
Concept explainers
(a)
Interpretation: To determine the functional group change that occurs in step 1 of a turn of the
Concept introduction: The
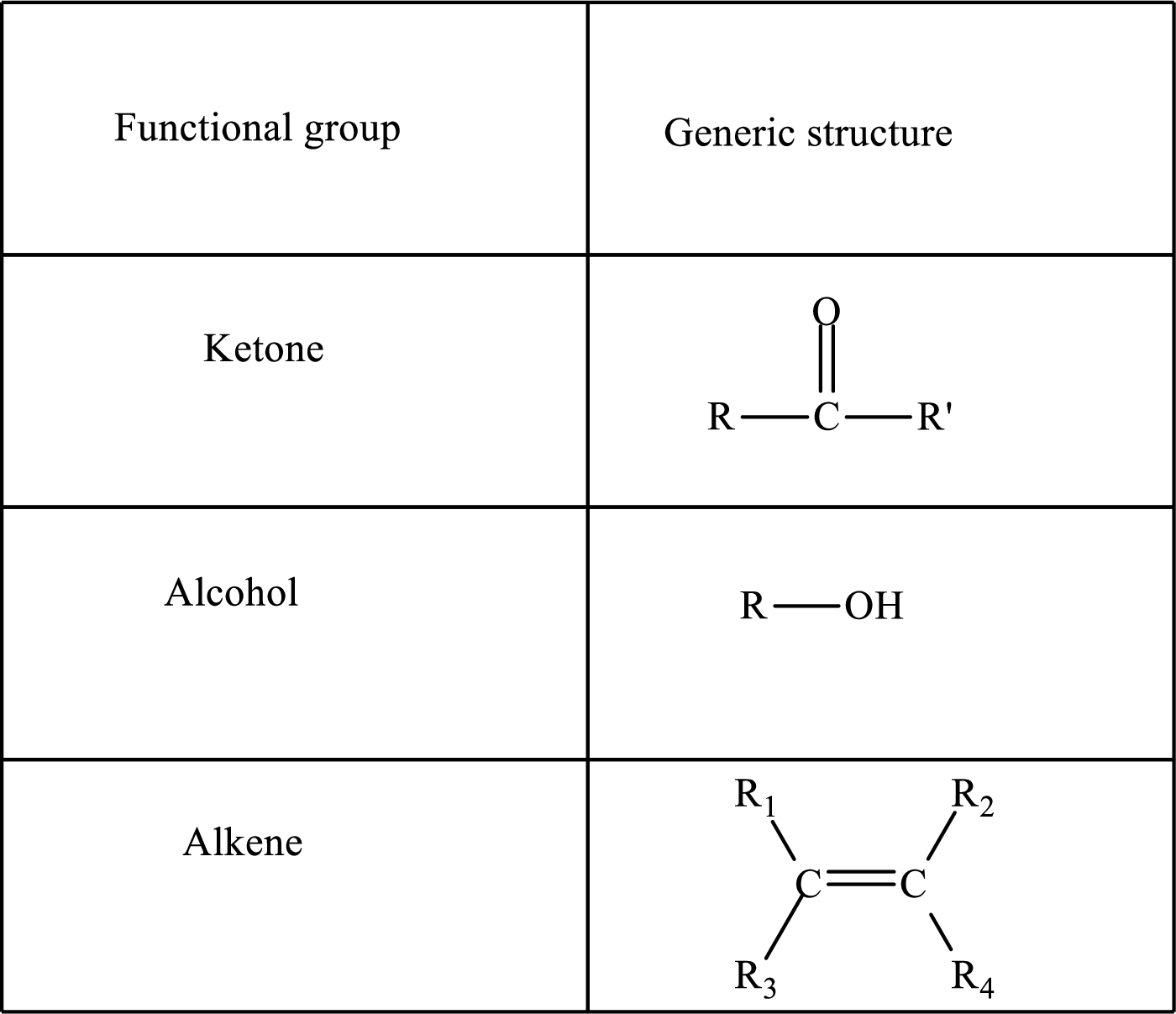
Here, R and
Alkanes are saturated hydrocarbons that contain covalently bonded hydrogen and carbon atoms.
