
Concept explainers
Interpretation:
The structural formulas for the enethiol forms of 6-mercaptopurine and 6-thioguanine has to be drawn.
Concept Introduction:
The functional group ketone (keto group) will be in equilibrium with the enol (alcohol group) form which is said to be the keto-enol tautomerization. The keto form is more stable when compared to the enol form. The keto-enol conversion reaction is
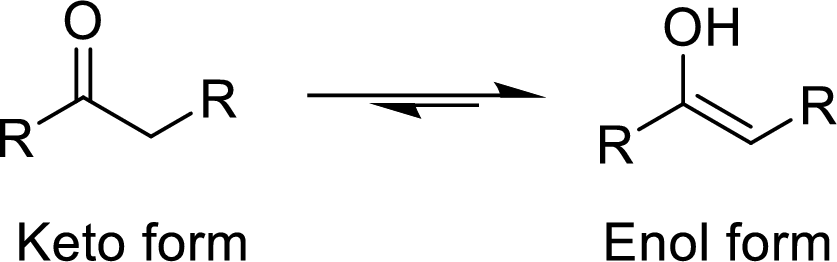
Explanation of Solution
The enethiol is also the enol form which contains Sulphur molecule instead of Oxygen. The enethiol form is the alcoholic form of the Sulphur. The double bonded Sulphur is converted to thiol form of the Sulphur.
The enethiol form of 6-mercaptopurine is
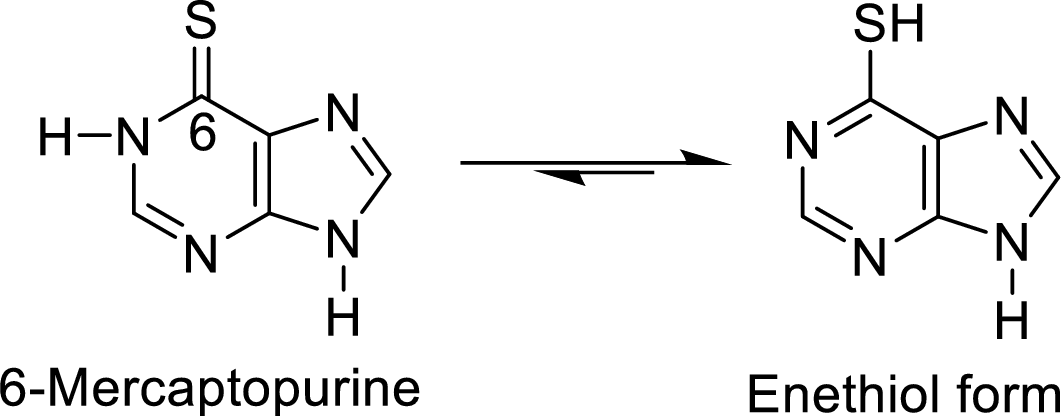
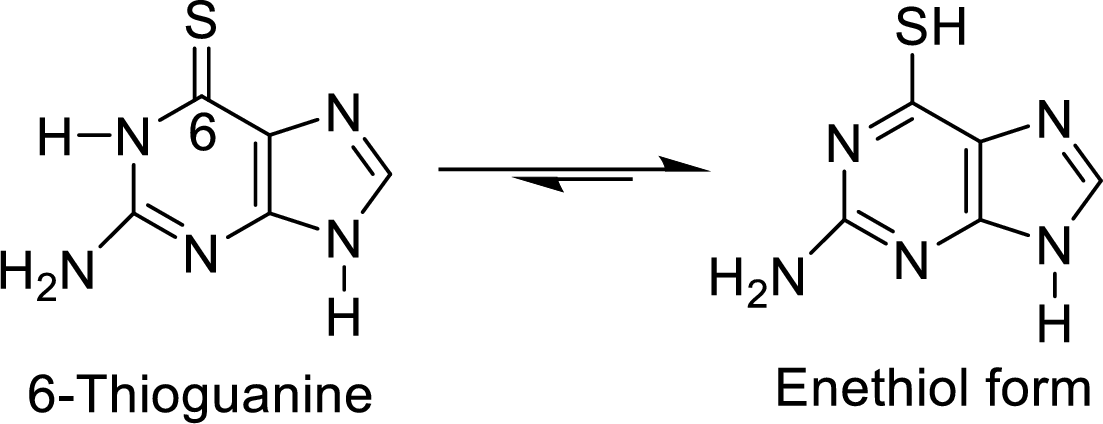
The enethiol form of 6-thioguanine is
Want to see more full solutions like this?
