
Draw the product formed when
a.
![]() , then
, then
b.![]() , then
, then
c.
![]() , then
, then
d.
(a)
Interpretation: The product formed when
Concept introduction: The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of a molecule with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 20.40P
The product formed when
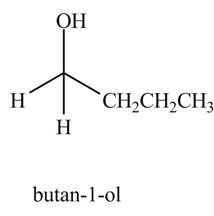
Explanation of Solution
The chemical equation when
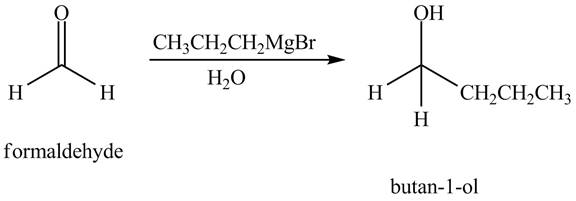
Figure 1
The above equation shows that formaldehyde reacts with
The product formed when
(b)
Interpretation: The product formed when
Concept introduction: The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of a molecule with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 20.40P
The product formed when
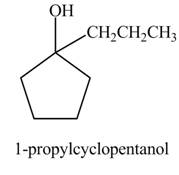
Explanation of Solution
The chemical equation when
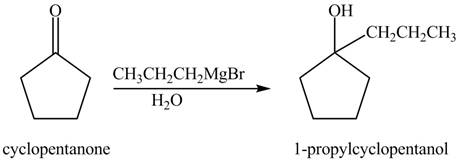
Figure 2
The above reaction indicates that that cyclopentanone reacts with
The product formed when
(c)
Interpretation: The product formed when
Concept introduction: The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of a molecule with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 20.40P
The product formed when
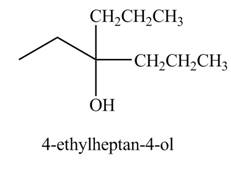
Explanation of Solution
The chemical equation when
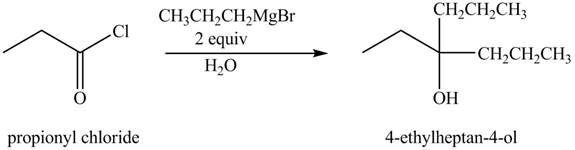
Figure 3
The above equation shows that propionyl chloride reacts with two equivalents of
The product formed when
(d)
Interpretation: The product formed when
Concept introduction: The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of a molecule with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 20.40P
The product formed when
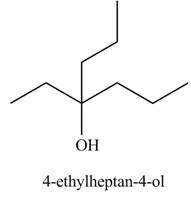
Explanation of Solution
The chemical equation when
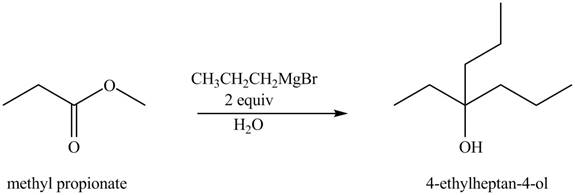
Figure 4
The above equation shows that methyl propionate reacts with two equivalents of
The product formed when
(e)
Interpretation: The product formed when
Concept introduction: The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of a molecule with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 20.40P
The products formed when
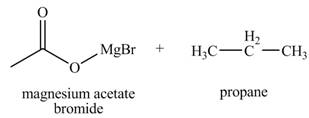
Explanation of Solution
The chemical equation when
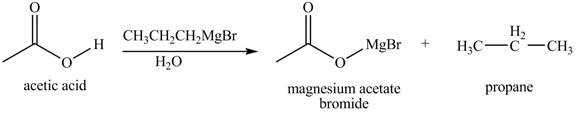
Figure 5
The above equation shows that acetic acid reacts with
The product formed when
(f)
Interpretation: The product formed when
Concept introduction: The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of a molecule with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 20.40P
The products formed when
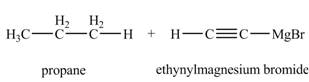
Explanation of Solution
The chemical equation when
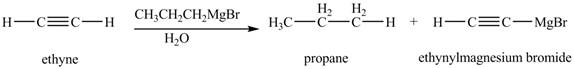
Figure 6
The above equation shows that ethyne reacts with
The product formed when
(g)
Interpretation: The product formed when
Concept introduction: The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of a molecule with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 20.40P
