
Concept explainers
(a)
Interpretation: To indicate whether
Concept introduction: Electron transport chain is a sequence of biochemical reactions in which electrons and hydrogen atoms from the citric acid cycle are transferred to various intermediate carriers and finally reacts with molecular oxygen to form a water molecule.
There are four complexes associated with the electron transport chain that is present in the inner mitochondrial membrane. The four complexes that help in the electron transfer in the electron transport chain are:
Complex I:
Complex II:
Complex III:
Complex IV:
An overview of the electron transport chain is as follows:
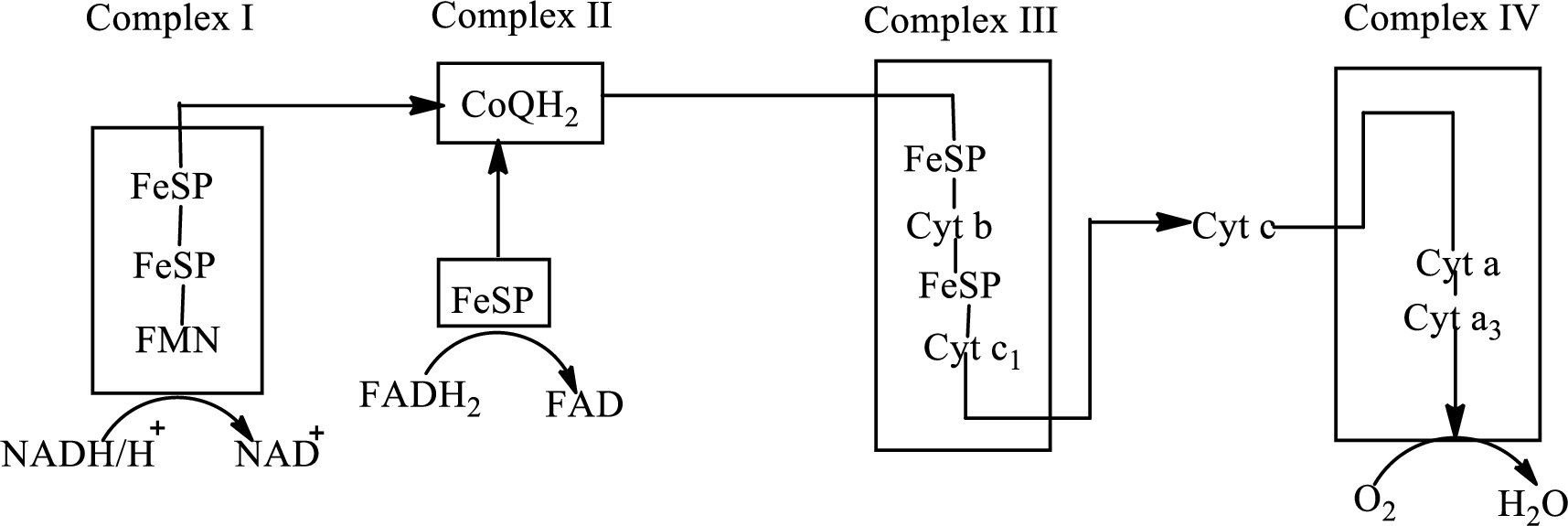
Redox
Here A is oxidized form and AH is reduced form.
(a)
Answer to Problem 23.87EP
Explanation of Solution
(b)
Interpretation: To indicate whether
Concept introduction: Electron transport chain is a sequence of biochemical reactions in which electrons and hydrogen atoms from the citric acid cycle are transferred to various intermediate carriers and finally reacts with molecular oxygen to form a water molecule.
There are four complexes associated with the electron transport chain that is present in the inner mitochondrial membrane. The four complexes that help in the electron transfer in the electron transport chain are:
Complex I:
Complex II:
Complex III:
Complex IV:
An overview of the electron transport chain is as follows:
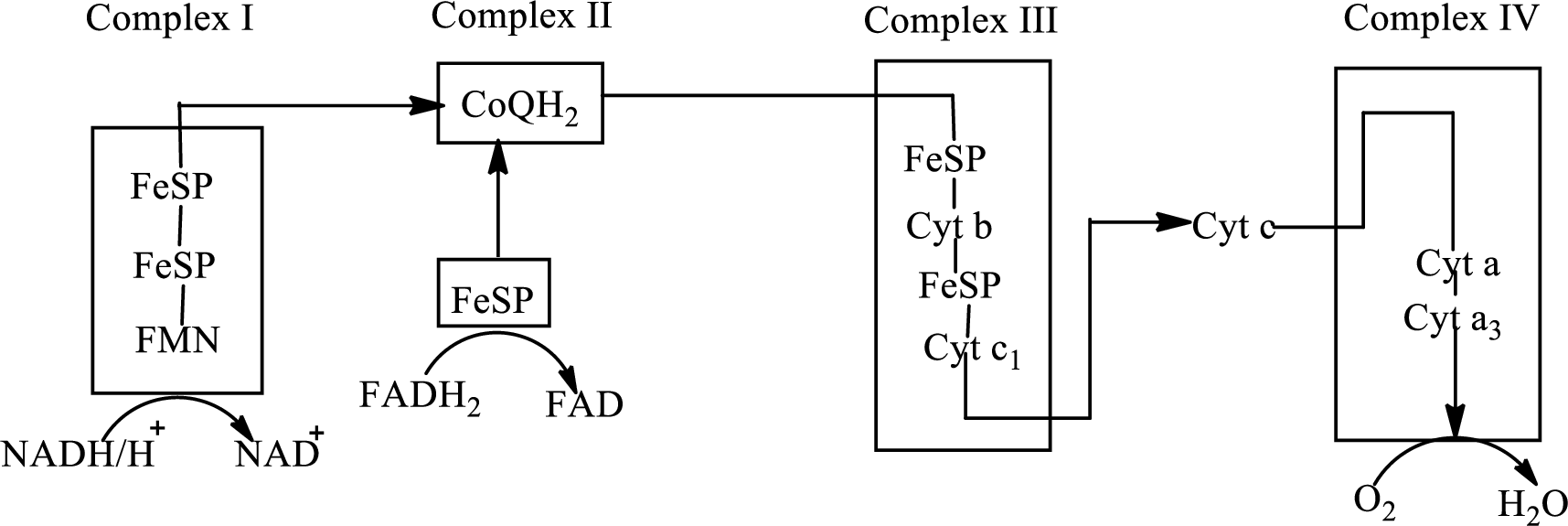
Redox reactions involve oxidation and reduction reaction occurring simultaneously so that one species is oxidized and the other one is reduced. The species that gain hydrogen or electron is known as reduced form and the species that loss hydrogen or electron is known as oxidized form. The general representation of redox reaction is
Here A is oxidized form and AH is reduced form.
(b)
Answer to Problem 23.87EP
Explanation of Solution
(c)
Interpretation: To indicate whether
Concept introduction: Electron transport chain is a sequence of biochemical reactions in which electrons and hydrogen atoms from the citric acid cycle are transferred to various intermediate carriers and finally reacts with molecular oxygen to form a water molecule.
There are four complexes associated with the electron transport chain that is present in the inner mitochondrial membrane. The four complexes that help in the electron transfer in the electron transport chain are:
Complex I:
Complex II:
Complex III:
Complex IV:
An overview of the electron transport chain is as follows:
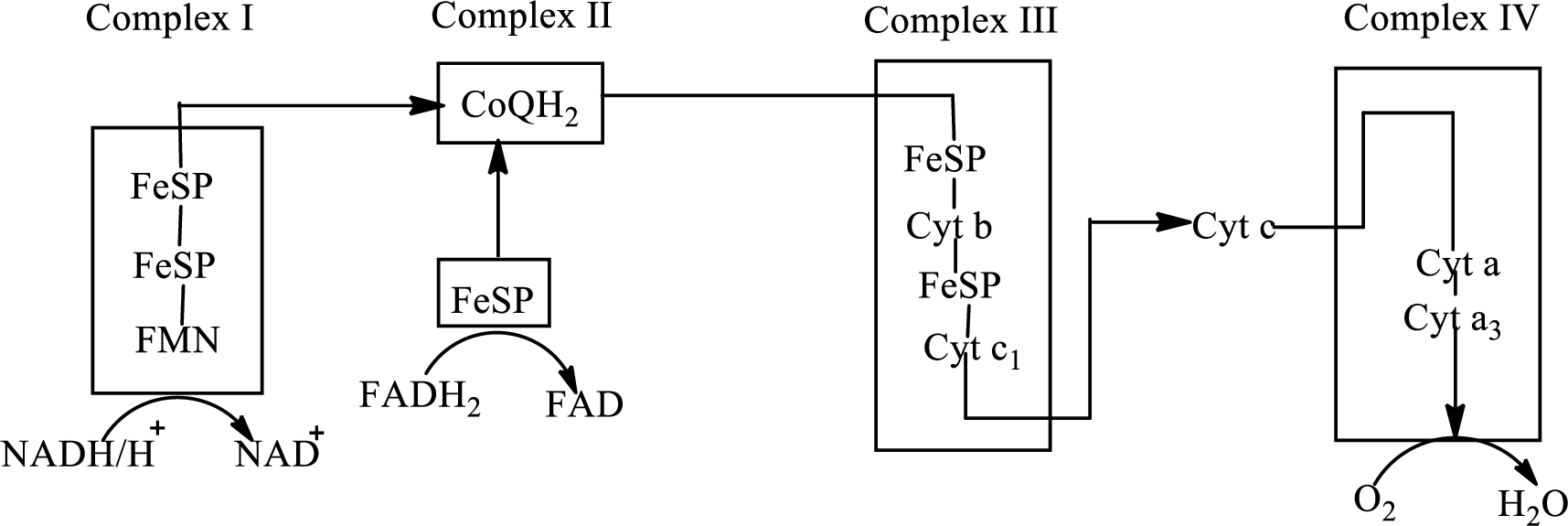
Redox reactions involve oxidation and reduction reaction occurring simultaneously so that one species is oxidized and the other one is reduced. The species that gain hydrogen or electron is known as reduced form and the species that loss hydrogen or electron is known as oxidized form. The general representation of redox reaction is
Here A is oxidized form and AH is reduced form.
(c)
Answer to Problem 23.87EP
NADH is the reduced form of the nicotinamide adenine dinucleotide.
Explanation of Solution
In complex I, electrons are transferred from the
The reaction of the oxidation of
Here,
(d)
Interpretation: To indicate whether is in its oxidized form or its reduced form.
Concept introduction: Electron transport chain is a sequence of biochemical reactions in which electrons and hydrogen atoms from the citric acid cycle are transferred to various intermediate carriers and finally reacts with molecular oxygen to form a water molecule.
There are four complexes associated with the electron transport chain that is present in the inner mitochondrial membrane. The four complexes that help in the electron transfer in the electron transport chain are:
Complex I:
Complex II:
Complex III:
Complex IV:
An overview of the electron transport chain is as follows:
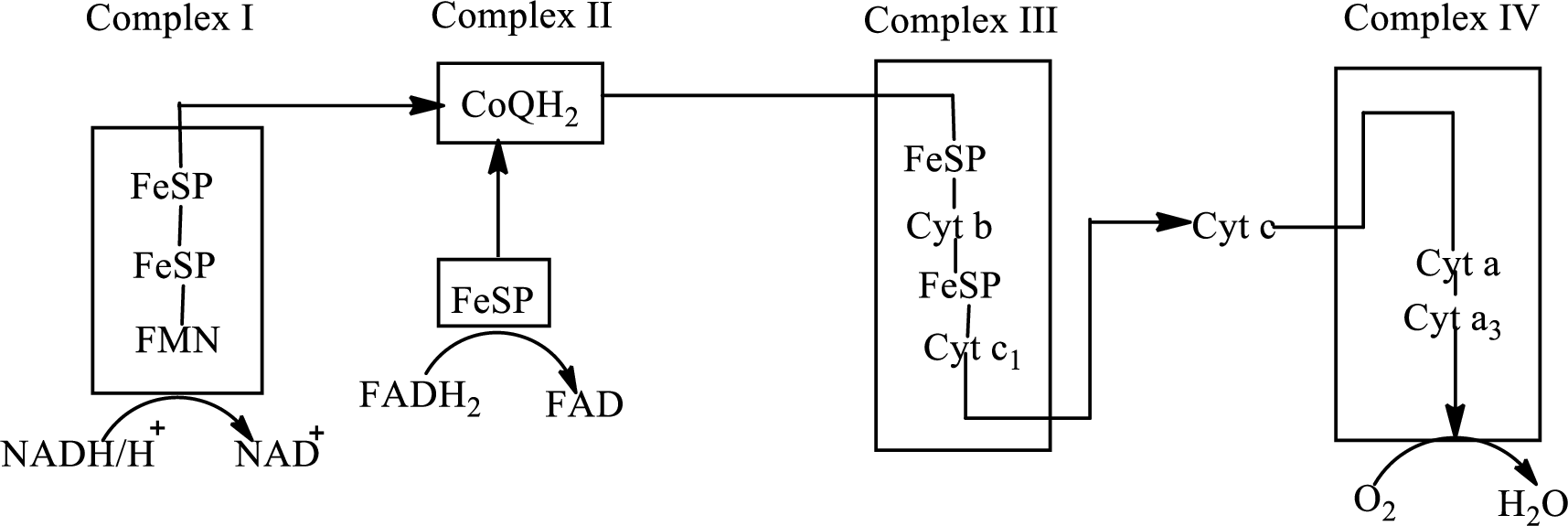
Redox reactions involve oxidation and reduction reaction occurring simultaneously so that one species is oxidized and the other one is reduced. The species that gain hydrogen or electron is known as reduced form and the species that loss hydrogen or electron is known as oxidized form. The general representation of redox reaction is
Here A is oxidized form and AH is reduced form.
(d)
Answer to Problem 23.87EP
FAD is the oxidized form of the flavin adenine dinucleotide.
Explanation of Solution
In the complex II, electrons are transferred from the
Here,
Want to see more full solutions like this?
Chapter 23 Solutions
General, Organic, and Biological Chemistry Seventh Edition
- Indicate whether each of the following electron carriers is in its oxidized form or its reduced form. Fe(III) SP c) NADH Cyt b (Fe+3) d) FADarrow_forwardMark the following statements as true or false. If a statement is false, correct it to make a true statement. a. During an oxidation-reduction reaction, the compound with the greatest electron affinity accepts electrons and is oxidized. b. The electromotive force is generated by the flow of electrons from one substance that is oxidized to another substance that is reduced. c. NAD+ and FAD are electron carriers in the cell. d. ATP is the “money” that the cell uses to “pay for” the cell’s exergonic reaction.arrow_forwardWhich of the following statements concerning ATP is true? a. The free energy value for the hydrolysis of ATP is nearly the same for ADP. b. The free energy value for the hydrolysis of ATP is greater than that for ADP. c. ATP hydrolysis is more likely at pH 5 than at pH 7. d. One mole of glycerate-1,3-bisphosphate can phosphorylate one mole of AMP to yield ATP.arrow_forward
- During cell respiration, what happens to the strength of the bonds between atoms in glucose vs those in the products water and carbon dioxide? Choose all answers that are correct. a. the carbon-hydrogen bonds in glucose are stronger than the carbon-oxygen bonds in carbon dioxide b. an oxidizing agent c.the carbon-hydrogen bonds in glucose are weaker than the carbon-oxygen bonds in carbon dioxide d.the carbon-hydrogen bonds in glucose are weaker than the oxygen-hydrogen bonds in water e.the carbon-hydrogen bonds in glucose are stronger than the oxygen-hydrogen bonds in waterarrow_forwardIn the reaction catalyzed by malate dehydrogenase, which of the following molecules is classified as the reduced donor? a. Malate b. Oxaloacetate c. Fumerate d. FAD e. FADH2 f. NAD+ g. NADHarrow_forwardWhich of the following final electron acceptors is used during anaerobic respiration? a. lactic acid b. pyruvate c. glucose d. nitrate e. waterarrow_forward
- Explain why the hydrolysis of the following compounds releases energy. A. phosphoenol pyruvate B. 1,3-diphosphoglycerate C. succinyl CoAarrow_forwardDoes the following inhibitors decrease ATP production or Blocks ATP production? a. DCCD b. FCCP c. Amytal d. Carboxin e. Cyanidearrow_forwardThe aspartate–malate shuttle transfers electrons from cytoplasmic NADH to the electron transport system. Which of the following molecules is not directly involved in this pathway? a. malate b. oxaloacetate c. citrate d. α-ketoglutaratearrow_forward
- Consider the function of the cofactor FAD. Which of the following makes it unique (different) from NAD+? Select all that apply. a. Can facilitate single electron transfers b. Is associated with an enzyme and not a mobile electron carrier c. Serves to facilitate redox reactions d. s an electron carrier in the TCA cyclearrow_forwardduring oxidative phosphorylation, one FADH2 produced by the krebs cycle will produce a. 1 ATP b. 2 ATP c. 3 ATP d. 4 ATP e. 5 ATParrow_forwardWhich one of the following statements about coenzyme Q is not true? a. coenzyme Q is lipid-soluble b. coenzyme Q can accept one or two electrons c. conenzyme Q often diffuses in the membrane from one protein complex to another in its semiquinone form d. Coenzyme Q is one of the five electron carriers used in the electron transport chainarrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