Chapter 28 Solutions
Student Study Guide and Solutions Manual for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- Q23: Dr. Alghe has just performed a restriction digest on DNA she extracted from her samples. She must now run her DNA out on a gel to separate the fragments based on size. To do this she must prepare a 1.1% agarose gel, where the percentage is determined as mass/vol. How many grams of agarose must she add to 413 mL of buffer in order to arrive at the correct percentage? (answer to 2 decimals) **Assume the dissolution of agarose will not alter the final volume of the solution**arrow_forward2.70 Why do you think an inhibitor molecule is needed to induce the polymerization of ethylene?arrow_forwardThere are 41 = 4 mononucleotides of DNA, there are 42 = 16 possible dinucleotides, and so on. If a segment of DNA were completely random, how many nucleotides long would it need to be in order to have one possible sequence for each person on Earth (currently about 7.5 billion people)?arrow_forward
- 5 its asking which mutation it would be and to predict what it would look like..arrow_forwardThe DNA of bacteriophage lambda has 1.2 x 10^5 nucleotides. How many proteins of molecular weight 40,000 could be coded by this DNA? Assume a molecular weight of 100 for the average amino acid.arrow_forwardDNA Replication1) Construct the complementary strand of DNA (strand II) that would pair with the DNA strand I sequence below. Remember that nucleotides are always added in the 5 prime to 3 prime direction. The base pairing rules are: DNA DNAadenine (A) always bonds with thymine (T) thymine (T) always bonds with adenine (A) guanine (G) always bonds with cytosine (C) cytosine (C) always bonds with guanine (G) STRAND I: 3’ T A C T A T A G T C T G T C T C C C A C T 5’ STRAND II: 5’ 3’arrow_forward
- Section one. Match enzyme or molecule with its function in bacteria. Answers may be used more than once or not at all. DNA ligase d. tRNA promoter а. g. b. DNA helicase 1RNA h. terminator е. RNA polymerase f. DNA polymerase III i. stop codon с. 37. main enzyme in catalyzing DNA → mRNA transcription 38. unwinds double-stranded DNA at the replication fork 39. Where transcription stops 40. adds new DNA bases in the 5'→3’ direction 41. the "translator" molecule 42. contains codons 43. contains anti-codonsarrow_forwardWhy are Guanidins and Amidins such strong bases?arrow_forward1. Draw the two Watson-Crick Bases Pairs and show how G- C and A-T recognize each other by hydrogen bonding. 2. Draw the full structure of two nucleotides - one from DNA and one from RNA. Point out the key difference between the two. 3. Write out an example of a condensation reaction forming a phosphate-sugar bond. 4. Write out an example of a condensation reaction between a nucleic acid base and a sugar. 5. State the "Central Dogma" of biochemistry.arrow_forward
- Shown are the configuration of a gel electrophoresis experiment. Charged plates establish an electric field on a region of gel between the plates. A solution of DNA fragments, which acquire a negative charge in solution, is analyzed by using an electric field to drive the fragments through the gel. A drag force opposes the motion, causing the fragments to move at a constant speed. Larger fragments move more slowly, so the fragments separate by size to give a “fingerprint” of the sample. What is the energy transformation as the fragments move through the gel?A. Electric potential energy to kinetic energyB. Electric potential energy to thermal energyC. Electric potential energy to kinetic energy and thermal energyD. Kinetic energy to thermal energyarrow_forward1arrow_forward0 0=P-0-CH₂ 0- H 1) How would you designate this acid in a abbreviation ? 09-0-962 6- H 3) Provide -NH₂ one-letter AR # # 2) 15 this a strand of DNA of RNA? How do you know? an Ntl₂ 0=P-O-CH₂ A K он н abbreviation for the Strand of DNA WITH complementary bases to the one shown above. Indicate the '5' and 3 endsarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





