
Essential Organic Chemistry, Global Edition
3rd Edition
ISBN: 9781292089034
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 41P
- a. List the following carboxylic acids in order from strongest acid to weakest acid:
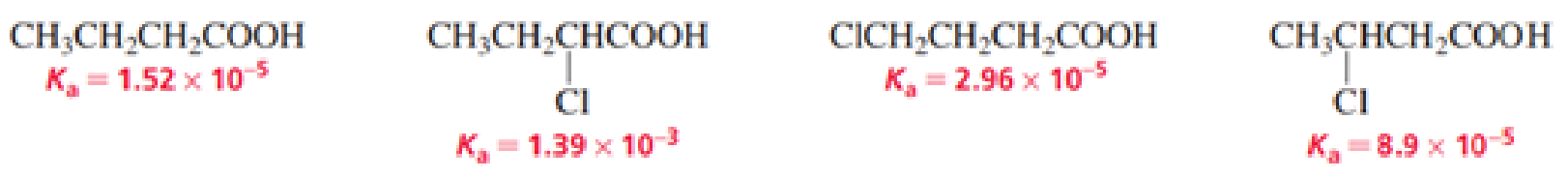
- b. How does the presence of an electronegative substituent such as Cl affect the acidity of a
carboxylic acid ? - c. How does the location of the substituent affect the acidity of the carboxylic acid?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Why are phenols more acidic than alcohol? Explain.
Alkynes exhibit acidic properties because of the differences in the electronegativity of sp-
hybridized carbons. Which of the following statements best explains the acidic property of
acetylene?
A. The electronegativity of sp hybridized carbon decreases bond dipole between C and H.
B. Hydrogen atom becomes ionized due to sp-s repulsion.
C. The sp-sp orbital overlap attracts the electron cloud between C and H.
D. The pi bonds in the triple bond promote ionization of carbon into carbocations
How does the presence of an electronegative substituent such as Cl affect the acidity of a carboxylic acid?
Chapter 2 Solutions
Essential Organic Chemistry, Global Edition
Ch. 2.1 - Which of the following are not acids? CH3COOH CO2...Ch. 2.1 - Draw the products of the acidbase reaction when a....Ch. 2.1 - a.What is the conjugate acid of each of the...Ch. 2.2 - a. Which is a stronger acid, one with a pKa of 5.2...Ch. 2.2 - Prob. 5PCh. 2.2 - Antacids are compounds that neutralize stomach...Ch. 2.2 - Are the following body fluids acidic or basic? a....Ch. 2.3 - Draw the conjugate acid of each of the following:...Ch. 2.3 - a. Write an equation showing CH3OH reacting as an...Ch. 2.3 - Prob. 10P
Ch. 2.3 - a. Which is a stronger base, CH3COO or HCOO? (The...Ch. 2.3 - Using the pKa values in Section 2.3, rank the...Ch. 2.4 - Prob. 13PCh. 2.5 - Prob. 14PCh. 2.5 - Ethyne has a pKa value of 25, water has a pKa...Ch. 2.5 - Which of the following bases can remove a proton...Ch. 2.6 - List the ions (CH3, NH2, HO, and F) in order from...Ch. 2.6 - List the carbanions shown in the margin in order...Ch. 2.6 - Which is a stronger acid?Ch. 2.6 - a. Draw the products of the following reactions: A...Ch. 2.6 - List the halide ions (F, Cl, Br, and I) in order...Ch. 2.6 - a. Which is more electronegative, oxygen or...Ch. 2.6 - Which is a stronger acid? a. HCl or HBr b....Ch. 2.6 - a. Which of the halide ions (F, Cl, Br, and I) is...Ch. 2.6 - Which is a stronger base? a. H2O or HO b. H2O or...Ch. 2.7 - Which is a stronger acid? a. CH3OCH2CH2OH or...Ch. 2.7 - Which is a stronger base?Ch. 2.8 - Fosamax has six acidic groups. The structure of...Ch. 2.8 - Which is a stronger acid? Why?Ch. 2.10 - For each of the following compounds (shown in...Ch. 2.10 - Prob. 33PCh. 2.11 - Write the equation that shows how a buffer made by...Ch. 2.11 - What products are formed when each of the...Ch. 2 - a. List the following alcohols in order from...Ch. 2 - Which is a stronger base? a. HS or HO b. CH3O or...Ch. 2 - Prob. 40PCh. 2 - a. List the following carboxylic acids in order...Ch. 2 - For the following compound, a. draw its conjugate...Ch. 2 - List the following compounds in order from...Ch. 2 - For each of the following compounds, draw the form...Ch. 2 - Give the products of the following acidbase...Ch. 2 - Prob. 46PCh. 2 - For each compound, indicate the atom that is most...Ch. 2 - Tenormin, a member of the group of drugs known as...Ch. 2 - From which acids can HO remove a proton in a...Ch. 2 - Prob. 50PCh. 2 - Which is a stronger acid? a. CH29CHCOOH or...Ch. 2 - Prob. 52PCh. 2 - Prob. 53PCh. 2 - How could you separate a mixture of the following...Ch. 2 - Prob. 1PCh. 2 - Prob. 2PCh. 2 - Draw the products of the following acidbase...Ch. 2 - Prob. 4PCh. 2 - Prob. 5PCh. 2 - Prob. 6PCh. 2 - Prob. 7PCh. 2 - Prob. 8PCh. 2 - Prob. 9PCh. 2 - Prob. 10PCh. 2 - Prob. 11PCh. 2 - Prob. 12PCh. 2 - Prob. 13PCh. 2 - Prob. 14P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Why are carboxylic acids more acidic than phenols?arrow_forward18. Which of the following compounds DOES NOT exhibit acidic properties? A. Adipic acid B. Phenol C. Toluene D. Benzenearrow_forward16. What is one use for sodium and potassium salts of long-chain carboxylic acids? Oa. food preservative b. athletes foot treatment C. soaps d. pH buffer e. blood anticoagulantarrow_forward
- 1. Give at least four medicinal uses of salicylic acid. 2. What are the roles sulfuric acid in the synthesis of salicylic acid? 3. What are the effects of the carboxyl group to the activity of salicylic acid?arrow_forwardDoes alcohol, ketones, and aldehydes considered acidic or a basic?arrow_forwardWhy is phenol a stronger kind of acid than other kinds of alcohol?arrow_forward
- Draw the following reactions and label it properly 1. stearic acid reaction with water chemical equation 2. Reaction with NaOH with benzoic acid 3. Reaction with NaOH with acetic acid 4. Reaction with NaOH with stearic acid 5. Reaction with sodium carbonate with acetic acidarrow_forward1. Write a balanced chemical equation to show the dissociation of Butanoic acid (C3H7COOH). Physical states must be included. 2. When butanoic acid (C3H7COOH) reacts with the strong base potassium hydroxide (KOH), potassium butanoate (C3H7COOK), a salt of the weak acid is formed. Write a balanced chemical equation to show the dissociation of the potassium butanoate (C3H7COOK). Physical states must be included.arrow_forwardWhat will happen if you add a base to a carboxylic acid? The acidic proton of the carboxylic acid will be donated to the base. Any of the protons of the carboxylic acid will be donated to the base. The base will donate an acidic proton to the carboxylic acid The base will donate any of its protons to the carboxylic acidarrow_forward
- Which member of each pair is the stronger base? a. ethylamine or aniline b. ethylamine or ethoxide ion c. phenolate ion or ethoxide ion d. phenolate ion or acetate ionarrow_forward18.12 Identify the compound in each pair that has the higher boiling point. Explain. a. CH3-CH,-CH,-CH3 or CH3-CH2 CH,NH, b. CH3-NH, or CH3-CH2-NH2 NH2 c. CH3-CH2 -CH2-OH or CH3-CH-CH;arrow_forwardWhat is the role of phenolphthalein in the neutralization reaction? Draw the structure of phenolphthalein under acidic and basic conditions.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY