
Concept explainers
Biaryls, compounds containing two
a.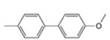 b.
b. 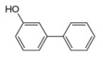 c.
c. 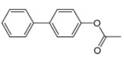
(a)
Interpretation: The synthesis of given biaryl compound using benzene as the starting material and any required organic or inorganic reagents is to be stated, and if more than one route is possible, then both routes are to be drawn.
Concept introduction: The treatment of an organic halide
Answer to Problem 26.47P
The synthesis of given biaryl compound (two routes) using benzene as the starting material and any required organic or inorganic reagents are shown below.
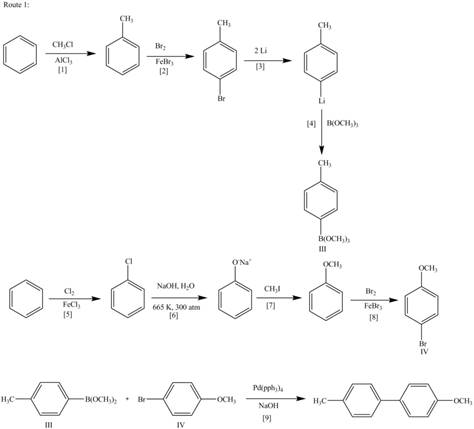
Explanation of Solution
The treatment of an organic halide
The retrosynthetic analysis of the given compound is,
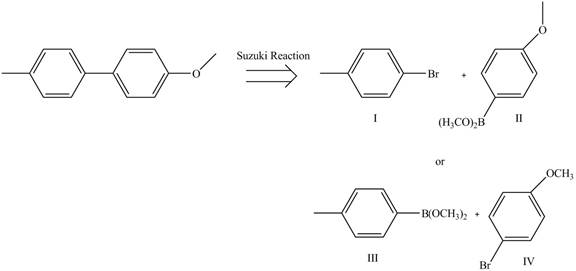
Figure 1
The above analysis shows that there are two possible routes to synthesize the given compound.
Route 1:
The Route 1 involves formation of desired biaryl in nine steps. The first step is Friedel-Crafts alkylation of benzene with
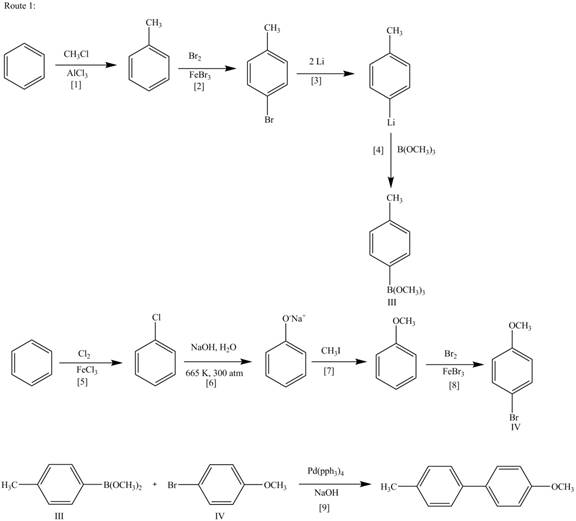
Figure 2
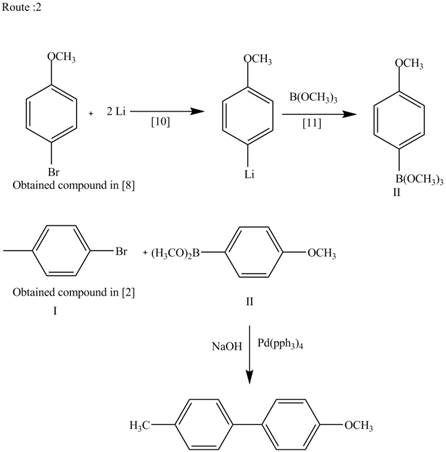
Route 2:
The Route 2 involves
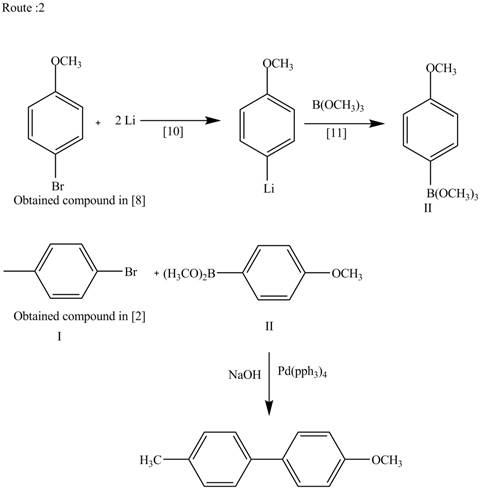
Figure 3
The synthesis of given biaryl compound (two routes) using benzene as the starting material and any required organic or inorganic reagents are shown in Figure 2, and Figure 3.
(b)
Interpretation: The synthesis of given biaryl compound using benzene as the starting material and any required organic or inorganic reagents is to be stated, and if more than one route is possible, then both routes are to be drawn.
Concept introduction: The treatment of an organic halide
Answer to Problem 26.47P
The synthesis of given biaryl compound (two routes) using benzene as the starting material and any required organic or inorganic reagents are shown below.
