
(a)
Interpretation:
The curved-arrow mechanism for the anionic oxyCope reaction of compound A is to be given and the stereoisomer B does not react under the same conditions-the statement is to be explained.
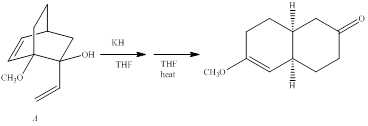
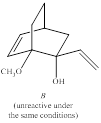
Concept introduction:
Generally, [3, 3] sigmatropic reactions of 1, 5-diences are called as cope reactions. When the diene is having the oxygen at C3 or C4 position the sigmatropic reaction is called as oxy cope reaction. Notably, when the alkoxide fragment is involved it is referred as anionic oxy cope reaction. Moreover, the enhanced increase in
(b)
Interpretation:
The structure of the product, including its stereochemistry expected from the anion oxyCope reaction of the following compound is to be given.

Concept introduction:
When the diene is having the oxygen at C3 or C4 position the sigmatropic reaction is called as oxy cope reaction. Notably, when the alkoxide fragment is involved it is referred as anionic oxy cope reaction.
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
EBK ORGANIC CHEMISTRY STUDY GUIDE AND S
- (a) Give chemical tests to distinguish between the following pairs of compounds :(i) Pentan-2-ol and Pentan-3-ol (ii) Methanol and Phenol(b) o-nitro phenol is more acidic than o-methoxy phenol. Explain why.arrow_forwardWrite the structure of the major organic product formed in the reaction of 1-pentene with each of the following: (a) Hydrogen chloride (b) Dilute sulfuric acid (c) Diborane in diglyme, followed by basic hydrogen peroxide (d) Bromine in carbon tetrachloride (e) Bromine in water (f) Peroxyacetic acid (g) Ozone (h) Product of part (g) treated with zinc and water (i) Product of part (g) treated with dimethyl sulfide (CH3)2Sarrow_forwardGive the structure of compound A.arrow_forward
- Explain why :(a) The dipole moment of chlorobenzene is lower than that of cyclohexyl chloride.(b) Alkyl halides, though polar, are immiscible with water.arrow_forwardExplain the following behaviours :(i) Alcohols are more soluble in water than the hydrocarbons of comparable molecular masses.(ii) Ortho-nitrophenol is more acidic than ortho-methoxyphenol.arrow_forward11:43 Q1. (a) (c) (d) (b) Two stereoisomers of but-2-ene are formed when 2-bromobutane reacts with ethanolic potassium hydroxide. (i) Explain what is meant by the term stereoisomers. Library Name and outline a mechanism for the reaction of 2-bromo-2-methylpropane with ethanolic potassium hydroxide to form the alkene 2-methylpropene, (CH3)2C=CH₂ Name of mechanism Mechanism (ii) Draw the structures and give the names of the two stereoisomers of but-2-ene. Stereoisomer 1 Name (iii) Name this type of stereoisomerism. Select Name Stereoisomer 2 When 2-bromo-2-methylpropane reacts with aqueous potassium hydroxide, 2-methylpropan-2-ol is formed as shown by the following equation. CH3 H₂C-C-CH3 + KOH Br Page 2 of 14 CH3 H3C-C-CH3 + KBr ОН State the role of the hydroxide ions in this reaction. Write an equation for the reaction that occurs when CH3CH₂CH₂CH₂Br reacts with an excess of ammonia. Name the organic product of this reaction. Equation Name of product 9,284 Photos, 1,166 Videos For You…arrow_forward
- Deduce the structure of compound C.arrow_forward(a) The Friedel-Crafts reaction of benzene with 2-chloro-3-methylbutane in the presence of AlCl3 occurs with a carbocation rearrangement. Give mechanistic explanation and the product formed. (b) Predict the product(s) will be formed from the following reactions: (i) Bromination of p-methylbenzoic acid (ii) Sulphonation of m-bromoanisole (iii) Friedel-craft acylation of o-bromonitrobenzenearrow_forward(a) Account for the following :(i) Propanal is more reactive than propanone towards nucleophilic reagents.(ii) Electrophilic substitution in benzoic acid takes place at meta position.(iii) Carboxylic acids do not give characteristic reactions of carbonyl group.(b) Give simple chemical test to distinguish between the following pairs of compounds:(i) Acetophenone and benzaldehyde(ii) Benzoic acid and ethylbenzoate.arrow_forward
- (a) (b) (c) Suggest a synthesis of the following alkene (A) using a Wittig reaction strategy. Draw the starting material(s), key reagent and a full reaction mechanism including an explanation of the observed geometry. Which of the following (B) and (C) will favour the enol form? Briefly explain your reasoning. Predict the product(s) and provide a mechanism for each of the following transformations: (i) (ii) OMe OMe Base OEt NaOEtarrow_forwardGive reasons :(a) n-Butyl bromide has higher boiling point than f-butyl bromide.(b) Racemic mixture is optically inactive.(c) The presence of nitro group (-NO2) at o/p positions increases the reactivity of haloarenes towards nucleophilic substitution reactions.arrow_forward(b) Suggest a reasonable biosynthesis for the naturally occurring alkaloid coniine (isolated from hemlock), starting from octanoic acid. Coniinearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