The product formed when
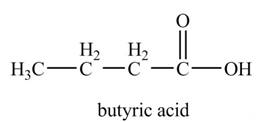
Explanation of Solution
The chemical equation when
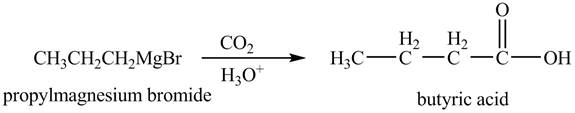
Figure 7
The above equation shows that propylmagnesium bromide reacts with
The product formed when
(h)
Interpretation: The product formed when
Concept introduction: The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of a molecule with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 20.40P
The product formed when
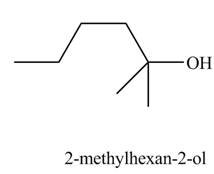
Explanation of Solution
The chemical equation when
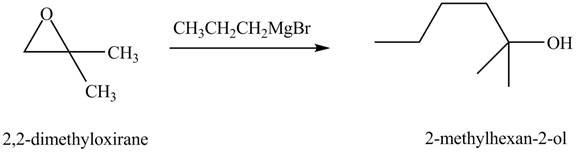
Figure 8
The above equation shows that
The product formed when
(i)
Interpretation: The product formed when
Concept introduction: The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of a molecule with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 20.40P
The product formed when

Explanation of Solution
The chemical equation when
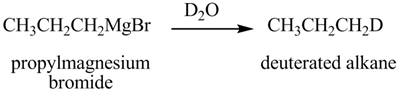
Figure 9
The above equation shows that propylmagnesium bromide reacts with
The product formed when
(j)
Interpretation: The product formed when
Answer to Problem 20.40P
The product formed when
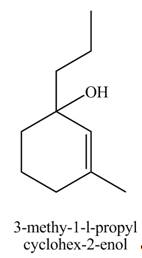
Explanation of Solution
The chemical equation when
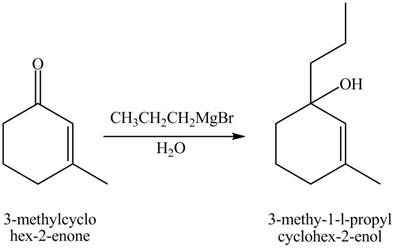
Figure 10
The compound
The product formed when
Want to see more full solutions like this?
Chapter 20 Solutions
Organic Chemistry
- What is the major product to the following reaction? 1. BH3-THF 2. NaOH, H202, H2O OH H. H. OH Warrow_forwardGive the product of the following reaction: a 'COOH COOH b COOH COOH KMnO4 но C COOH XX. COOH darrow_forward2. What is the product(s) of the following reaction? A HN B. N A NH2 [H+] -H₂O C. D. HN 3. What is/are the product(s) of the following reaction? A. B. D [H+] -H₂O C.arrow_forward
- Show how to bring about each conversion in good yield. a. b. C6H5 Cl OH COOH C6H5 COOHarrow_forwardWhat is the major product of the following reaction? 1. KMNO4, "OH, boil 2. H₂0¹arrow_forwardWhat is the best choice as the major product for this reaction? NaCN ų th OH CH2 a. b. OH он NH d. O a O b Oc O d earrow_forward
- Provide the correct product: 1. Hg(OAC)2, H2O 2. NABH4 కరరర ОН ОН OH H ОН ОН A. B. C. D. a. choice A. O b. choice B. O c. Choice C. O d. choice D.arrow_forwardQuestion 6 Give the reagents. O 1. BH3, 2. H₂O2, NaOH OmCPBA O 1. Hg(OAc)2, H₂O 2. NaBH4 NaBH4 OH2, Pd/C harrow_forwardWhat is the major product of this reaction? 1 equiv HCI -80 °C 身arrow_forward
- What is the organic product formed in the following reaction? Ono CH CH Br Br I III 0 Select one: O A. IV B. I C. II O D. III CH- CH- NH, NH, + 2 NaNH, II IV ? CH=CH-arrow_forward6S. What is the major product of this reaction? 65. NaOH a C d.arrow_forwardWhat is the reagent in this reaction? a. mCPBA, water, H+ b. H2, Pd c. NaBH4, water d. KMNO4arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





