according to IUPAC convention is given below.
| General formula | Functional group | Functional group name |
| Alcohol | ||
| Aldehyde | ||
| Ketone | ||
| Amine | ||
| Ether | ||
| Ester |
Answer to Problem 3.33P
The functional group present in the given ring is amide. The given structure represents a lactam-a cyclic amide.
Explanation of Solution
The given structure is,
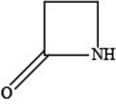
Figure 4
In the given structure, the functional group present is amide. The given functional group is in cyclic form. Therefore, the given structure represents a lactam-a cyclic amide.
The functional group present in the given ring is amide. The given structure represents a lactam-a cyclic amide.
Want to see more full solutions like this?
Chapter 3 Solutions
PKG ORGANIC CHEMISTRY
- What are the functional groups present in this antibacterial antibiotic? A. Amide, thioether, aldehyde, phenol, carboxylic acid B. Amide, thioether, ketone, amine, phenol, carboxylic acid C. Amide, thioether, ketone, phenol, carboxylic acid D. Thioether, ketone, amine, phenol, carboxylic acid A brief explanation would be highly appreciated + upvotearrow_forwardJj.166. Draw each amide. N-butyl-N-methylbenzamidearrow_forward1. Which statement best described the ability of amines to hydrogen bond? A. Primary, secondary and tertiary amines can all hydrogen bond with molecules identical to themselves. B. Primary and secondary amines can hydrogen bond with molecules identical to themselves, but tertiary amines cannot. C. Only primary amines can hydrogen bond with molecules identical to themselves. Secondary and tertiary amines cannot. D. Primary, secondary and tertiary amines cannot hydrogen bond with molecules identical to themselves, but the can hydrogen bond with water.arrow_forward
- Draw a structural formula for each amine and amine derivative. Q.) trans-2-Aminocyclohexanolarrow_forwardamine, (2) an amide, or (3) both an amine and an amide. 17-106 Classify each of the following compounds as (1) an amine, (2) an amide, or (3) both an amine and an NH2 b. `NH a. H2N H d. с.arrow_forwardErythronolide B is the biological precursor of erythromycin, a broad-spectrum antibiotic. What functional group Erythronolide B does contain? a. b. H₂CH₂C C. H₂C 1 H₂C 2 3 4 O Amide d. Amine OH Erythronolide B Ketone Aldehyde a CH₂ b C d CH₂ OH JCH₂ 'OH OHarrow_forward
- Identify the functional groups in the two molecules shown below. Then, for each ester or amide functional group, write a reaction showing how the molecule could be made. H. H.arrow_forwardAmide hydrolysis in basic conditions forms A. a carboxylic acid and an amine B. a carboxylate salt and an amine 3. an ester and an amine 4. a carboxylic acid and an amine saltarrow_forwardDraw a structural formula for each amine and amine derivative. Q.) Benzylaminearrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning





