
Organic Chemistry, Books a la Carte Edition (8th Edition)
8th Edition
ISBN: 9780134074580
Author: Bruice, Paula Yurkanis
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 28, Problem 41P
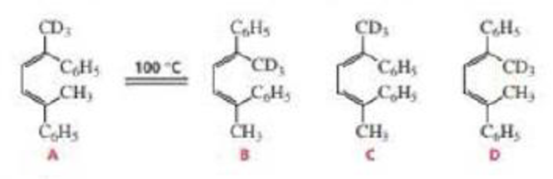
If isomer A is heated to about 100 °C, a mixture of isomers A and B is formed. Explain why there is no trace of isomer C and D.
Expert Solution & Answer
Learn your wayIncludes step-by-step video

schedule06:51
Students have asked these similar questions
4.
Draw the preferred conformation for the following compounds. Provide a
brief explanation as to why you chose that conformation.
a.
b.
Ph
Which isomer, A or B, would you expect to have the
greater heat of combustion? Explain.
A
B
Chemistry
5.66 A mixture contains equal amounts of compounds A-D.
D
B
C
A
a. Which compounds alone are optically active?
b. If the mixture was subjected to fractional distillation, how many fractions would be obtained?
c. How many of these fractions would be optically active?
Chapter 28 Solutions
Organic Chemistry, Books a la Carte Edition (8th Edition)
Ch. 28.1 - Prob. 1PCh. 28.2 - Prob. 2PCh. 28.2 - Prob. 3PCh. 28.2 - Give a molecular orbital description for each of...Ch. 28.3 - Prob. 5PCh. 28.3 - Prob. 6PCh. 28.3 - Prob. 7PCh. 28.3 - Prob. 8PCh. 28.4 - Prob. 10PCh. 28.4 - Prob. 11P
Ch. 28.5 - Prob. 12PCh. 28.5 - a. Draw the product of the following reaction: b....Ch. 28.5 - Prob. 14PCh. 28.5 - Prob. 15PCh. 28.5 - Prob. 17PCh. 28.5 - Prob. 18PCh. 28.6 - Prob. 19PCh. 28.6 - Explain why the hydrogen and the methyl...Ch. 28.6 - Chorismate mutase is an enzyme that promotes a...Ch. 28.7 - Convince yourself that the TE-AC method for...Ch. 28 - Draw the product of each of the following...Ch. 28 - Draw the product of each of the following...Ch. 28 - Prob. 25PCh. 28 - Show how norbornance can be prepared from...Ch. 28 - Prob. 27PCh. 28 - Prob. 28PCh. 28 - Draw the product of each of the following...Ch. 28 - Prob. 30PCh. 28 - Prob. 31PCh. 28 - Prob. 32PCh. 28 - Prob. 33PCh. 28 - When the following compound is heated, a product...Ch. 28 - Prob. 35PCh. 28 - Propose a mechanism for the following reaction:Ch. 28 - Prob. 37PCh. 28 - Prob. 38PCh. 28 - Prob. 39PCh. 28 - Prob. 40PCh. 28 - If isomer A is heated to about 100 C, a mixture of...Ch. 28 - Propose a mechanism for the following reaction:Ch. 28 - Prob. 43PCh. 28 - A student found that heating any one of the...Ch. 28 - Prob. 45PCh. 28 - Prob. 46PCh. 28 - Prob. 47P
Additional Science Textbook Solutions
Find more solutions based on key concepts
Q1. What is the empirical formula of a compound with the molecular formula
Chemistry: A Molecular Approach (4th Edition)
23. Give the symbol and name for (a) an isotope with a mass number of 37 and an atomic number of 17 and (b) an ...
Chemistry For Changing Times (14th Edition)
Without looking at the structures, give molecular formulas for the compounds in Problem 3-8 (a) and (b). Use th...
Organic Chemistry (9th Edition)
Predict whether a precipitation reaction will occur when aqueous solutions of the following substances are mixe...
CHEMISTRY-TEXT
In qualitative analysis, Ca2+ and Ba2+ are separated from Na+, K+, and Mg2+ by adding aqueous (NH4)2CO3 to a so...
General Chemistry: Atoms First
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Identify circle the alkanelike portions of the following molecules: a. b. c. d.arrow_forward5. Which of compounds is likely to adopt a planar conformation? Why? .. A. С. D. В.arrow_forwardWhat is the other isomeric substitution product in the reaction shown below? A. B. C. D. E. CH3OH OCH3 OCH3 OCH3 OCH3 OCH3 OCH3 + isomerarrow_forward
- 18. How many isomers does dichlorocyclobutane have? Consider both constitutional isomers and stereoisomers! A.2 B. 3 C. 4 D. 5 E. 6arrow_forwardProblem 5.26 Without drawing out the structures, label each pair of compounds as enantiomers or diastereomers. a. (2R,3S)-hexane-2,3-diol and (2R,3R)-hexane-2,3-diol b. (2R,3R)-hexane-2,3-diol and (2S,3S)-hexane-2,3-diol c. (2R,3S,4R)-hexane-2,3,4-triol and (2S,3R,4R)-hexane-2,3,4-triolarrow_forwardMake sure all requirements are filled drawing the isomer. from a to farrow_forward
- v 8.42b e Get help answering Molecular Drawing questions. Modify the given carbon skeleton to draw the major product of the following reaction. If a racemic mixture of enantiomers is expected, draw both enantiomers. Note: you can select a structure and use Copy and Paste to save drawing time. HCI H;C, Editarrow_forwardDefine Carbocation Stability?arrow_forwardConsider the following molecule. which Natos is more basic, A or B? CH Aarrow_forward
- How many stereoisomers estuary for this molecule? Show how you know. A. 8 B. 3 C. 4 D. 6arrow_forwardConsider the following molecule: CI a) Determine the absolute configuration (R or S) at each chiral center by clearly indicating on the structure above which atoms are chiral and whether they are R or S. b) The geometric configuration of the alkene (circle one)... cannot be is 'cis'. is 'trans'. is E. is Z. determined. C) Draw both possible chair conformations and identify which conformer would be the most stable. You may use an 'R' group to represent the hydrocarbon substituent. Place the chlorine atom on the (*) starred position on the structure on the left.arrow_forward1. Which term best describes the alkene below? a. cis only b. trans only c. both terms may be used d. neither cis nor transarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning
GCSE Chemistry - Differences Between Compounds, Molecules & Mixtures #3; Author: Cognito;https://www.youtube.com/watch?v=jBDr0mHyc5M;License: Standard YouTube License, CC-BY