
Life: The Science of Biology
11th Edition
ISBN: 9781319010164
Author: David E. Sadava, David M. Hillis, H. Craig Heller, Sally D. Hacker
Publisher: W. H. Freeman
expand_more
expand_more
format_list_bulleted
Question
Chapter 24.1, Problem 2R
Summary Introduction
To explain:
Mark boundaries between the sites given in Figure, which were formed, during the same geological time period.
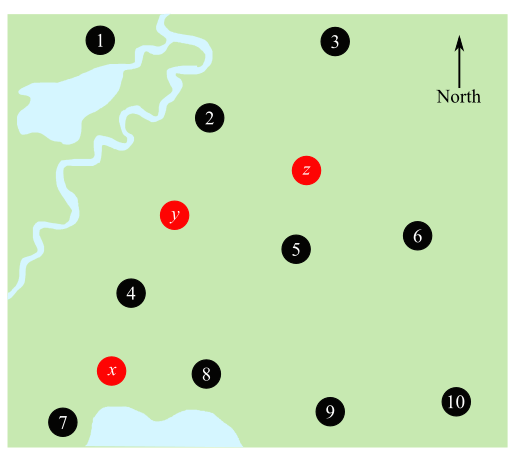
Given:
Table 1 given below shows the ratio between the 206Pb and 238U and the estimated age of the sites given.
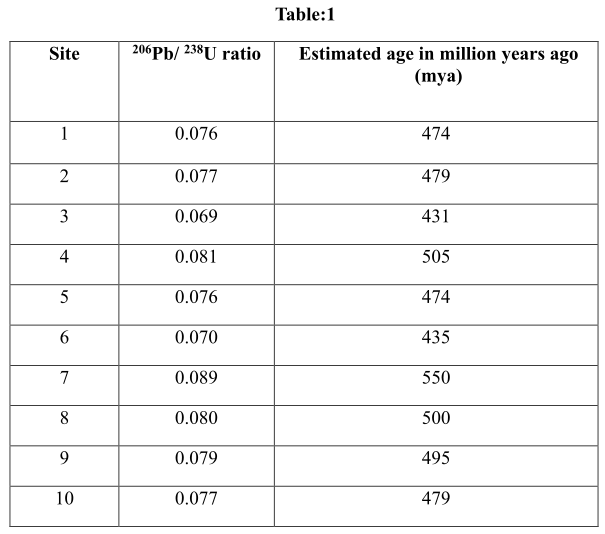
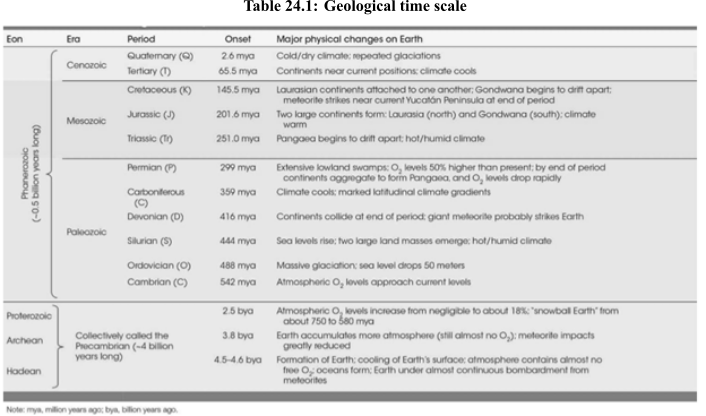
Introduction:
The ratio of 206Pb to 238U is known as uranium and lead dating. It is a radioactive dating technique used in earlier times to date the rocks that are formed in between 1 million years to 4.5 billion years ago. The number of radioactive isotopes of uranium and amount of other materials into which the radioactive isotopes would be decompose.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
The most useful method for dating ancient calcium rich cave deposits would be
Group of answer choices
A: Argon-Argon Dating
B: Paleomagnetic Reversals
C: Electron Spin Resonance Dating
D: Uranium-Thorium Dating
E: Potassium-Argon Dating
Radiometric dating does not measure the age of an individual atom. It is a measure of the age of a quantity of atoms—a statistic. As with any statistical measure, its values may deviate around an average (sampling error, Section 1.8). Imagine that one sample of rock is dated ten different ways. Nine of the tests yield an age close to 225,000 years. One test yields an age of 3.2 million years. Do the nine consistent results imply that the one that deviates is incorrect, or does the one odd result invalidate the nine that are consistent?
Which item is best dated using the potassium-argon (K-Ar) method?
