
(a)
Interpretation:
Chair conformations and the position of substituents need to be identified for the given set of compounds.
Concept introduction:
Ring-flipping is a phenomenon known as ring inversion that involves rotation about the single bonds of cyclic conformers. Usually this happens in cyclohexane ring. When ring flip happens in the cyclohexane, the axially and equatorially substituted groups are inverted. The ring-flipping also happens in a fused ring system. Decalin is a fused ring system. There are two stereoisomers for decalin, namely cis-decalin and trans-decalin. If the substituent is facing away from the ring or which makes
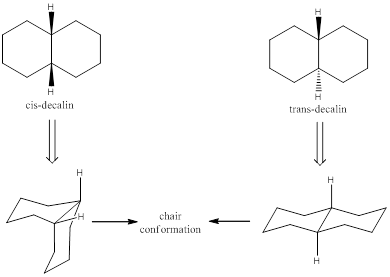
When viewing the molecule from the top the bonds which are said to be in parallel with the viewing angle are in axial position and which are not parallel are known to be in equatorial position.
To draw and explain: chair conformation and identify the substituent position.
(b)
Interpretation:
Chair conformations and the position of substituents need to be identified for the given set of compounds.
Concept introduction:
Ring-flipping is a phenomenon known as ring inversion that involves rotation about the single bonds of cyclic conformers. Usually this happens in cyclohexane ring. When ring flip happens in the cyclohexane, the axially and equatorially substituted groups are inverted. The ring-flipping also happens in a fused ring system. Decalin is a fused ring system. There are two stereoisomers for decalin, namely cis-decalin and trans-decalin. If the substituent is facing away from the ring or which makes

When viewing the molecule from the top the bonds which are said to be in parallel with the viewing angle are in axial position and which are not parallel are known to be in equatorial position.
To draw and explain: chair conformation and identify the substituent position.
(c)
Interpretation:
Chair conformations and the position of substituents need to be identified for the given set of compounds.
Concept introduction:
Ring-flipping is a phenomenon known as ring inversion that involves rotation about the single bonds of cyclic conformers. Usually this happens in cyclohexane ring. When ring flip happens in the cyclohexane, the axially and equatorially substituted groups are inverted. The ring-flipping also happens in a fused ring system. Decalin is a fused ring system. There are two stereoisomers for decalin, namely cis-decalin and trans-decalin. If the substituent is facing away from the ring or which makes

When viewing the molecule from the top the bonds which are said to be in parallel with the viewing angle are in axial position and which are not parallel are known to be in equatorial position.
To draw and explain: chair conformation and identify the substituent position.
Want to see the full answer?
Check out a sample textbook solution
Chapter 26 Solutions
ORG. CHEM. TEXT/STUDY GUIDE/EBOOK (LL)
- 4. Propose a synthesis of the target molecules from the respective starting materials. a) b) LUCH C Br OHarrow_forwardThe following mechanism for the gas phase reaction of H2 and ICI that is consistent with the observed rate law is: step 1 step 2 slow: H2(g) +ICI(g) → HCl(g) + HI(g) fast: ICI(g) + HI(g) → HCl(g) + |2(g) (1) What is the equation for the overall reaction? Use the smallest integer coefficients possible. If a box is not needed, leave it blank. + → + (2) Which species acts as a catalyst? Enter formula. If none, leave box blank: (3) Which species acts as a reaction intermediate? Enter formula. If none, leave box blank: (4) Complete the rate law for the overall reaction that is consistent with this mechanism. (Use the form k[A][B]"..., where '1' is understood (so don't write it) for m, n etc.) Rate =arrow_forwardPlease correct answer and don't use hand rating and don't use Ai solutionarrow_forward
- 1. For each of the following statements, indicate whether they are true of false. ⚫ the terms primary, secondary and tertiary have different meanings when applied to amines than they do when applied to alcohols. • a tertiary amine is one that is bonded to a tertiary carbon atom (one with three C atoms bonded to it). • simple five-membered heteroaromatic compounds (e.g. pyrrole) are typically more electron rich than benzene. ⚫ simple six-membered heteroaromatic compounds (e.g. pyridine) are typically more electron rich than benzene. • pyrrole is very weakly basic because protonation anywhere on the ring disrupts the aromaticity. • thiophene is more reactive than benzene toward electrophilic aromatic substitution. • pyridine is more reactive than nitrobenzene toward electrophilic aromatic substitution. • the lone pair on the nitrogen atom of pyridine is part of the pi system.arrow_forwardThe following reactions are NOT ordered in the way in which they occur. Reaction 1 PhO-OPh Reaction 2 Ph-O -CH₂ heat 2 *OPh Pho -CH2 Reaction 3 Ph-O ⚫OPh + -CH₂ Reaction 4 Pho Pho + H₂C OPh + CHOPh H₂C -CH₂ Reactions 1 and 3 Reaction 2 O Reaction 3 ○ Reactions 3 and 4 ○ Reactions 1 and 2 Reaction 4 ○ Reaction 1arrow_forwardSelect all possible products from the following reaction: NaOH H₂O a) b) ОН HO O HO HO e) ОН f) O HO g) h) + OHarrow_forward
- 3. Draw diagrams to represent the conjugation in these molecules. Draw two types of diagram: a. Show curly arrows linking at least two different ways of representing the molecule b. Indicate with dotted lines and partial charges (where necessary) the partial double bond (and charge) distribution H₂N* H₂N -NH2arrow_forwardQuestion 2 of 25 point Question Attempt 3 of Ulimited Draw the structure for 3-chloro-4-ethylheptane. Part 2 of 3 Click and drag to start drawing a structure. Draw the structure for 1-chloro-4-ethyl-3-lodooctane. Click and drag to start drawing a structure. X G X B c Part 3 of 30 Draw the structure for (R)-2-chlorobutane. Include the stereochemistry at all stereogenic centers. Check Click and drag to start drawing a structure. G X A 。 MacBook Pro G P Save For Later Submit Assignment Privacyarrow_forwardPlease correct answer and don't used hand raitingarrow_forward
- In a silicon and aluminum alloy, with 12.6% silicon, what are the approximate percentages of the phases present in the constituent that is formed at the end of solidification? Temperature (°C) 1500 1000 L B+L 1415- α+L 577' 500 1.65 12.6 99.83 α+B B 0 Al 20 40 60 Weight percent silicon 80 Siarrow_forwardPlease correct answer and don't used hand raitingarrow_forwardPlease correct answer and don't used hand raitingarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





