
Concept explainers
In an alternate universe, the smallest negatively chargedparticle, analogous to our electron, is called a blorvek. Todetermine the charge on a single blorvek, an experiment likeMillikan’s with charged oil droplets ‘as carried out, and thefollowing results were recorded:
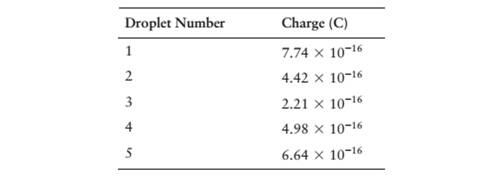
(a) Based on these observations, what is the largest possiblevalue for the charge on a blorvek?
(b) Further experiments found a droplet with a charge of
Trending nowThis is a popular solution!

Chapter 2 Solutions
CHEMISTRY-TEXT
- 2-89 Assume that a new element has been discovered with atomic number 117. Its chemical properties should be similar to those of astatine (At). Predict whether the new element’s ionization energy will be greater than, the same as, or smaller than that of: (a)At (b)Raarrow_forwardWhich of the following is true about an individual atom? Explain. a. An individual atom should be considered to be a solid. b. An individual atom should be considered to be a liquid. c. An individual atom should be considered to be a gas. d. The state of the atom depends on which element it is. e. An individual atom cannot be considered to be a solid, liquid, or gas. Justify your choice, and for choices you did not pick, explain what is wrong with them.arrow_forward2.84 Early attempts to arrange the elements often focused on atomic weight. Mendeleev considered a number of properties in addition to atomic weight, so he realized that some elements seemed out of place when ordered by atomic weight. Using the modern periodic table, identify elements for which Mendeleev must have had to switch the order in order to get the correct sequence of elements.arrow_forward
- Discussing this chapter, a classmate says, “Since elementsthat form cations are metals and elements that form anionsare nonmetals, elements that do not form ions are metalloids.”Do you agree or disagree?arrow_forward(a) Group 1A positive + The elements in group 1A configuration. an electron (obtain a charge) in order to achieve a noble gas ...arrow_forwardRubidium and bromine atoms are depicted at right. (a) What monatomic ions do they form? (b) What electronic feature characterizes this pair of ions, and which noble gas are they related to? (c) Which pair best represents the relative ionic sizes?arrow_forward
- Illustrate the sharing of electrons of each covalent compound using the Bohr Model. Apply the octet rule if possible. (A) A molecule composed of two chlorine atoms. (b) A molecule composed of a hydrogen atom and a bromine atom. (c) A molecule composed of 2 hydrogen atoms. (d) A molecule composed of a hydrogen atom and a chlorine atom.arrow_forwardQ2. This question is about the periodicity of the Period 3 elements. (a) State and explain the general trend in first ionisation energy across Period 3. Give one example of an element which deviates from the general trend in first (b) ionisation energy across Period 3. Explain why this deviation occurs. Page 3 of 12 (c) The table shows successive ionisation energies of an element Y in Period 3. lonisation number 1 2 3 4 5 6 7 8 lonisation energy kJ mol- 1000 2260 3390 4540 6990 8490 27 100 31 700 Identify element Y Explain your answer using data from the table. (d) Identify the Period 3 element that has the highest melting point. Explain your answer by reference to structure and bonding.arrow_forwardHow many inner, outer, and valence electrons are present in an atom of each of the following elements? (a) Br (b) Cs (c) Cr (d) Sr (e) Farrow_forward
- a ) Eva l u a te t h e expre s s i o n s 2 x 1, 2 x (1 + 3),2 x(1 + 3 + 5), and 2 x (1 + 3 + 5 + 7). (b) How do the atomic numbers of the noble gases relate to the numbersfrom part (a)? (c) What topic discussed is thesource of the number “2” in the expressions in part (a)?arrow_forwardAccording to Coulomb’s law, what happens to the potential energy of two oppositely charged particles as they get closer together?(a) Their potential energy decreases.(b) Their potential energy increases.(c) Their potential energy does not change.arrow_forward1 (a)What charge is expected on F? Enter answer as -# or +# where # is the charge. b What charge is expected on Xe? Enter answer as -# or +# where # is the charge. c What charge is expected on P? Enter answer as -# or +# where # is the charge.arrow_forward
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning





