
Concept explainers
(a)
Interpretation:
The stronger acid has to be determined.
Concept Introduction:
Relative Acid strength: As going down the group, the size of the atoms increases. The larger the size, the negative charge can be easily polarizable over the larger volume of the atom. Hence, the proton attached to the larger sized atom, the acid becomes the stronger.
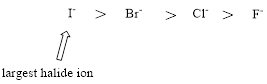
(b)
Interpretation:
The stronger acid has to be determined.
Concept Introduction:
Relative Acid strength: As going down the group, the size of the atoms increases. The larger the size, the negative charge can be easily polarizable over the larger volume of the atom. Hence, the proton attached to the larger sized atom, the acid becomes the stronger.
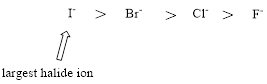
(c)
Interpretation:
The stronger acid has to be determined.
Concept Introduction:
Relative Acid strength: As going down the group, the size of the atoms increases. The larger the size, the negative charge can be easily polarizable over the larger volume of the atom. Hence, the proton attached to the larger sized atom, the acid becomes the stronger.
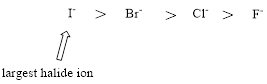
(d)
Interpretation:
The stronger acid has to be determined.
Concept Introduction:
Relative Acid strength: As going down the group, the size of the atoms increases. The larger the size, the negative charge can be easily polarizable over the larger volume of the atom. Hence, the proton attached to the larger sized atom, the acid becomes the stronger.
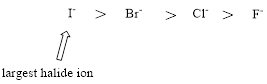
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Essential Organic Chemistry, Global Edition
- Use Table 14.3 to help order the following acids from strongest to weakest HNO3,H2O,NH4+,C5H5NH+arrow_forward. A strong acid has a weak conjugate base, whereas a weak acid has a relatively strong conjugate base. Explain.arrow_forwardSeveral acids and their respective equilibrium constants are: Which is the strongest acid? Which is the weakest acid? Which acid has the weakest conjugate base? Which acid has the strongest conjugate base?arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





