
Concept explainers
(a)
Interpretation:
The hydrocarbon B and the intermediate A (both with the formulas C11H10) in the following reaction sequence is to be identified. Compound B is formed spontaneously from A in a pericyclic reaction.
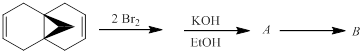
Concept introduction:
Commonly, electrocyclic reactions are a pericyclic reaction which occur intramolecularly. These reactions will result in the formation of ring compounds under the influence of heat or light. Notably, in this process one new sigma bond is formed and one old π-bond is consumed. Intriguingly, the reverse ring opening electrocyclic reaction can also be possible to occur under the same reaction mechanism but in reverse manner.
(b)
Interpretation:
The proton NMR spectrum of B consists of a complex absorption at δ 7.1 (8H) and a singlet at δ (-0.5) (2H).The negative absorption for protons is to be explained.
Concept introduction:
The usage of nuclear magnetic resonance in NMR spectroscopy of hydron nuclei is termed as proton nuclear magnetic resonance spectroscopy. This is used to determine the structure of molecules with respect to 1H found within the molecules. In order to avoid the interference from solvent molecules, the analysis of 1H spectra is usually carried out in deuterated solvents such as D2O, CDCl3, DMSO-d6.Besides, tetramethyl silane is used as an internal standard.
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
EBK ORGANIC CHEMISTRY STUDY GUIDE AND S
- (d) Ozonolysis of 3,4-dimethyl-3-hexene gives compound D. (i) Write the reaction step for the formation of D by including the appropriate reagents. (ii) The mass spectrum of compound D shows a fragment ion peak at m/z 57. Propose the structure of this fragment ion. (ii) Identify the more stable resonance of the proposed structure in (ii).arrow_forwardCompounds A and B are isomers having the molecular formula C4H8O3. Identify A and B on the basis of their 1H NMR spectra.Compound A: δ 1.3 (3H, triplet); 3.6 (2H, quartet); 4.1 (2H, singlet); 11.1 (1H, broad singlet)Compound B: δ 2.6 (2H, triplet); 3.4 (3H, singlet); 3.7 (2H triplet); 11.3 (1H, broad singlet)arrow_forwardThe treatment of (CH3)2C=CHCH2Br with H2O forms B (molecular formulaC5H10O) as one of the products. Determine the structure of B from its 1H NMR and IR spectra.arrow_forward
- The following sequence of steps converts (R)-2-octanol to (S)-2-octanol. Propose structural formulas for intermediates A and B, specify the configuration of each, and account for the inversion of configuration in this sequence.arrow_forwardPropose structures for molecules that fit the following descriptions:(a) An aldehyde with the formula C5H10O(b) An ester with the formula C6H12O2(c) A compound with the formula C3H7NOS that is both anamide and a thiolarrow_forward(a) The Friedel-Crafts reaction of benzene with 2-chloro-3-methylbutane in the presence of AlCl3 occurs with a carbocation rearrangement. Give mechanistic explanation and the product formed. (b) Predict the product(s) will be formed from the following reactions: (i) Bromination of p-methylbenzoic acid (ii) Sulphonation of m-bromoanisole (iii) Friedel-craft acylation of o-bromonitrobenzenearrow_forward
- Compounds W and X are isomers. They have the molecular formula C9H8O. The IR spectrum ofeach compound shows a strong absorption band near 1715 cm−1. Oxidation of either compound withhot, basic potassium permanganate followed by acidification yields phthalic acid. The proton NMRspectrum of W shows a multiplet at δ 7.3 and a singlet at δ 3.4. The proton NMR spectrum of Xshows a multiplet at δ 7.5, a triplet at δ 3.1, and a triplet at δ 2.5. Propose structures for W and X.arrow_forwardThere are several isomeric alcohols and ethers of molecular formula C5H12O. Two of these exhibit the following 1H-NMR spectra. Propose a structure for each of the isomers. Isomer A: δ = 0.92 (t, 7.8 Hz, 3 H), 1.20 (s, 6H), 1.49 (q, 7.8 Hz, 2H), 1.85 (s, 1H) ppm Isomer B: δ = 1.19 (s, 9 H), 3.21 (s, 3H) ppmarrow_forwardThe treatment of (CH3)2C=CHCH2Br with H2O forms B (molecular formula C5H10O) as one of the products. Determine the structure of B from its 1H NMR and IR spectra.arrow_forward
- The enamine prepared from acetone and dimethylamine is shown here in its lowest-energy form. (a) What is the geometry and hybridization of the nitrogen atom? (b) What orbital on nitrogen holds the lone pair of electrons? (c) What is the geometric relationship between the p orbitals of the double bond and the nitrogen orbital that holds the lone pair? Why do you think this geometry represents the minimum energy?arrow_forward(b) Compound C has the molecular formula of C-H12. The IR spectrum shows the important absorption bands at 2950, 2830 and 2250 cm-1 while the 'H NMR spectrum shows two singlets at dH 1.24 (9H) and ôH 1.80 (3H). Based on these spectral data, deduce the structure of compound C.arrow_forward(b) 3-methyl-2-butanol reacts with concentrated sulphuric acid to form 2-methyl-2- butene. Write the mechanism for the reaction.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