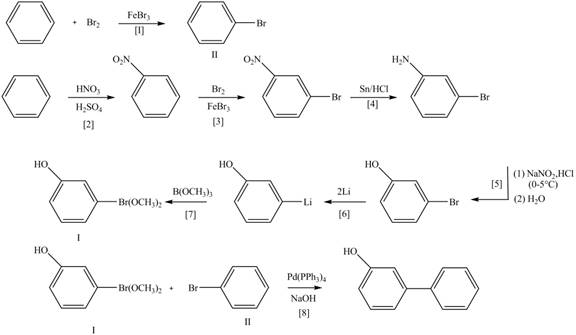
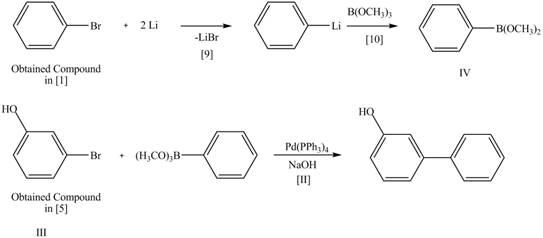
Explanation of Solution
The treatment of an organic halide
The retrosynthetic analysis of the given compound is,
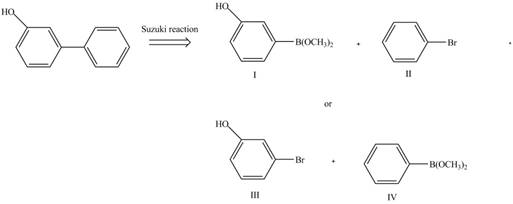
Figure 4
The above analysis shows that there are two possible routes to synthesize the given compound.
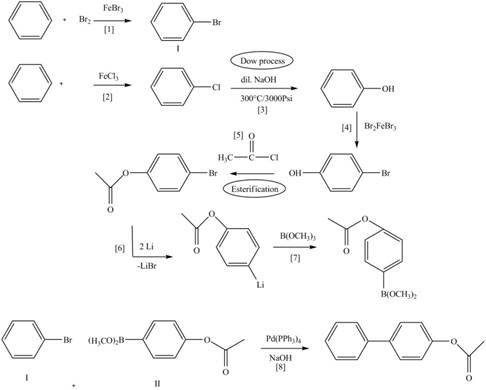
Route 1:
The Route 1 involves eight steps. The first step is bromination of benzene in the presence of
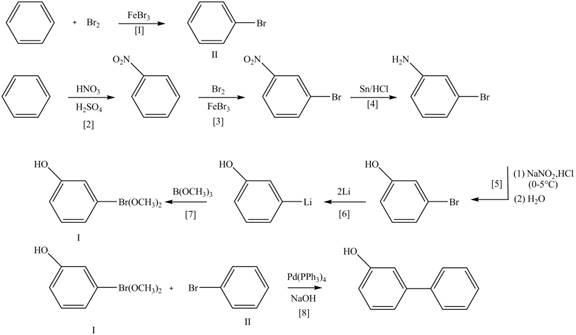
Figure 5
Route 2:
The Route 2 involves
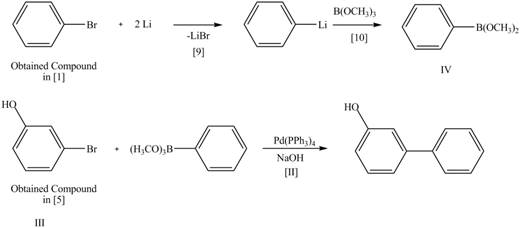
Figure 6
The synthesis of given biaryl compound (two routes) using benzene as the starting material and any required organic or inorganic reagents are shown in Figure 5, and Figure 6.
(c)
Interpretation: The synthesis of given biaryl compound using benzene as the starting material and any required organic or inorganic reagents is to be stated, and if more than one route is possible, then both routes are to be drawn.
Concept introduction: The treatment of an organic halide
Answer to Problem 26.47P
The synthesis of given biaryl compound (two routes) using benzene as the starting material and any required organic or inorganic reagents are shown below.
Explanation of Solution
The treatment of an organic halide
The retrosynthetic analysis of the given compound is,
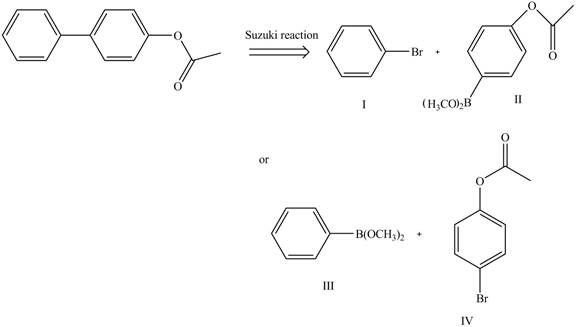
Figure 7
The above analysis shows that there are two possible routes to synthesize the given compound.
