
Concept explainers
A plasmid that is both ampicillin and tetracycline resistant is cleaved with Pstl, which cleaves within the ampicillin resistance gene. The cut plasmid is ligated with Pstl-digested Drosophila DNA to prepare a genomic library, and the mixture is used to transform E. coli K12.
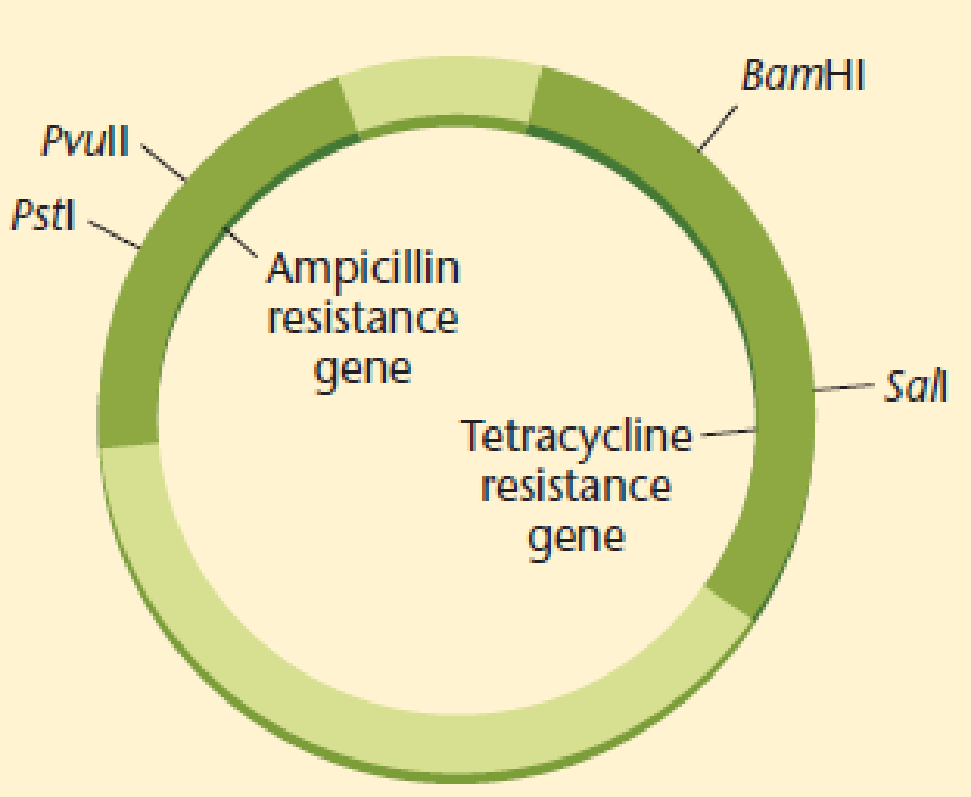
- (a) Which antibiotic should be added to the medium to select cells that have incorporated a plasmid?
- (b) If recombinant cells were plated on medium containing ampicillin or tetracycline and medium with both antibiotics, on which plates would you expect to see growth of bacteria containing plasmids with Drosophila DNA inserts?
- (c) How can you explain the presence of colonies that are resistant to both antibiotics?
(a)
To determine: The antibiotic that an individual should add to the medium to select cells that contain the recombinant plasmid.
Introduction: Plasmid refers to an circular, extrachromosomal DNA (deoxyribonucleic acid) molecule that replicates chromosome independently.
Explanation of Solution
The gene that confers resistance to tetracycline is intact in the recombinant plasmid. An individual would add tetracycline to the medium to select the recombinant plasmd in the cells. In the medium, the bacteria that have been transformed with recombinant plasmid will be resistant to tetracycline.
Thus, an individual would use tetracycline to select cells that contain the recombinant plasmid.
(b)
To determine: The plates on which an individual might see the growth of bacteria having plasmids with Drosophila DNA inserts.
Introduction: Genetically modified bacterial plasmids were the first developed vectors, used for cloning purpose.
Explanation of Solution
Cloning is a important process in which bacterial cells containing recombinant DNA can be readily identified. This process is also accomplished is through the use of selectable marker genes. Antibiotic resistant genes provide very effective selectable marker genes. Therefore, the colonies that grow on tetracycline medium but do not grow on ampicillin medium would contain the Drosophila DNA insert.
