
(a)
Interpretation:
The number of
Concept Introduction:
There are three types of
(a)
Explanation of Solution
The given compound is shown here:
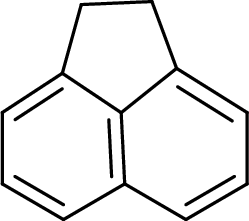
No. of double bonds:
So, the total number of
(b)
Interpretation:
The number of
Concept Introduction:
There are three types of
(b)
Explanation of Solution
The given compound is shown here:
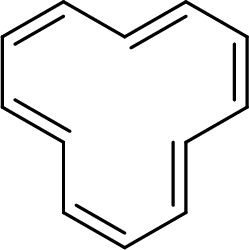
No. of double bonds:
So, the total number of
(c)
Interpretation:
The number of
Concept Introduction:
There are three types of
(c)
Explanation of Solution
The given compound is shown here:
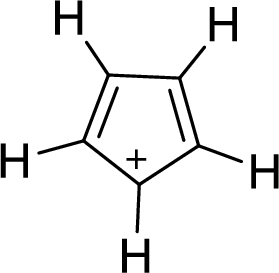
No. of double bonds:
So, the total number of
(d)
Interpretation:
The number of
Concept Introduction:
There are three types of
(d)
Explanation of Solution
The given compound is shown here:
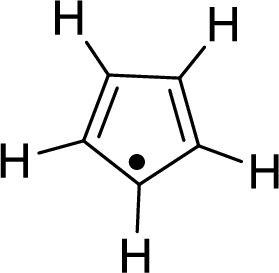
No. of double bonds:
So, the number of
There is an unpaired free-radical electron which is represented by a single dot. This unpaired free-radical electron will be in resonance with the
Therefore, the total number of
(e)
Interpretation:
The number of
Concept Introduction:
There are three types of
(e)
Explanation of Solution
The given compound is shown here:
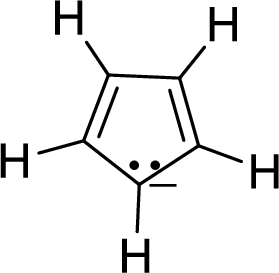
No. of double bonds:
So, the number of
There is a pair of electrons which is represented by two dots with a negative sign. This pair of electrons will be in resonance with the
Therefore, the total number of
(f)
Interpretation:
The number of
Concept Introduction:
There are three types of
(f)
Explanation of Solution
The given compound is shown here:
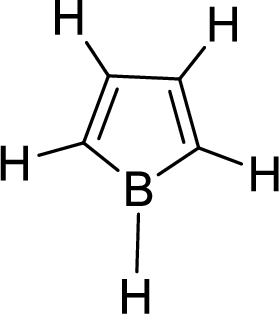
No. of double bonds:
So, the total number of
(g)
Interpretation:
The number of
Concept Introduction:
There are three types of
(g)
Explanation of Solution
The given compound is shown here:
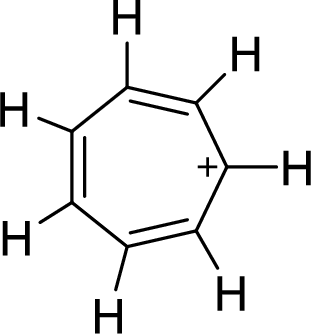
No. of double bonds:
So, the total number of
(h)
Interpretation:
The number of
Concept Introduction:
There are three types of
(h)
Explanation of Solution
The given compound is shown here:
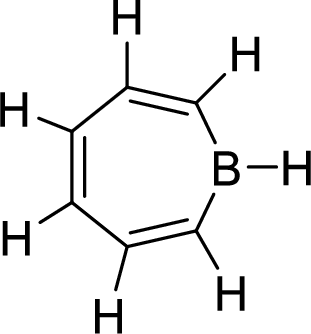
No. of double bonds:
So, the total number of
(i)
Interpretation:
The number of
Concept Introduction:
There are three types of
(i)
Explanation of Solution
The given compound is shown here:
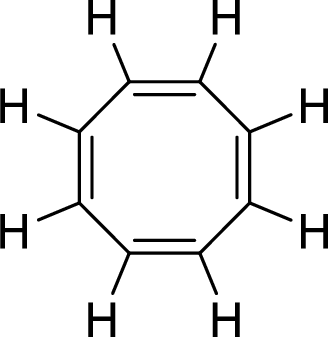
No. of double bonds:
So, the total number of
(j)
Interpretation:
The number of
Concept Introduction:
There are three types of
(j)
Explanation of Solution
The given compound is shown here:
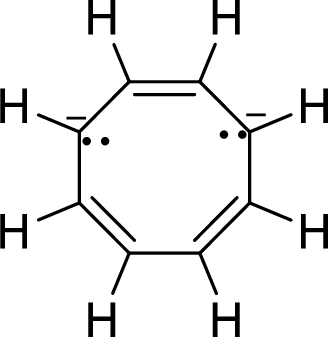
No. of double bonds:
So, the number of
There are two pairs of electrons and each pair is represented by two dots with a negative sign. These two pair of electrons will be in resonance with the
Therefore, the total number of
Want to see more full solutions like this?