Group of answer choices
A: Cave layers in South Africa
B: stone tools
C: Ancient dinosaur bones
D: organic materials
E: Lava flows found in the Olduvai Gorge
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Use the following figure to answer questions 2 and 3. mya - million years ago 1,00 mya = 1 bilhan yeas ago |4,600 mya: Formation of Earth 4000 mya |3,500 mya: Oldest known fosll Present day |2,000mya:Rrstmulticelled organisms 1,000 myu 3,000 myn 2000 mya 2. How old is the oldest known fossil? 3. How much time passed between the Earth's formation and the appearance of the first multicellular organ- ism? 4. What key evolutionary event occurred 370 million years ago? Would this have been considered macro- evolution or microevolution? Briefly explain your answer.arrow_forwardA fossil is found to contain the radioactive parent and daughter isotopes uranium 235U, and lead 207Pb, in the ratio 12.5% to 87.5%. Assuming that no 207Pb was present when the fossil first formed, estimate the age of this sample.Please show some thought process and a therefore statement. Thank you very much, have a great day!arrow_forwardSuppose a scientist finds a rock with an interesting fossil. Curious to know the age of the fossil, she takes the rock to a lab. The lab determines the amount of rubidium (Rb) and strontium (Sr) in the rock. The scientist is familiar with radiometric dating and knows that she can now calculate the age of the rock, 1, using an isochron. The Rb-Sr isochron equation is 87. Rb. [edr – 1] 86 °Sr. 87 87 Sro 86 Sro 86 Sr. 87. Rb 3.807 and 89.60. In a published table, she finds that Using the lab's values, she calculates the isotope ratios as 86 Sr. 87 = 0.7129. She also finds that the decay constant, 86 the ratio of strontium-87 to strontium-86 at the formation of the rock was Sro 2, for rubidium-87 is 1.42 x 10-11. Use the equation and provided data to calculate t. Put your answer in scientific notation and round to the nearest tenth. years x10 What is the approximate age of the rock in billions of years? Round to the nearest tenth. billions of years t =arrow_forward
- Radiometric dating uses the decay of to to determine how old fossils of biological material are using the rate of decay. Group of answer choices 14C, 12C 14N, 12C 12C, 14N 14C, 14Narrow_forwardA paleontologist estimates that when a particular rock formed, it contained 12 mg of the radioactive isotope potassium-40. The rock now contains 3 mg of potassium-40. The half-life of potassium-40 is 1.3 billion years. From this information, you can conclude that the rock is approximately ______________ billion years old.arrow_forwardGive two lines of evidence supporting the asteroid impact hypothesis for the Cretaceous extinctions.arrow_forward
- Using the principles of relative dating, put the rock layers, faults, and igneous intrusions in a sequence from youngest to oldestarrow_forwardGive 1 reason from the reading that shows that there are no life forms on Venus.arrow_forwardWhich parent isotope is most useful for dating samples that are greater than 100 million years old? a potassium-40 b lead-206 c argon-40 d uranium-238arrow_forward
- List several significant events in the history of life on earth that are associated with the K-T Boundary.arrow_forwardMatch the reliable age range of the right with the appropriate technique on the left. Thermoluminescence [ Choose] Choose] Ages greater than 1,000 years ago. Ages between 1,000 years ago and 600 kya. Ages greater than 100 kya Ages between 300 BP and 300 kya. Ages between 0- 50 kya. [Choose ] Uranium-Thorium K-Ar (Potassium-Argon) Fission Track [ Choose ] Radiocarbon (14C) [Choose ]arrow_forwardIn radiometric dating, scientists use the half-life of a radioactive isotope, to determine the approximate age of rocks, and the fossils that are embedded in them. Half-life is the time it takes for half of a radioactive element to become stable.Uranium 238 (238U) is a radioactive element that decays into Lead (206Pb) after 4.5 billion years. 6) Scientists found a fossil in a layer of rock that tested as 1/8th U and 7/8th Pb. What is the approximate age of the fossil? A) 562 million years old B) 4.5 billion years old C) 9 billion years old D) 13.5 billion years old Not Gradedarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning

Biology Today and Tomorrow without Physiology (Mi...
Biology
ISBN:9781305117396
Author:Cecie Starr, Christine Evers, Lisa Starr
Publisher:Cengage Learning

Necrosis vs. Apoptosis: Cell Death; Author: AMBOSS: Medical Knowledge Distilled;https://www.youtube.com/watch?v=zFrBwGfOQs0;License: Standard Youtube License