(a)
Answer to Problem 25.29EP
In step 1 of a turn of the
Explanation of Solution
The reaction in step 1 of a turn of the
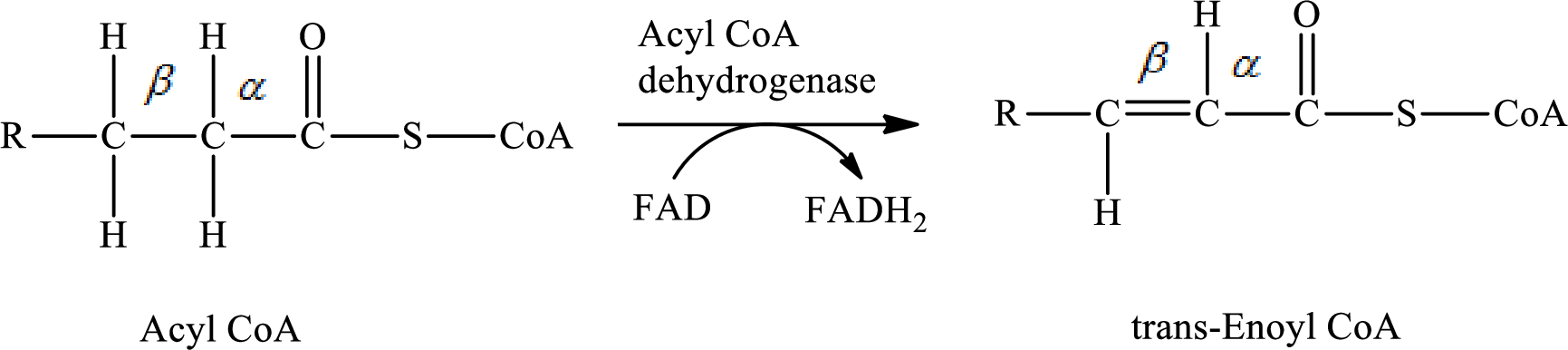
Therefore,
Carbon atoms are bonded with other carbon atoms by a single covalent bond in
(b)
Interpretation: To determine the functional group change that occurs in step 2 of a turn of the
Concept introduction: The