Chapter 21 Solutions
Student Study Guide and Solutions Manual for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- • predict the geometry of a molecule from its, Lewis structure.arrow_forwardWhen one electron is added to an oxygen molecule, a superoxide ion (O2) is formed. The addition of two electrons gives a peroxide ion (O22) . Removal of an electron from O2 leads to O2+ . (a) Construct the correlation diagram for O2 . (b) Give the valence electron configuration for each of the following species: O2+,O2,O2,O22 . (c) Give the bond order of each species. (d) Predict which species are paramagnetic. (e) Predict the order of increasing bond dissociation energy among the species.arrow_forwardState the net number of bonding electrons between xenon and the four fluorines in [XeF4] that is indicated by the mo diagram containing the proper total number of electrons.?arrow_forward
- (a) Determine the molecular geometry of SeF4using the VSEPR model. The molecular geometry of SeF4 is (___________________) linear or trigonal planar or bent tetrahedral or trigonal pyramidal or trigonal bipyramidal or seesaw or T-shaped or octahedral or square pyramidal or square planar . (b) Explain why the molecular geometry of SeF4 is different from that of IBr3, although they have similar electron-domain geometries. Although both SeF4 and IBr3 have the electron-domain geometry of (_________________) linear or trigonal planar or bent tetrahedral or trigonal pyramidal or trigonal bipyramidal or seesaw or T-shaped or octahedral or square pyramidal or square planar, IBr3 has _____ lone pair(s) of electrons whereas SeF4 has only ________ lone electron pair(s) in the (______________) axial or equatorial positions. Accordingly, the molecular geometry of SeF4 is (________________) linear or trigonal planar or bent tetrahedral or trigonal pyramidal or trigonal bipyramidal or seesaw or…arrow_forwardAnswer the questions in the table below about the shape of the phosphorus pentabromide (PBr,) molecule. How many electron groups are around the central phosphorus atom? Note: one "electron group" means one lone pair, one single bond, one double bond, or one triple bond. What phrase best describes the arrangement of these electron groups around the central phosphorus atom? (You may need to use the scrollbar to see all the choices.) (choose one) Ar Explanation Check © 2021 McGraw Hill LLC. AlI Rights Reserved. Terms of Use Privacy Center | Accessibilityarrow_forwardExplain what is wrong with each molecular geometry and provide the correct molecular geometry, given the numbers of lone pairs and bonding groups on the central atom. Match the words in the left column to the appropriate blanks in the sentences on the right. a bent a trigonal bipyramidal an octahedral a trigonal planar a square planar a trigonal pyramidal a tetrahedral a seesaw a linear In structure (a), four pairs of electrons give cause lone pair-bonded pair repulsions and would have Reset electron geometry. The lone pair would molecular geometry. Help In structure (b), five pairs of electrons give electron geometry. The lone pair occupies an equatorial position to minimize lone pair-bonded pair repulsions, and the molecule would have molecular geometry. In structure (c), six pairs of electrons give electron geometry. The two lone pairs would occupy opposite positions to minimize lone pair-lone pair repulsions, and the molecule would have molecular geometry.arrow_forward
- Which of the following elements are capable of expanding their octet? Select all that apply. Br Se Farrow_forwardWrite the best Lewis dot structure for POCl₃, be sure to give the electronic geometry, molecular geometry, hybridization of the central element, polarity, and bond angle around the central element.arrow_forwardDescribe the bonding in the OF2 bent molecule. Predict its energy level diagram and electron configuration.arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning