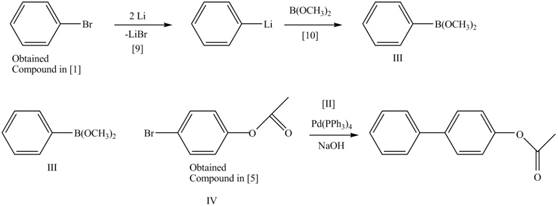
Route 1:
The Route 1 involves eight steps. The first step is bromination of benzene in the presence of
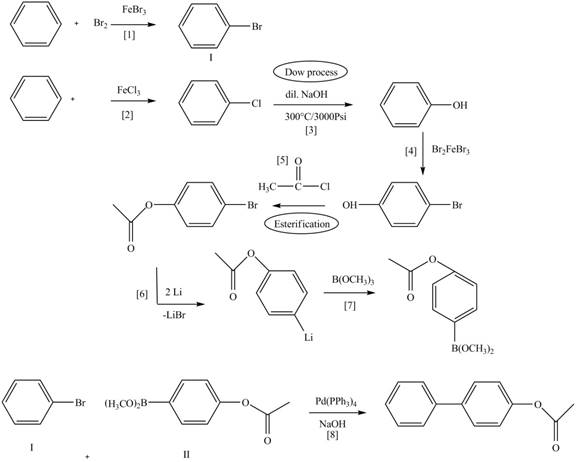
Figure 8
Route 2:
The Route 2 involves
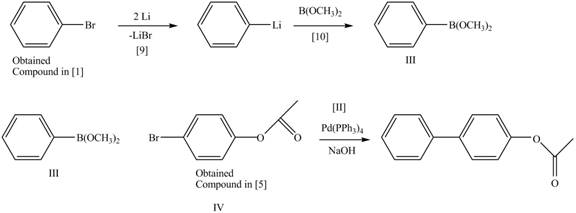
Figure 9
The synthesis of given biaryl compound (two routes) using benzene as the starting material and any required organic or inorganic reagents are shown in Figure 8, and Figure 9.
Want to see more full solutions like this?
Chapter 26 Solutions
Package: Loose Leaf for Organic Chemistry with Biological Topics with Connect Access Card
- A problem often encountered in the oxidation of primary alcohols to acids is that esters are sometimes produced as by-products. For example, oxidation of ethanol yields acetic acid and ethyl acetate: Propose a mechanism to account for the formation of ethyl acetate. Take into account the reversible reaction between aldehydes and alcohols:arrow_forwardEnamines can serve as enolate surrogates in reactions at the a-carbon. In the reaction sequence, the structures of the enamine addition product – the initial zwitterion and its neutral tautomer – are shown. Draw the structures of the two reactants forming these intermediates, and draw the structure of the final product, obtained via hydrolysis of the neutral intermediate. initial zwitterionic intermediate neutral intermediate tautomerization Reactants H,0 hydrolysis product Draw the two reactants. Draw the hydrolysis product. Select Draw Rings More Erase Select Draw Rings More Erase H Harrow_forwardElectrophilic aromatic substitution usually occurs at the 1-position of naphthalene, also called the a position. Predict the major products of the reactions of naphthalene with fuming sulfuric acid.arrow_forward
- Draw the principal organic product for the reaction of 1-bromopentane with lithium in diethyl ether, followed by formaldehyde in diethyl ether, and then followed by dilute acid.arrow_forwardWhich of the following statements is correct? A) The haloform reaction proceeds under very difficult conditions and the yield is very low. B) The haloform reaction is used only for the identification of compounds containing secondary alcohol groups. C) Methyl ketones or alcohols are oxidized with halogens in acidic solutions to give carboxylic acids and the appropriate haloform product. D) Enolate anions react rapidly with halogens to give alpha-halocarbonyl compounds.arrow_forwardTwo methods convert an alkyl halide to a carboxylic acid having one more carbon atom. Depending on the structure of the alkyl halide, one or both of these methods may be employed. For each alkyl halide, write out a stepwise sequence that converts it to a carboxylic acid with one more carbon atom. If both methods work, draw both routes. If one method cannot be used, state why it can't.arrow_forward
- Draw the tautomer of this enol. Include all lone pairs. Ignore inorganic byproducts. :OH: :0: H3O+ Draw Tautomerarrow_forwardWhich reagent(s), if any, may be used to carry out the following reactionarrow_forwardConvert phenylacetylene to butyl benzene using any necessary organic and inorganic reagents. It will require multiple steps.arrow_forward
- Enamines can serve as enolate surrogates in reactions at the a-carbon. In the following reaction sequence, the structures of the enamine addition product – the initial zwitterion and its neutral tautomer – are shown. Draw the structures of the two reactants forming these intermediates, and draw the structure of the final product, obtained via hydrolysis the neutral intermediate. reactants initial zwitterionic intermediate tautomerization hydrolysis product neutral intermediate NH н,о Н,о ↑arrow_forwardChemistry Please help explain this textbook question: Although N, N -dimethylaniline is extremely reactive toward electrophilic aromatic substitution and is readily substituted by weak electrophiles, such as diazonium and nitronium ions, this reactivity is greatly diminished by the introduction of an alkyl substituent in an ortho position.arrow_forwardDraw the structure(s) of the major organic product(s) from the following reactionsarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

