
Organic Chemistry
7th Edition
ISBN: 9780321803221
Author: Paula Y. Bruice
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 20.6, Problem 11P
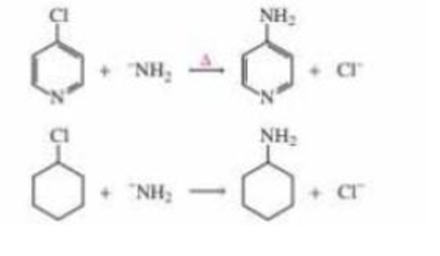
How to the mechanisms of the following reactions differ?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What is the mechanism of the following reactions?
How do the mechanisms of the following reactions differ?
What is the synthesis and mechanism for the following?
Chapter 20 Solutions
Organic Chemistry
Ch. 20.1 - Name the following:Ch. 20.2 - Prob. 2PCh. 20.2 - Prob. 3PCh. 20.3 - Draw the product of each of the following...Ch. 20.5 - Prob. 6PCh. 20.5 - When pyrrole is added to a dilute solution of...Ch. 20.5 - Explain why cyclopentadiene (pKa = 15) is more...Ch. 20.6 - Prob. 10PCh. 20.6 - How to the mechanisms of the following reactions...Ch. 20.6 - Prob. 12P
Ch. 20.6 - Rank the following compounds from easiest to...Ch. 20.7 - Prob. 14PCh. 20.7 - Prob. 15PCh. 20.7 - Prob. 16PCh. 20.7 - Prob. 17PCh. 20.7 - Prob. 18PCh. 20.7 - Prob. 19PCh. 20.7 - Prob. 20PCh. 20 - Name the following:Ch. 20 - Prob. 22PCh. 20 - Prob. 23PCh. 20 - Which of the following compounds is easier to...Ch. 20 - Rank the following compounds from most reactive to...Ch. 20 - One of the following compounds undergoes...Ch. 20 - Benzene undergoes electrophilic aromatic...Ch. 20 - The dipole moments of furan and tetrahydrofuran...Ch. 20 - Prob. 29PCh. 20 - Prob. 30PCh. 20 - The chemical shifts of the C-2 hydrogen in the...Ch. 20 - Explain why protonating aniline has a dramatic...Ch. 20 - Prob. 33PCh. 20 - Propose a mechanism for the following reaction:Ch. 20 - Prob. 35PCh. 20 - Propose a mechanism for the following reactions:Ch. 20 - Prob. 37PCh. 20 - a. Draw resonance contributors to show why...Ch. 20 - Prob. 39PCh. 20 - Pyrrole reacts with excess...Ch. 20 - Prob. 41PCh. 20 - Prob. 42PCh. 20 - Organic chemists work with tetraphenylporphyrins...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which of the following reactions would result in the correct product?arrow_forwardIdentify the mechanisms by which each reactions above proceeds from the choices listed.arrow_forwardProvide the major product of each of the following reactions: What do all these reactions have in common? How do all reactions differ?arrow_forward
- Draw the mechanism, including stereochemistry, of the following reaction, predicting the products formed. What is the relationship of the products that are formed?arrow_forwardDescribe the mechanism of the following reaction. 1) H2N NH2 (excess) SH SH `CO2H AcO 2) КОН 3) H +arrow_forwardPredict the product and draw the mechanisms of the following reactions. Write down the rate, molecularity, and order of reaction. (hint: Slow step is the rate - determining step)arrow_forward
- 2-chloro-1,1,3-trimethylcyclohexane can undergo either substitution or elimination or BOTH reactions. Determine ALL possible type of reaction occur (E1, E2, SN1 or SN2), propose a detailed mechanism (or mechanisms) for reaction and identity the MAJOR compounds when; the reagent used is butanol he reagent used is butoxide the reagent used is (CH),0arrow_forwardCan you explain the mechanism for this question HO OH H2SO4 cat. H₂Oarrow_forwardGive the reactants and the mechanism to synthesize the following:arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
SAR of Anticancer(Antineoplastic) Drug/ Alkylating agents/ Nitrogen Mustard; Author: Pharmacy Lectures;https://www.youtube.com/watch?v=zrzyK3LhUXs;License: Standard YouTube License, CC-BY