
Interpretation: Each amide group present in oxytocin is to be classified as
Concept introduction: The amide is identified as primary
Answer to Problem 22.1P
The classification of each amide group present in oxytocin as
Explanation of Solution
The classification of each amide group present in oxytocin as
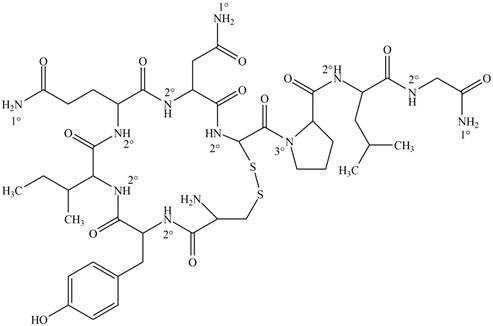
Figure 1
Amides are the organic compounds which contain
The classification of each amide group present in oxytocin as
Want to see more full solutions like this?
Chapter 22 Solutions
PKG ORGANIC CHEMISTRY
- Label each nitrogen-containing functional group in lidocaine, a local anesthetic, as an amine or amide, and classify it as 1 °, 2 °, or 3 °.arrow_forwardWhat carboxylic acid and amine are needed to synthesize the pain reliever phenacetin? Phenacetin was once a component of the over-the-counter pain reliever APC (aspirin, phenacetin, caffeine), but it is no longer used because of its kidney toxicity.arrow_forwardWhat ammonium salt is formed when each amine is treated with HCl? Draw the structure of the resulting salt.arrow_forward
- Acetaminophen is an analgesic marketed under the brand name Tylenol, among others. Draw the amine that results from the base hydrolysis of acetaminophen.arrow_forwardGive a systematic name for each amide ?arrow_forwardExplain why many amines with useful medicinal properties are sold as their ammonium saltsarrow_forward
- Indicate whether the following statement is true or false. Aliphatic amines are more basic than ammonia, whereas aromatic amines are less basic than ammonia.RightFalsearrow_forwardWhat products are formed when each amide is hydrolyzed with water and HCl?arrow_forwardWhat amine is formed by reduction of each amide?arrow_forward
- Give an acceptable name for each amine.arrow_forwardIn the 1880's, Acetanilide, sold under the name Antifebrin, was widely used as a pain reliever and fever reducer. However, it had many adverse side effects, including cyanosis as a result of methemoglobinemia. The toxic side effects were the result of a small portion of acetanilide being hydrolyzed to aniline. Acetanilide was discontinued and replaced with phenacetin. Later studies show that both acetanilide and phenacetin are metabolized to acetaminophen. This metabolite, which we know as Tylenol, is responsible for the analgesic and antipyretic properties. Part 1: Show a detailed arrow pushing mechanism of the acid catalyzed hydrolysis of acetanilide to aniline Part 2: Propose a synthesis of Acetaminophen from phenol NH NH NH Phenacetin inophen Acetanilide Attach File Browse Local Files Browse Content Collectionarrow_forwardWhat carbonyl compound and amine are formed by the hydrolysis of each compound?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning



