
DRAW IT Ø Label a hydrogen bond and a polar covalent bond in the diagram of live water molecules. Is a hydrogen bond a covalent bond? Explain.
To label: The hydrogen bond and covalent bonds in water molecules.
Introduction:
Water is a polar molecule consisting of an oxygen atom and two hydrogen atoms.
Explanation of Solution
Water (H2O) consists of an oxygen atom and two hydrogen atoms. Hydrogen and oxygen share their valence electrons to form a strong bond that known as a covalent bond (Fig.1). As oxygen is more electronegative than hydrogen, the shared electrons in the H-O bond tend to be pulled towards the oxygen atom. There are two regions of partial negative charge on oxygen and a partial positive charge on each hydrogen atom. Thus, the covalent bond between H-O in the water molecule is a polar bond.
Water is a polar molecule due to the difference in electronegativity between O and H atoms. The partial negatively charged oxygen atom of a water molecule is attracted to the partial positively charged hydrogen atom of an adjacent water molecule (Fig.1). This forms the hydrogen bond among different water molecules.
Pictorial representation:
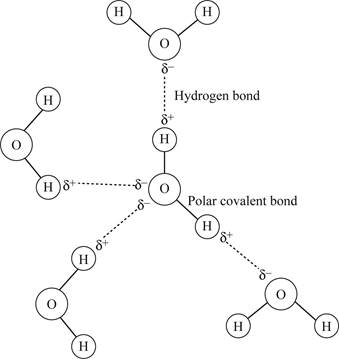
Fig. 1 Interactions among water molecules
To explain: The hydrogen bonds are not covalent bonds.
Introduction: Covalent bonds are formed by the sharing of valence electrons between two atoms. Hydrogen bonds are formed due to the attraction between two adjacent atoms due to partial charges.
Explanation of Solution
Covalent bonds are strong bonds, formed by the sharing of electrons between atoms. Hydrogen bonds do not involve the sharing of valence electrons. They are formed on the basis of attraction due to partial charges on neighboring atoms, and are hence very weak bonds. Therefore, a hydrogen bond is not a covalent bond.
Want to see more full solutions like this?
Chapter 3 Solutions
Campbell Biology (11th Edition)
Additional Science Textbook Solutions
Human Biology: Concepts and Current Issues
Essentials of Genetics (9th Edition) - Standalone book
Genetics: Analysis and Principles
Biological Science (6th Edition)
Fundamentals of Anatomy & Physiology Plus Mastering A&P with eText - Access Card Package (10th Edition) (New A&P Titles by Ric Martini and Judi Nath)
- Figure 2.24 Which of the following statements is false? Molecules with the formulas CH3CH2COOH and C3H6O2 could be structural isomers. Molecules must have a double bond to be cis-trans isomers. To be enantiomers, a molecule must have at least three different atoms or groups connected to a central carbon. To be enantiomers, a molecule must have at least four different atoms or groups connected to a central carbon.arrow_forwardAn ________ can help keep the pH of a solution stable. a. covalent bond c. buffer b. hydrogen bond d. free radicalarrow_forwardWhat kinds of bonds often control the shape (or tertiary form) of large molecules such as proteins? a. hydrogen b. ionic c. covalent d. inert e. singlearrow_forward
- Watch this video (http://openstaxcollege.org/l/disaccharide) to observe the formation of a disaccharide. What happens when water encounters a glycosidic bond?arrow_forwardIn a single molecule of water, the two hydrogen atoms are bonded to a single oxygen atom by nonpolar covalent bonds. polar covalent bonds. ionic bonds. hydrogen bonds. van der Waals interactionsarrow_forwardWhat is the maximum number of hydrogen bonds a water molecule can form with neighboring water molecules? four B) five C) one D) three E) two What determines the cohesiveness of water molecules? covalent bonds hydrogen bonds ionic bonds high specific heat hydrophobic interactionsarrow_forward
- Picture below showing four water molecules interacting via hydrogen bonds? be sure lo label partial charges in each molecule and use dotted line to represent a hydrogen bond.arrow_forwardWhich of the following statements is correct regarding polar/nonpolar molecules? 1. Molecules comprised of nonpolar covalent bonds are likely to be hydrophilic 2. Polar molecules like sugar tend to dissolve in solutions of other polar molecules like water 3. Nonpolar molecules like sugar tend to dissolve in solutions of polar molecules like water 4. Polar covalent bonds tend to form between molecules that consist of two atoms of the same element, like oxygen gas (O2) or nitrogen gas (N2)arrow_forwardWhich of the following represents a covalent interaction? Choose any/all answers that apply. A. salt bridge B. peptide bond C. hydrogen bond D. Van der Waals interaction E. disulfide bondarrow_forward
- Water displays many unique properties making it essential to life. Which of the following chemical characteristic confers water its unique properties? Water displays many unique properties making it essential to life. Which of the following chemical characteristic confers water its unique properties? Hydrogen bond is found within a water molecule. Water is dipolar. Water is highly electronegative. Water is abundant on Earth. Water is a small molecule that makes up of two hydrogen and one oxygen.arrow_forwardExplain in detail why water molecules are polar. Your explanation can be as long as you wish. Your answer must be in your own words. Your answer must include the following terms: Polar, polar covalent bonds, electronegativity, oxygen, hydrogen, partial positive, partial negartive, electrons.arrow_forwardWhich of the following factor(s) may contribute to the stability of folded protein structures? Disulfide bonds. Hydrophobic effect. Electrostatic interactions. Hydrogen bonds. All of the above.arrow_forward

 Human Biology (MindTap Course List)BiologyISBN:9781305112100Author:Cecie Starr, Beverly McMillanPublisher:Cengage Learning
Human Biology (MindTap Course List)BiologyISBN:9781305112100Author:Cecie Starr, Beverly McMillanPublisher:Cengage Learning Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781938168130Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark WomblePublisher:OpenStax College
Anatomy & PhysiologyBiologyISBN:9781938168130Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark WomblePublisher:OpenStax College