Functional groups are defined as the group of atoms which are attached to the carbon backbone of organic compounds. These are generally heteroatoms which are attached to the parent hydrocarbon chain. Some examples of functional groups are as follows:
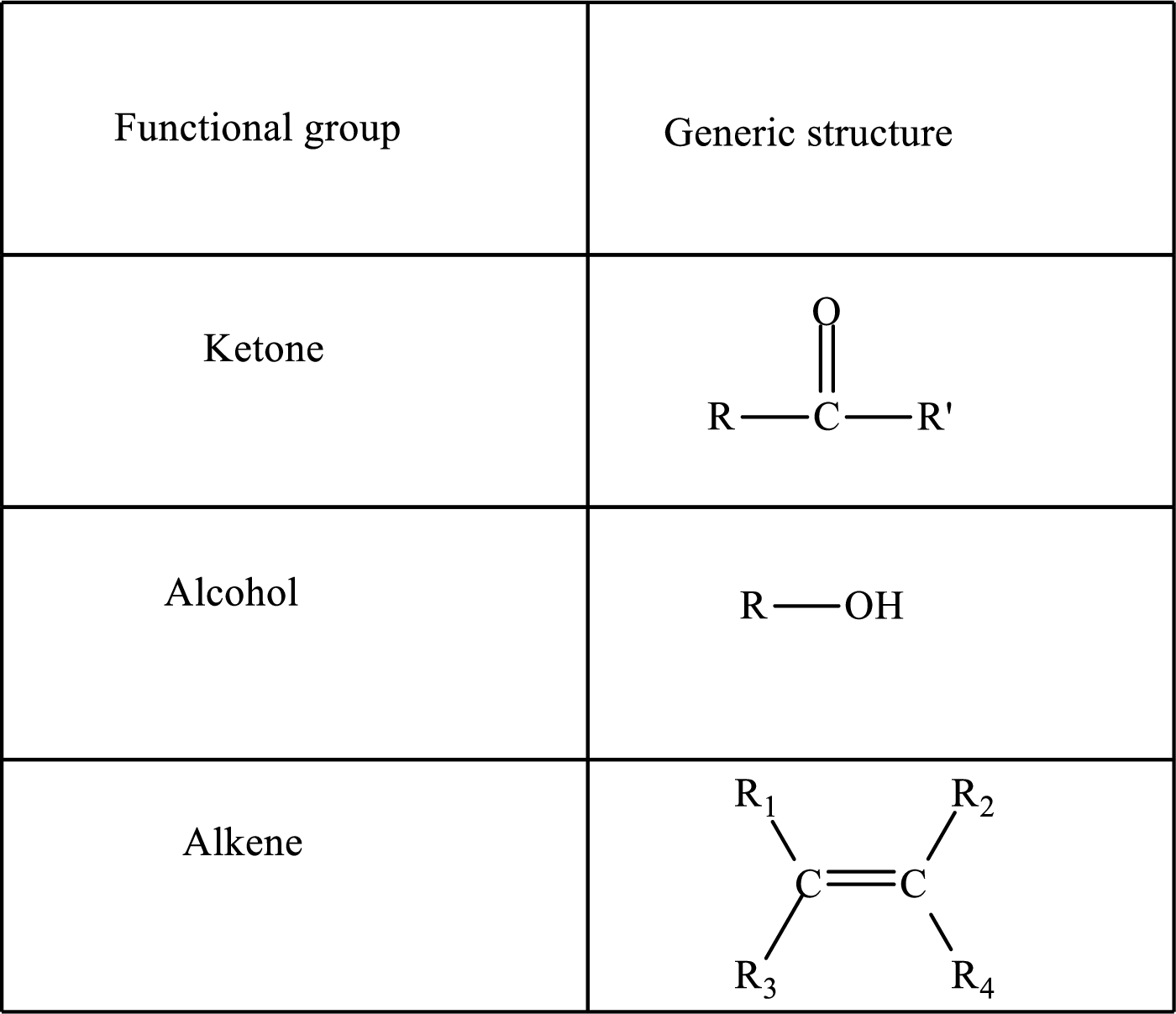
Here, R and
Alkanes are saturated hydrocarbons that contain covalently bonded hydrogen and carbon atoms. In secondary alcohol, the carbon atom of the hydroxyl group
