
(a)
Interpretation:
The potassium element needs to be classified as metals, non-metals,
Concept introduction:
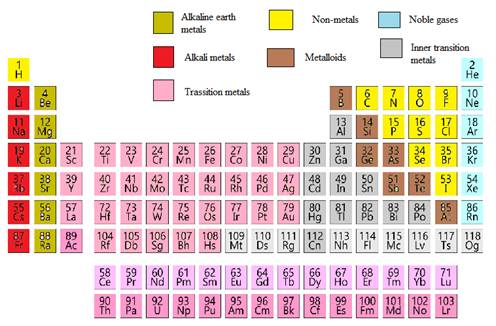
In the modern periodic table, all the known elements are arranged in organized tabular manner (having groups and periods) based on their
Elements are also classified based on metal, non-metals, transition element and post- transition elements in modern periodic table.
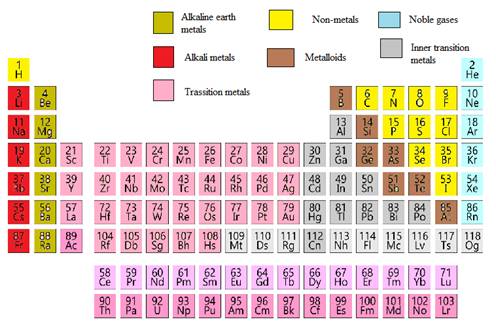
Modern periodic table is represented as follows:
(b)
Interpretation:
The cadmium element needs to be classified as metals, non-metals, transition elements or post transition elements.
Concept introduction:
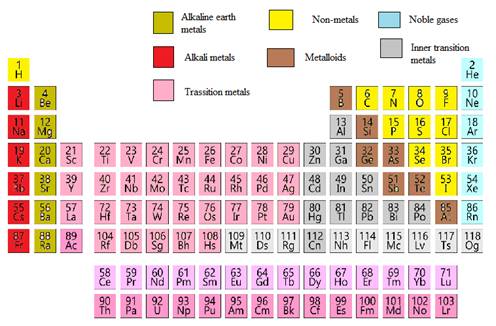
In the modern periodic table, all the known elements are arranged in organized tabular manner (having groups and periods) based on their atomic number. In this table, atomic number, chemical symbol with its chemical name and average atomic mass is written.
Elements are also classified based on metal, non-metals, transition element and post- transition elements in modern periodic table.
Modern periodic table is represented as follows:
(c)
Interpretation:
The aluminum element needs to be classified as metals, non-metals, transition elements or post transition elements.
Concept introduction:
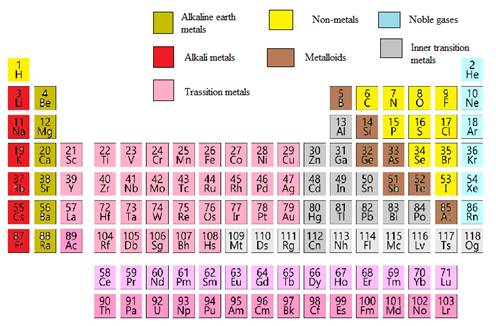
In the modern periodic table, all the known elements are arranged in organized tabular manner (having groups and periods) based on their atomic number. In this table, atomic number, chemical symbol with its chemical name and average atomic mass is written.
Elements are also classified based on metal, non-metals, transition element and post- transition elements in modern periodic table.
Modern periodic table is represented as follows:
(d)
Interpretation:
The antimony element needs to be classified as metals, non-metals, transition elements or post transition elements.
Concept introduction:
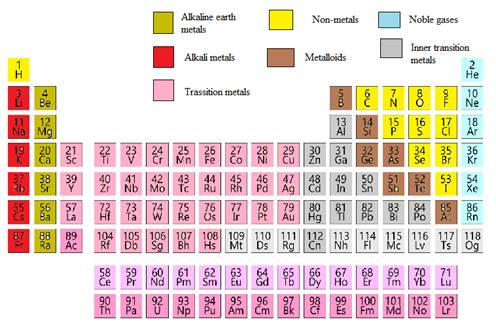
In the modern periodic table, all the known elements are arranged in organized tabular manner (having groups and periods) based on their atomic number. In this table, atomic number, chemical symbol with its chemical name and average atomic mass is written.
Elements are also classified based on metal, non-metals, transition element and post- transition elements in modern periodic table.
Modern periodic table is represented as follows:
(e)
Interpretation:
The phosphorus element needs to be classified as metals, non-metals, transition elements or post transition elements.
Concept introduction:
In the modern periodic table, all the known elements are arranged in organized tabular manner (having groups and periods) based on their atomic number. In this table, atomic number, chemical symbol with its chemical name and average atomic mass is written.
Elements are also classified based on metal, non-metals, transition element and post- transition elements in modern periodic table.
Modern periodic table is represented as follows:
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Chemistry: Principles and Reactions
- Mass spectrometric analysis showed that there are four isotopes of an unknown element having the following masses and abundances: Three elements in the periodic table that have atomic weights near these values are lanthanum (La), atomic number 57, atomic weight 138.9055; cerium (Ce), atomic number 58, atomic weight 140.115; and praseodymium (Pr), atomic number 59, atomic weight 140.9076. Using the data above, calculate the atomic weight, and identify the element if possible.arrow_forwardArrange the following in the order of increasing mass. (a) a potassium ion, K+ (b) a phosphorus molecule, P4 (c) a potassium atom (d) a platinum atomarrow_forwardFrom the following written description, write the balanced chemical equation for the reaction including state symbols. A diatomic gaseous molecule that contains 17 protons per atom is reacted with a solid element that has an atomic number of 19 to yield an ionic compound.arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





