
Organic Chemistry (6th Edition)
6th Edition
ISBN: 9781260119107
Author: Janice Gorzynski Smith
Publisher: McGraw Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 46P
What is the
a. 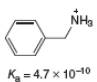
b. 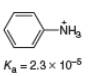
c. 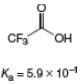
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
How does the reaction shown effect the water solubilty of the product?
H
HCI
dhe
A. product is more water soluble than the parent compound
B. product is less water soluble than the parent compound
What does a value of K eq greater than 1 mean?A. More reactants than products exist at equilibrium.B. More products than reactants exist at equilibrium.C. The rate of the forward reaction is high atequilibrium.D. The rate of the reverse reaction is high atequilibrium.
The boiling points of a range of organic compounds (A-D) of similar Mol. Wt.s are given below.
OH
OMe
A
в
D
b.p. 46-50°C
Explain why the boiling point of C is higher than that of D.
Explain why the boiling point of B is higher than that of A.
Explain why the boiling point of C is higher than that of A.
Which of these four compounds is most acidic? What makes it so acidic?
b.p. 78'C
b.p. 141'C
b.p. 79'C
(i)
(ii)
(iii)
(iv)
Chapter 2 Solutions
Organic Chemistry (6th Edition)
Ch. 2.1 - a. Which compounds are Bronsted-Lowry acids:...Ch. 2.2 - a. Draw the conjugate acid of each base:...Ch. 2.2 - Label each statement as True or False.
a. is the...Ch. 2.2 - Decide which compound is the acid and which is the...Ch. 2.2 - Draw the products formed from the acid-base...Ch. 2.3 - Which compound in each pair is the stronger acid?...Ch. 2.3 - Use a calculator when necessary to answer the...Ch. 2.3 - Rank the conjugate bases of each of group of acids...Ch. 2.3 - Problem-2.10 Considers two acids: (formic acid,)...Ch. 2.3 - Prob. 11P
Ch. 2.4 - Draw the products of each reaction and determine...Ch. 2.4 - Prob. 13PCh. 2.5 - Without reference to a pKa table, decide which...Ch. 2.5 - Rank the labeled H atoms in the following compound...Ch. 2.5 - Which hydrogen in pseudoephedrine, the nasal...Ch. 2.5 - Which compound in each pair is the stronger acid?...Ch. 2.5 - Glycolic acid, HOCH2CO2H, is the simplest member...Ch. 2.5 - Explain the apparent paradox. HBr is a stronger...Ch. 2.5 - The CH bond in acetone, (CH3)2C=O, has a pKa of...Ch. 2.5 - Prob. 23PCh. 2.5 - For each pair of compounds: [1] Which indicated H...Ch. 2.5 - Rank the compounds in each group in order of...Ch. 2.5 - Prob. 26PCh. 2.5 - Prob. 27PCh. 2.6 - Prob. 28PCh. 2.7 - Problem 2.29
Compounds like amphetamine that...Ch. 2.8 - Problem 2.30 Which species are Lewis bases?
a. b....Ch. 2.8 - Which species are Lewis acids?
a. b. c. d.
Ch. 2.8 - For each reaction, label the Lewis acid and base....Ch. 2.8 - Prob. 33PCh. 2.8 - Prob. 34PCh. 2.8 - Label the Lewis acid and base. Use curved arrow...Ch. 2 - 2.36 Propranolol is an antihypertensive agent—that...Ch. 2 - 2.37 Amphetamine is a powerful stimulant of the...Ch. 2 - 2.38 What is the conjugate acid of each base?
a....Ch. 2 - 2.39 What is the conjugate base of each acid?
a....Ch. 2 - Draw the products of each proton transfer...Ch. 2 - Prob. 43PCh. 2 - What is Ka for each compound? Use a calculator...Ch. 2 - What is the pKa for each compound? a. b. c.Ch. 2 - Which of the following bases are strong enough to...Ch. 2 - Draw the products of each reaction. Use the pKa...Ch. 2 - a. What is the conjugate acid of A? b. What is the...Ch. 2 - Dimethyl ether (CH3OCH3) and ethanol (CH3CH2OH)...Ch. 2 - 2.59 Atenolol is a (beta) blocker, a drug used to...Ch. 2 - 2.60 Use the principles in Section 2.5 to label...Ch. 2 - 2.61 Label the three most acidic hydrogen atoms in...Ch. 2 - Prob. 66PCh. 2 - 2.63 Classify each compound as a Lewis base, a...Ch. 2 - 2.64 Classify each species as a Lewis acid, a...Ch. 2 - Label the Lewis acid and Lewis base in each...Ch. 2 - 2.66 Draw the products of each Lewis acid-base...Ch. 2 - Prob. 71PCh. 2 - 2.68 Answer the following questions about the four...Ch. 2 - Prob. 73PCh. 2 - 2.70 Hydroxide can react as a Brønsted-Lowry base...Ch. 2 - 2.71 Answer the following questions about esmolol,...Ch. 2 - Prob. 76PCh. 2 - Prob. 77PCh. 2 - Prob. 82P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 24. Rank the following in decreasing order of basicity (hint: the stronger the acid, the weaker the conjugate base). a. A. CH3NH₂ b. CH3CH₂O B. OH C.F d. D. NH3 -Harrow_forward17. The correct reagent is:arrow_forwardWhat is the hybridization of O in the aldehyde methanal? sp² a. 1. b. sp c. sp³ Which of the following compounds is most acidic? a. p-nitrophenol 2. b. p-hydroxyaniline phenol C.arrow_forward
- 16.34 For which compounds can a second resonance structure be drawn? Draw an additional resonance structure for each resonance-stabilized compound. a. OCH₂ b. OCH₂ C. d. NHCH,arrow_forwardExplain why HCl reacts more rapidly with compound A than compound B. A H H₂C H CH H Barrow_forwardWhich compound in each pair is the stronger acid? c. CH3COOH or O,NCH,COOH a. CICH,COOH or FCH,COOH b. Cl,CHCH,OH or Cl,CHCH,CH,OHarrow_forward
- Which is the stronger acid? a. CH3CH3 or HC CH b. H2C CH2 or HC CH c. H2C CH2 or CH3CH3arrow_forwardMC 15 How many H atoms of the compound shown can be replaced by D with D2O under basic conditions? a. b. C. d. e. 1 63 ENT 2 3 9arrow_forwardWhat is the product of the following reaction: Zn(Hg) HC он OH B) C ii. В ii. A iv. None 39arrow_forward
- Draw the structure of a constitutional isomer of compound B that fits each description. a. an isomer that is at least 105 times more acidic than B b. an isomer that is at least 105 times less acidic than B c. an isomer that is comparable in acidity to Barrow_forward1. b. Which is a stronger base? 2. orarrow_forwarda. b. Draw the correct product for the each of the following O H P.(Ph)3 O Bind H P(Ph)3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY