
Concept explainers
What is the
a. 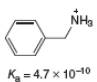
b. 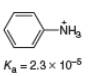
c. 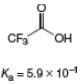
(a)
Interpretation: The value of
Concept introduction: The equilibrium constant of an acid dissociation reaction depends on the strength of an acid involved in the reaction. The
Acid dissociation constant
For an acid dissociation reaction,
The strength of an acid decreases as the value of
Answer to Problem 2.46P
The value of
Explanation of Solution
The given value of
The value of
Substitute the value of
Simplify the above equation,
Hence, the value of
The value of
(b)
Interpretation: The value of
Concept introduction: The equilibrium constant of an acid dissociation reaction depends on the strength of an acid involved in the reaction. The
Acid dissociation constant
For an acid dissociation reaction,
The strength of an acid decreases as the value of
Answer to Problem 2.46P
The value of
Explanation of Solution
The given value of
The value of
Substitute the value of
Simplify the above equation,
Hence, the value of
The value of
(c)
Interpretation: The value of
Concept introduction: The equilibrium constant of an acid dissociation reaction depends on the strength of an acid involved in the reaction. The
Acid dissociation constant
For an acid dissociation reaction,
The strength of an acid decreases as the value of
Answer to Problem 2.46P
The value of
Explanation of Solution
The given value of
The value of
Substitute the value of
Simplify the above equation.
Hence, the value of
The value of
Want to see more full solutions like this?
Chapter 2 Solutions
PKG ORGANIC CHEMISTRY
- 24. Rank the following in decreasing order of basicity (hint: the stronger the acid, the weaker the conjugate base). a. A. CH3NH₂ b. CH3CH₂O B. OH C.F d. D. NH3 -Harrow_forward17. The correct reagent is:arrow_forwardWhat is the hybridization of O in the aldehyde methanal? sp² a. 1. b. sp c. sp³ Which of the following compounds is most acidic? a. p-nitrophenol 2. b. p-hydroxyaniline phenol C.arrow_forward
- 16.34 For which compounds can a second resonance structure be drawn? Draw an additional resonance structure for each resonance-stabilized compound. a. OCH₂ b. OCH₂ C. d. NHCH,arrow_forwardExplain why HCl reacts more rapidly with compound A than compound B. A H H₂C H CH H Barrow_forwardWhich compound in each pair is the stronger acid? c. CH3COOH or O,NCH,COOH a. CICH,COOH or FCH,COOH b. Cl,CHCH,OH or Cl,CHCH,CH,OHarrow_forward
- Which is the stronger acid? a. CH3CH3 or HC CH b. H2C CH2 or HC CH c. H2C CH2 or CH3CH3arrow_forwardMC 15 How many H atoms of the compound shown can be replaced by D with D2O under basic conditions? a. b. C. d. e. 1 63 ENT 2 3 9arrow_forwardWhat is the product of the following reaction: Zn(Hg) HC он OH B) C ii. В ii. A iv. None 39arrow_forward
- Draw the structure of a constitutional isomer of compound B that fits each description. a. an isomer that is at least 105 times more acidic than B b. an isomer that is at least 105 times less acidic than B c. an isomer that is comparable in acidity to Barrow_forward1. b. Which is a stronger base? 2. orarrow_forwarda. b. Draw the correct product for the each of the following O H P.(Ph)3 O Bind H P(Ph)3arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

