
Concept explainers
What reagent is needed to convert
a.
b.
(a)
Interpretation:
The reagent needed to convert
Concept introduction:
Acid chlorides, which contains good leaving group
Answer to Problem 20.44P
The reagent needed to convert
Explanation of Solution
The given compound is aldehyde. Acid chlorides on reaction with mild reducing agent like lithium tri-tert-butoxyaluminium hydride
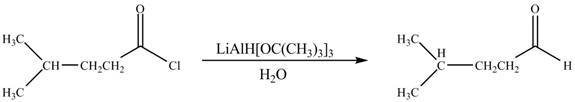
Figure 1
Therefore,
The reagent needed to convert
(b)
Interpretation:
The reagent needed to convert
Concept introduction:
Acid chlorides, which contains good leaving group
Answer to Problem 20.44P
The reagent needed to convert
Explanation of Solution
The structure of given ketone compound shows that one ethene molecule is attached to carbonyl carbon. Acid chlorides on reaction with one equivalent of organocuprate reagent convert into ketone. The organocuprate reagent removes the good leaving
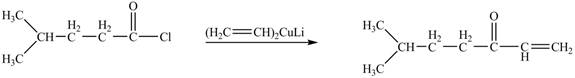
Figure 2
Therefore,
The reagent needed to convert
(c)
Interpretation:
The reagent needed to convert
Concept introduction:
Acid chlorides, which contains good leaving group
Answer to Problem 20.44P
The reagent needed to convert
Explanation of Solution
The structure of given ketone compound shows that the hydroxyl group is attached to tertiary carbon atom. Acid chlorides on reaction with two equivalents of Grignard reagent convert into tertiary alcohol. The first equivalent of Grignard reagent remove good leaving
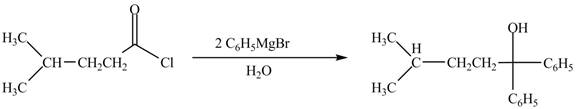
Figure 3
Therefore,
The reagent needed to convert
(d)
Interpretation:
The reagent needed to convert
Concept introduction:
Acid chlorides, which contains good leaving group
Answer to Problem 20.44P
The reagent needed to convert
Explanation of Solution
Acid chlorides on reaction with strong reducing agent like lithium aluminium hydride
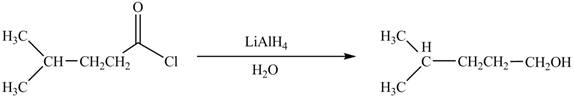
Figure 4
Therefore,
The reagent needed to convert
Want to see more full solutions like this?
Chapter 20 Solutions
Organic Chemistry-Package(Custom)
- Draw the products formed when CH3CH2C ≡ C−Na+reacts with each compound. a. CH3CH2CH2Brb. (CH3)2CHCH2CH2Clc. (CH3CH2)3CCld. BrCH2CH2CH2CH2OHe. ethylene oxide followed by H2Of. propene oxide followed by H2Oarrow_forwardGive the IUPAC name for each compound. CH3 CH2CH3 Br a. PHCH(CH3)2 b. С. d.arrow_forwardIndicate the product obtained by reacting A and B. A || H3C-S-CH₂ Na* || CH 3 B CH2. C. CH3 H3C CH3arrow_forward
- Draw the products formed when CH3CH2C=C Na+ reacts with each compound. a. CH3CH2CH2Br b.(CH3)2CHCH2CH2Cl c.(CH3CH2)3CCl d.BrCH2CH2CH2CH2OH e. ethylene oxide followed by H2O f.propene oxide followed by H2Oarrow_forwardHCI CH3OH CH₁₂arrow_forwardGive the IUPAC name for each compound. Part 1 of 4 HO H₂C-C-CH₂CH₂ H Part 2 of 4 X OH H₂C-C-CH(CH₂CH₂), H Ś X $arrow_forward
- MCQ 3: The carbonyl group in aldehyde is A. C=O B. C-O C. CO D. CHOarrow_forward18. Ketone reduction Dicyclohexyl ketone Reduce the ketone. 1. NaBH4, ethanol 2. H3O+ H OH Dicyclohexylmethanol (88%) (a 2° alcohol)arrow_forwardOH conc H3PO4 A A MCPBA CH₂Cl₂ B NaOH H₂O 8. Identify the products A,B and C.arrow_forward
- acetone H3C, HO H3C OH The product of the following reaction is: A OH H3C OH C D OBarrow_forwardWhat alkenes are formed when each alcohol is dehydrated with TSOH? Label the major product when a mixture results. OH Xom OH a. OH b. CH₂CH3 OH C. d. CH₂CH₂CH₂CH₂OH e.arrow_forward3. What alcohol is formed when each alkene is treated with H2O in the presence of H2SO4 (as the catalyst)? а. СНЗСН-СНСНЗ b. CH3CH2CH=CH2 С. CH3 CH3arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