(b)
Answer to Problem 25.29EP
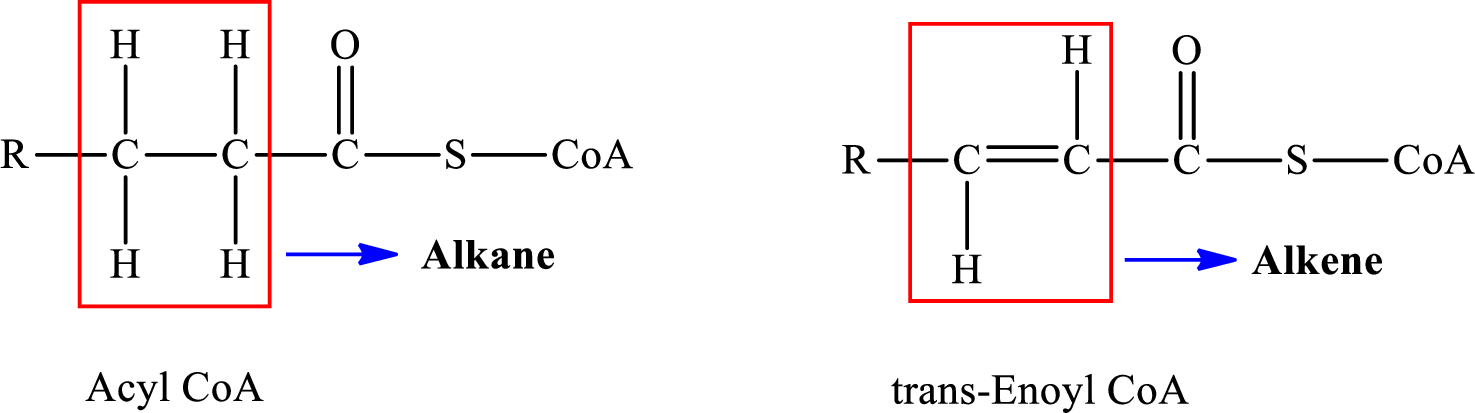
In step 2 of a turn of the
Explanation of Solution
The reaction in step 2 of a turn of the
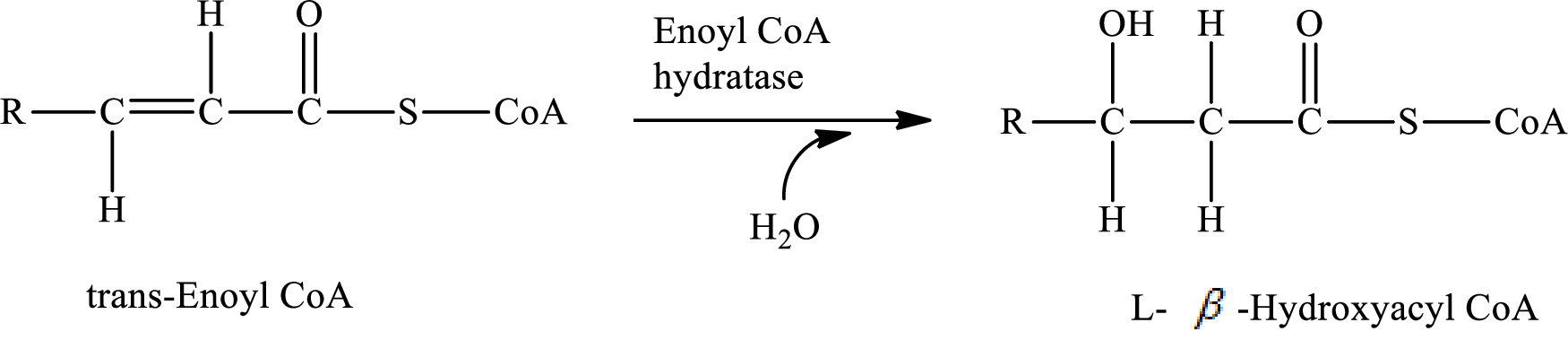
Therefore,
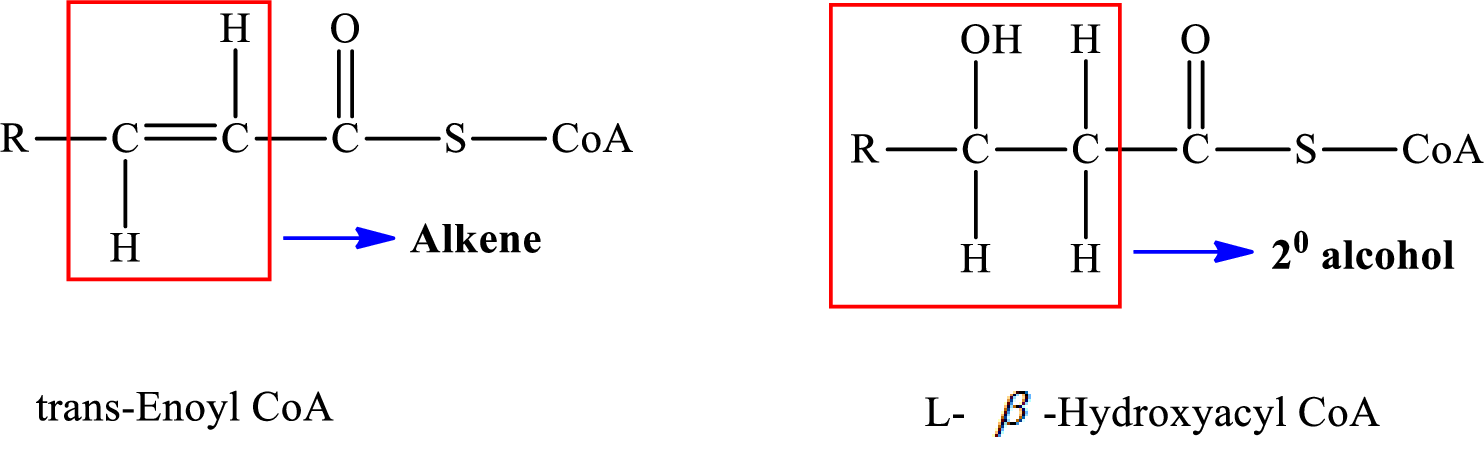
A double bond is present in
(c)
Interpretation: To determine the functional group change that occurs in step 3 of a turn of the
Concept introduction: The
Functional groups are defined as the group of atoms which are attached to the carbon backbone of organic compounds. These are generally heteroatoms which are attached to the parent hydrocarbon chain. Some examples of functional groups are as follows:
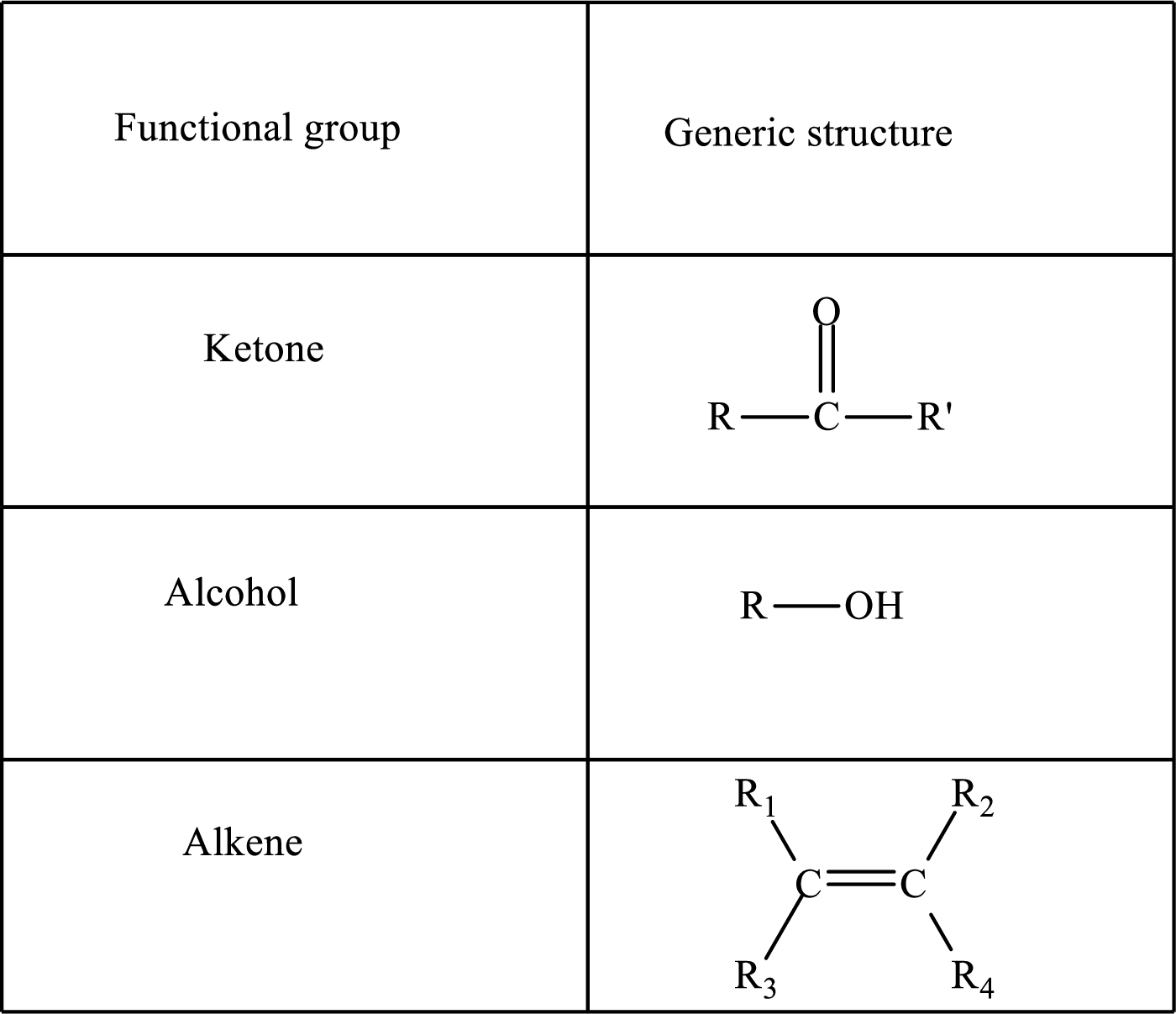
Here, R and
