
Organic Chemistry
7th Edition
ISBN: 9780321803221
Author: Paula Y. Bruice
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 20, Problem 24P
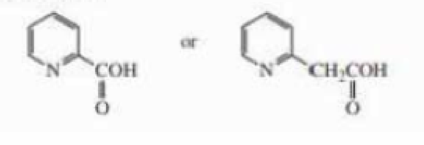
Which of the following compounds is easier to decarboxylate
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Which of the following molecules will undergo decarboxylation with mild heating?
Arrange the following compounds in order of decreasing reactivity towards hydrolysis.
H2N.
H3CO
NAI
IYK
КАН
BCS
Which of the following will readily undergo decarboxylation upon heating?
Chapter 20 Solutions
Organic Chemistry
Ch. 20.1 - Name the following:Ch. 20.2 - Prob. 2PCh. 20.2 - Prob. 3PCh. 20.3 - Draw the product of each of the following...Ch. 20.5 - Prob. 6PCh. 20.5 - When pyrrole is added to a dilute solution of...Ch. 20.5 - Explain why cyclopentadiene (pKa = 15) is more...Ch. 20.6 - Prob. 10PCh. 20.6 - How to the mechanisms of the following reactions...Ch. 20.6 - Prob. 12P
Ch. 20.6 - Rank the following compounds from easiest to...Ch. 20.7 - Prob. 14PCh. 20.7 - Prob. 15PCh. 20.7 - Prob. 16PCh. 20.7 - Prob. 17PCh. 20.7 - Prob. 18PCh. 20.7 - Prob. 19PCh. 20.7 - Prob. 20PCh. 20 - Name the following:Ch. 20 - Prob. 22PCh. 20 - Prob. 23PCh. 20 - Which of the following compounds is easier to...Ch. 20 - Rank the following compounds from most reactive to...Ch. 20 - One of the following compounds undergoes...Ch. 20 - Benzene undergoes electrophilic aromatic...Ch. 20 - The dipole moments of furan and tetrahydrofuran...Ch. 20 - Prob. 29PCh. 20 - Prob. 30PCh. 20 - The chemical shifts of the C-2 hydrogen in the...Ch. 20 - Explain why protonating aniline has a dramatic...Ch. 20 - Prob. 33PCh. 20 - Propose a mechanism for the following reaction:Ch. 20 - Prob. 35PCh. 20 - Propose a mechanism for the following reactions:Ch. 20 - Prob. 37PCh. 20 - a. Draw resonance contributors to show why...Ch. 20 - Prob. 39PCh. 20 - Pyrrole reacts with excess...Ch. 20 - Prob. 41PCh. 20 - Prob. 42PCh. 20 - Organic chemists work with tetraphenylporphyrins...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which of the following compounds can serve as Michael acceptors?arrow_forwardWhich of the following choices is a product of reacting a ketone with HCN?arrow_forwardThe major product that will form during the nitration of benzoic acid is? OA H₂N OR O-N ос OD. NO₂ COOH COOH COOR NH₂ COOH OZN H-N COOH COOHarrow_forward
- Prepare the following substances from different aldehydes or ketonesarrow_forwardThioester hydrolysis yielding more energy than O-ester hydrolysis is ultimately due to Bond between S and carbonyl carbon is of lower energy Thioesters are more resonance stabilized Products of O-ester hydrolysis have lower degree of solvation than its reactants in water Sulphur being a larger atom than oxygenarrow_forwardWhich of the attached compounds can serve as Michael acceptors?arrow_forward
- Explain why pentane-2,4-dione forms two alkylation products (A and B) when the number ofequivalents of base is increased from one to two.arrow_forwardComplete the following proposed acid–base reactions, and predict whether the reactants or products are favored.arrow_forwardWhich of the following compounds CANNOT serve as Michael acceptor? МеO Aarrow_forward
- Show how acid derivatives hydrolyze to carboxylic acids under either acidic or basicconditions. Explain why some acid derivatives (amides, for example) require muchstronger conditions for hydrolysis than other derivatives.arrow_forwardWhether you choose to perform an acid-catalyzed or base-catalyzed hydrolysis, the end product should be the same. Predict the product of the following reaction conditions.arrow_forwardThe following molecule undergoes an intramolecular reaction in the presence of pyrrolidinium acetate, the protonated form of pyrrolidine. Draw the product of this reaction, assuming that a dehydration reaction takes place.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Characteristic Reactions of Benzene and Phenols; Author: Linda Hanson;https://www.youtube.com/watch?v=tjEqEjDd87E;License: Standard YouTube License, CC-BY
An Overview of Aldehydes and Ketones: Crash Course Organic Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=-fBPX-4kFlw;License: Standard Youtube License