
Concept explainers
(a)
Interpretation: The products formed by the treatment of
Concept introduction: The
Answer to Problem 28.20P
The product formed by the treatment of
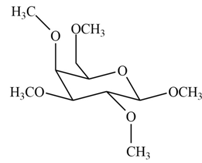
Explanation of Solution
The
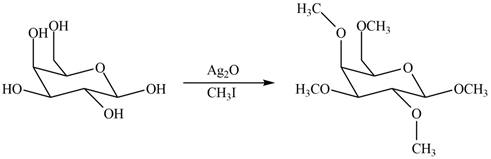
Figure 1
The products formed by the treatment of
(b)
Interpretation: The products formed by the treatment of
Concept introduction: The
Answer to Problem 28.20P
The product formed by the treatment of
Explanation of Solution
The
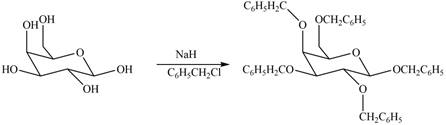
Figure 2
The products formed by the treatment of
(c)
Interpretation: The products formed by the treatment of
Concept introduction: The characteristic bond of glycoside is
Answer to Problem 28.20P
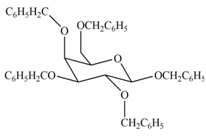
The products formed by the treatment of
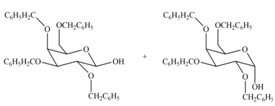
Explanation of Solution
The characteristic bond of glycoside is
The products formed by the treatment of
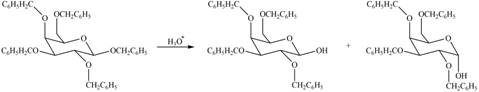
Figure 3
The products formed by the treatment of
(d)
Interpretation: The products formed by the treatment of
Concept introduction: The
Answer to Problem 28.20P
The products formed by the treatment of
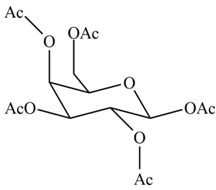
Explanation of Solution
The
The products formed by the treatment of
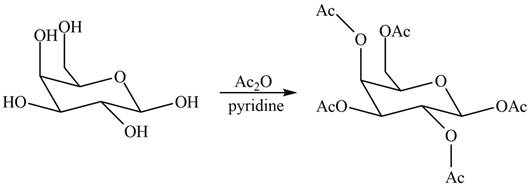
Figure 4
The products formed by the treatment of
(e)
Interpretation: The products formed by the treatment of D- galactose with
Concept introduction: The
Answer to Problem 28.20P
The product formed by the treatment of
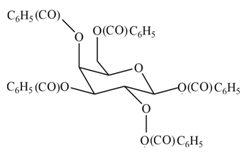
Explanation of Solution
The
The products formed by the treatment of
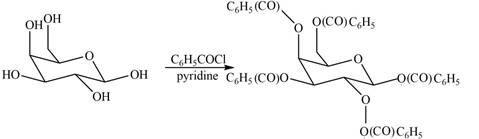
Figure 5
The products formed by the treatment of
(f)
Interpretation: The products formed by the treatment of
Concept introduction: The
Answer to Problem 28.20P
The products formed by the treatment of
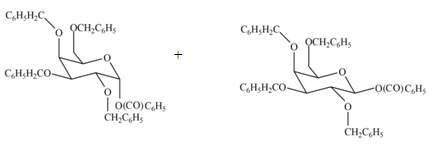
Explanation of Solution
The
The products formed by the treatment of
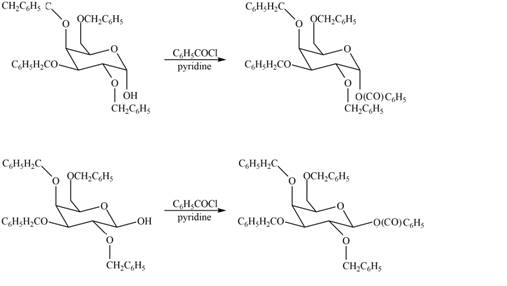
Figure 6
The products formed by the treatment of
Want to see more full solutions like this?
Chapter 28 Solutions
Organic Chemistry
- D-Arabinose can exist in both pyranose and furanose forms.a. Draw the a and ß anomers of D-arabinofuranose.b. Draw the a and ß anomers of D-arabinopyranosearrow_forward1. Classify the molecule shown according to the location of its carbonyl group and the number of carbon atoms. CH₂OH C=O PIL a. Aldotriose b. Aldotetrose Aldopentose d., Ketotriose Ketotetrose HO-C-H L CH₂OHarrow_forward10. Draw the reaction of Galactose with Benedict solution. Answer the following questions based on these structures. Here is the terminology you can use to describe the relationships. Structural isomers, Enantiomers, Diastereomers, and no relationship. CHO CHO -H H- Н- CHO HO-H OH H- НО- Н- Н- OH OH CH₂OH CHO H H -ОН -ОН но- Н- CH₂OH H -ОН CH2OH 2 Н- Н- но- НО- CHO -ОН -ОН -H -H CH2OH 8 НО Н- H- НО- -ОН -Н CH₂OH 3 CH₂OH 7 11. What is the relationship between 1 and 4? 12. What is the relationship between 1 and 3? 13. What is the relationship between 1 and 5? 14. What is the relationship between 2 and 9? Н- но 15. What is the relationship between 4 and 10? 16. What is the relationship between 5 and 6? ОН 17. What is the relationship between 2 and 10? CH2OH -ОН -Н CH2OH 9 НО- Н- Н- CH2OH :0 -H OH OH CH2OH 4 НО- НО- Н- H- но- но- CH2OH H——OH -Н -Н CH₂OH 10 H H OH OH CH₂OH 5 Н- но- но- Н- CHO -OH -H H -OH CH2OH 6arrow_forward
- + Mannose is a monosaccharide involved in the glycosylation of certain proteins. Several genetic disorders are associated with enzymes involved in mannose metabolism. Monosaccharides form a new functional group when the straight chain sugar cyclizes. What is the type of functional group formed in mannose after it undergoes cyclization to form the furanose ring? HO OH OH 0- OH OH A) aldehyde B) ketone C) hemi-acetal D) ether E) esterarrow_forward2. Sucrose is a disaccharide formed from the condensation of glucose and fructose sugars. Draw the structure of sucrose that results from the condensation reaction between the indicated -OH groups. CH2OH 1, CH,OH он 1. OH но CH,OH OH HO H он OHarrow_forwardWhat hydrolysis products are formed when each wax is treated with aqueous sulfuric acid? a. CH 3(CH 2) 18COO(CH 2) 29CH 3b. CH 3(CH 2) 24COO(CH 2) 23CH 3c. CH 3(CH 2) 14COO(CH 2) 17CH 3arrow_forward
- Which structure of Erythrose is referred to by the given Fischer projection? A. L-Erythrose B. D-Erythrose но- -H HO H CH2OHarrow_forwardDetermine the product formed when butanone (CH3CH,COCH3) Is treated with each reagent. If no reaction occurs, label the reaction with "no reaction". PCC H2 Pd-C HO OH NazCr,O, H,SO.H,O [1] LIAIH, [2] H,O no reaction Ag20 NH,OH NABH, CH;OH Resetarrow_forwardDraw the structure of each type of compound. a. a D-aldotriose b. an L-ketohexose c. a four-carbon aldonic acidarrow_forward
- 9. Melibiose is a disaccharide that is 30 times sweeter than sucrose. a. What are the monosaccharide units in melibiose? HO H b. What type of glycosidic bond links the monosaccharides? c. Identify the structure as a- or 6-melibiose. CH₂OH O H OH H H OH H O–CH2 H HO H OH H Melibiose O H OH H OHarrow_forwardDraw the products formed when B-D-galactose is treated with each reagent. a. Ag,0 + CH3I b. NaH + CgHsCH2CI c. The product in (b), then H30* d. Ac20 + pyridine e. CeHsCOCI + pyridine f. The product in (c), then CgHsCOCI + pyridinearrow_forward16.60 Draw the condensed structural formulas for the products from each of the following reactions: (16.2, 16.3) a. CH3-C-OH + NAOH → b. CH3-C-OH + H,O 2 ОН + КОНarrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