Alkanes are saturated hydrocarbons that contain covalently bonded hydrogen and carbon atoms. In secondary alcohol, the carbon atom of the hydroxyl group
(c)
Answer to Problem 25.29EP
In step 3 of a turn of the
Explanation of Solution
The reaction in step 3 of a turn of the
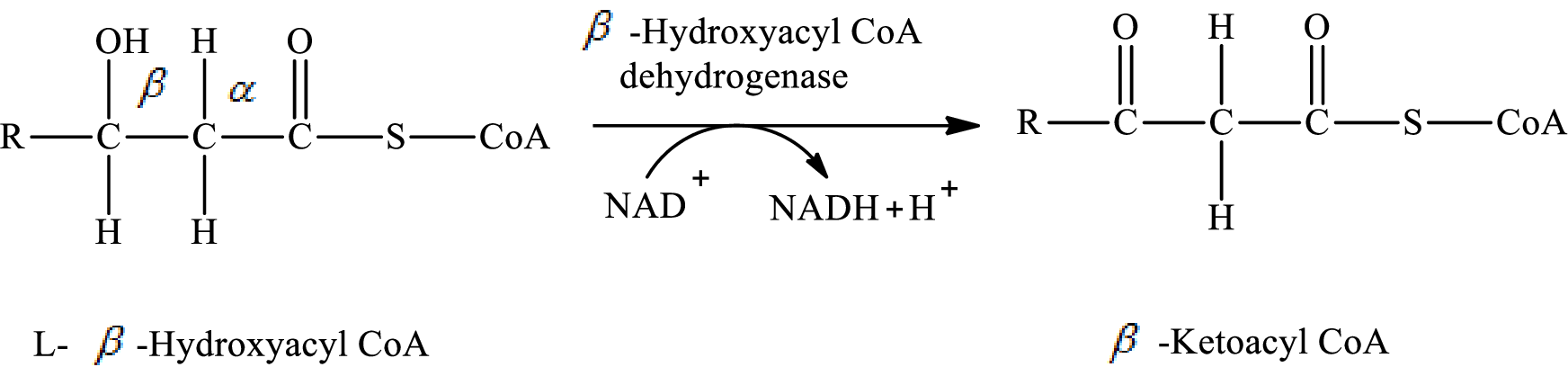
Therefore,
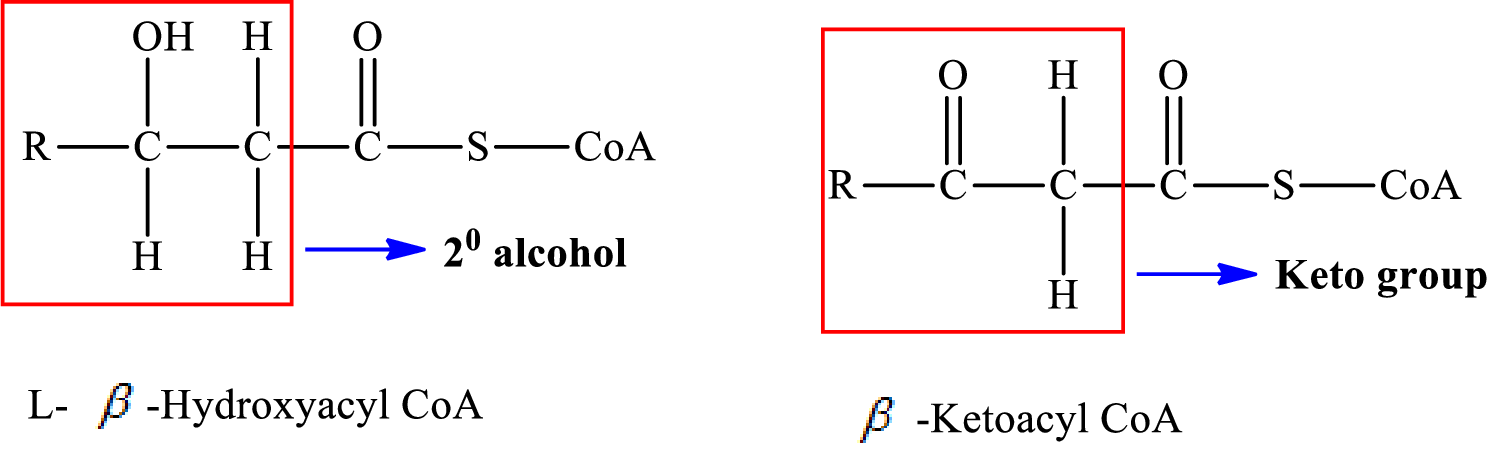
In
Want to see more full solutions like this?
Chapter 25 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- What type of inhibition is observed from the shift of the Lineweaver-Burke plot ahown in the graph below where the solid Ine represents the uninhibited enzymatic reaction while the broken line represents the inhibited enzymatic reaction? A. Irreveraible inhíbítion B. Noncompetitive inhíbition C. Competitive inhíbition D. Uncompetitive inhibition Potassium cyanide ia a polson which combines with cytochrome A3 to prevent binding of oxygen to the enzyme without altering the Km of the reaction with respect to reduced cytochrome c. Which type of inhibition does this represent? A. Irreveraible inhibition c. Competitive inhibition D. Uncompetitive inhibition B. Noncompetitive inhibition _10. Which of the following enzyme classes catalyze reactions in which two molecules become dissociated from each other? A. Kinase в. Нуdrolase c. Isomerase D. Ligase Which of the following enzyme classes catalyze reactions in which two molecules become covalently linked to each other? A. Kinase B. Hydrolase…arrow_forwardHow to differentiate the following simply by seeing the reaction: a. oxidoreductase b. transferase c. hydrolase d. lyase e. isomerase f. ligasearrow_forwardWhich of the following is a degradation product of both cytidine and deoxycytidine? a. β-alanine b. β-aminoisobutyrate c. succinyl-CoA d. uric acidarrow_forward
- Match each reaction description to the type of enzyme that catalyzes the reaction. 1. Oxidation and reduction of compounds 2. Transfers a functional group from one compound to another compound 3. Utilizes water to break bonds within a compound 4. Addition/removal of a group of atoms and bonds within a compound 5. Forms a bond between two compounds A. Ligase B. Transferase C. Hydrolase D. Oxidoreductase E. Isomerase F. Lyasearrow_forwardWhat intermediate in purine synthesis will accumulate if a strain of bacteria is lacking each of the following biochemicals? a. Aspartate b. Tetrahydrofolate c. Glycine d. Glutaminearrow_forwardAlkaptonuria is a rare organic aciduria involving a deficiency in homogentistic acid oxidase resulting in accumulation of homogentisic acid which is an intermediate in the degradative pathway of which of the following amino acids? a. Threonine b. Alanine c. Tyrosine d. Valinearrow_forward
- The diagram below shows an outline of the aminotransferase mechanism that skips the specific steps that show how electrons flow when a Schiff base is formed or is hydrolyzed. Using the mechanistic details given below A. Draw the mechanism for Enzyme- PLP Schiff base formation using pyridoxal and the lysine amine group using arrow to indicate electron flow. B.Draw the mechanism for hydrolysis of the Schiff base to form the a-keto acid, which is the reverse of the first reaction.arrow_forwardWhich of the following best explains why enzyme catalysis is affected by a change in pH? A. Change in pH alters ionization states of serine in the active site involved in nucleophilic catalysis B. The ionization states of his, asp and glu involved in acid/base catalysis are altered with change in pH C. Change in pH alters ionization states of contact amino acids in the active site D. All enzymes have optimum pHarrow_forwardChoose the statement that best explains WHY the aldol condensation is considered base-catalyzed. a The base lowers the activation energy of the intermediates. b The base is used throughout the reaction but not consumed in the reaction, since it is regenerated in the final steps. C The base deprotonates the ketone, generating the nucleophile. d The base reacts with any acid that is present in the solution, accelerating the rate.arrow_forward
- The following are the negative regulators of phosphofructokinase except Select one: a. AMP +b. H c. Citrate d. ATParrow_forwardFoF1 ATPase is the enzyme that catalyzes ATP synthesis. The enzyme itself is deactivated by ATP. What mode of enzyme regulation is being exemplified? Select the correct response: Trasncriptional control Covalent modification Proteolytic modification Allosteric regulation Compartmentationarrow_forwardWhat is the activated reactant in the biosynthesis of each of the following compounds? a. Phosphoribosylamine c. Orotidylate (from orotate) b. Carbamoylaspartate d. Phosphoribosylanthranilatearrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