Thus, colonies that only grow in tetracycline medium probably contain the Drosophila DNA insert.
(c)
To determine: The reason that some colonies can grow on tetracycline medium as well as ampicillin medium.
Introduction: Plasmids have an origin of replication (ori) site, which makes it possible to produce several hundred copies of a plasmid in a single host cell.
Explanation of Solution
When performing cloning process, it is not necessary for incorporation of all DNA plasmids need to be cloned. A plasmid cut with a restriction enzyme generating sticky ends can self-ligate if cut ends of the plasmid rejoin. Also, if cleavage with PstI was incomplete, then no change in biological characteristics of the uncut plasmid would be expected.
Thus, self-ligation of plasmid and incomplete cleavage with PstI are responsible for the growth of colonies that are resistant to both antibiotics.
Want to see more full solutions like this?
Chapter 20 Solutions
Concepts of Genetics Plus Mastering Genetics with Pearson eText -- Access Card Package (12th Edition) (What's New in Genetics)
- Using the plasmid map of pBCH2.0 provided above, predict how many DNA fragments would be formed if this plasmid was digested with restriction enzyme BamHI.arrow_forwardA shuttle vector is a vector (usually a plasmid) constructed so that it can propagate in two different host species. One of the most common types of shuttle vectors is the yeast shuttle vector. Examples of such vectors derived from yeast are Yeast Episomal Plasmid (YEP), Yeast Integrating Plasmid (YIP) and Yeast Replicating Plasmid (YRP). Why is YEP preferred over YIP and YRP? Give your thoughts on this.arrow_forwardPlease DESCRIBE, in outline form, the method you will use to select for bacterial cells that have taken up the pL311 plasmid, and to screen those cells for the presence of plasmids that are likely to contain a cloned gene. Be sure to mention the specific media you will use. In addition, please explain the rationale behind this specific selection and screening procedure. (Remember that you have available the following types of media: (i) media containing neither kanamycin nor X-gal, (ii) media containing BOTH kanamycin and X-gal, (iii) media containing tetracycline, and (iv) media containing ampicillin.arrow_forward
- When cloning a foreign DNA fragment into a plasmid, it is often useful to insert the fragment at a site that interrupts a selectable marker(such as the tetracycline-resistance gene of pBR322). The loss of function of the interrupted gene can be used to identify clones containing recombinant plasmids with foreign DNA. With a yeast artificial chromosome (YAC) vector, it is not necessary to do this; the researcher can still distinguish vectors that incorporate large foreign DNA fragments from those that do not. How are these recombinant vectors identified?arrow_forwardWhen using a conventional plasmid cloning vector containing a b-galactosidase gene, it is possible to perform a "blue-white screen" to determine which bacteria have taken up a plasmid into which a DNA fragment as been inserted, as opposed to those that have taken up just reclosed plasmid vector, by growing the transformed cells on nutrient agar plates containing the artificial b-gal substrate X-gal. Will bacteria that have taken up a plasmid into which a DNA fragment has been inserted form a blue colony or a white colony when grown on this medium? Briefly explain why these bacteria would form a colony of the color you chose.arrow_forwardYou have set up a recombinant DNA experiment using the plasmid PBR322 as the vector (see plasmid below). You use the BamHI restriction site on the plasmid to insert the target DNA. The plasmid is then used to transform E.coli colls Is the following statement True or False? Growth of the transformed cells on agar containing both ampicillin and tetracycline will eliminate any cells that do not contain a plasmid. Clal Hindlll EcoRI Pvul BamHI Pstl amp tet PBR322 -Sall ori rop Pvull True Falsearrow_forward
- To determine if the antibiotic resistance in MH1 was carried on a plasmid, you first isolate the plasmid in MH1 using the plasmid DNA purification technique. Then, you transform bacteria that are not resistant to penicillin/ampicillin with the plasmid isolated from MH1. For the bacterial transformation experiment, you set up the three controls listed below. Match each control with its appropriate purpose (i.e. what it is controlling for) Please note: Transformed bacteria are bacteria that received the plasmid from MH1 and untransformed bacteria are bacteria that did not receive a plasmid. Testing to ensure that the bacteria used in the transformation experiment are viable (i.e. can grow on LB media) (Choose) [ Choose ) after transformation. Untransformed bacteria plated on LB only plate Testing to ensure that the bacteria used in the transformation experiment are viable (ie. can grow on LB media) before transformation. Transformed bacteria plated on LB only plate Untransformed bacteria…arrow_forwardAssume you have successfully cloned a small (200 bp) fragment of DNA into the polylinker region of a pUC18 cloning vector. Describe the appearance of transformed colonies you would expect to see on each of the following plates: plain media, media containing ampicillin, media containing tetracycline, media containing ampicillin and X-Gal.arrow_forwardThe modifiedplasmid is reintroduced back into Rhizobium(step 4) and the genetically transformed bacteria are then selected based on the amp and lacZgenes present within the plasmid. The plasmid may or may not integrate the BBW resistancegene. The treated bacteria may or may not take up the modified plasmid. a) Complete the table below with a yes or no in each space stating whether you would expect these bacteria to grow or not. Type of treated bacteria culture plate(no amp) Culture plate treated with ampicillin Plasmid not taken up Plasmid taken up (WithoutBBW resistancegene) Plasmid taken up (WithBBW resistancegene) B) Outline how the genetically transformed bacteria containing the BBW resistance gene can beselected based on the amp and lacZgenes present within the plasmid.arrow_forward
- A plasmid, pUC18, contains the ampicillin-resistance gene, the origin of replication, and the ß - gal gene, which codes for the B-galactosidase protein. This protein can break down the synthetic chemical X-gal, producing a blue product that stains the entire cell blue (but is harmless to the bacteria). At the beginning of the B-gal gene there are several unique restriction sites (some of them are shown in the diagram below). You wish to clone a 1.0-kb Xbal fragment into the pUC18 plasmid, so you cut the plasmid with Xbal and, after removing the enzyme, mix the Xbal-cut plasmid with the 1.0-kb fragment, ligate, and transform competent bacteria. Pati Xbal EcoRI B-gal A Amp ori Figure: pUC18 plasmid map (a) On what medium would you grow your transformed bacteria? (b) Do you expect the bacteria carrying plasmid pUC18 (without the insert) to be blue or white when grown in the presence of X-gal? Explain.arrow_forwardMany resistance mechanisms are encoded on plasmids. These mechanisms are of great clinical significance, because they can spread very easily through horizontal gene transfer. A culture of the bacterial isolate is grown, and plasmid DNA is isolated using a spin column-based solid phase extraction method. The purified plasmid DNA is then submitted for next-generation sequencing. Bioinformatic analyses of the sequencing results suggests that the following gene is likely involved in antibiotic resistance: > putative antibiotic resistance gene ATGCGTGTATTAGCCTTATCGGCTGTGTTTTTGGTGGCATCGATT ATCGGAATGCCTGCGGTAGCAAAGGAATGGCAAGAAAACAAAAGT TGGAATGCTCACTTTACTGAACATAAATCACAGGGCGTAGTTGTG CTCTGGAATGAGAATAAGCAGCAAGGATTTACCAATAATCTTAAA CGGGCGAACCAAGCATTTTTACCCGCATCTAGTGCGAAAATTCCC AATAGCTTGATCGCCCTCGATTTGGGCGTGGTTAAGGATGAACAC CAAGTCTTTAAGTGGGATGGACAGACGCGCGATATCGCCACTTGG AATCGCGATCATAATCTAATCACCGCGATGAAATATTCAGTTGTG CCTGTTTATCAAGAATTTGCCCGCCAAATTGGCGAGGCACGTATG…arrow_forwardIn this western blot, the levels of phosphorylated TBK (PTBK) decrease with increasing amounts/expression of the viral protein (VP). Figure description: Increasing amounts of a plasmid expressing the viral protein (0.5, 1, or 2ug) were cotransfected with TBK1 expression plasmid. Cells were harvested 24 h post-transfection and analyzed for phosphorylated TBK1 (anti- TBK1 Ser172), total TBK1 (anti-TBK1), B-actin (anti-B-actin), and viral protein (anti-VP) expression by Western blot analysis. VP PTBK1 (S172) TBK1 actin Virus proteins O True O Falsearrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





