
Concept explainers
PRACTICE PROBLEM
Cyclobutadiene (below) is like benzene in that it has alternating single and double bonds in a ring. However, its bonds are not the same length, the double bonds being shorter than the single bonds; the molecule is rectangular, not square. Explain why it would be incorrect to write resonance structures as shown.

Interpretation: The given resonance structure written in an incorrect way, is to be explained.
Concept introduction:
A resonance structure is very helpful, as it describes the molecule, and the ions, completely.
While writing a resonance structure, only the delocalization of PI–electrons takes place. The nuclei positions remain the same in all the structures.
All the resonance structures must be proper Lewis structures.
More stable the structure, greater is the contribution to the resonance hybrid.
Answer to Problem 1PP
Solution:
In the two representations, the four carbon atoms occupy different positions.
Explanation of Solution
Given information:
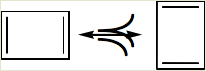
According to the rules of writing resonance structures, in all the structures, the positions of the nuclei remain the same and only the delocalization of PI - electrons occurs and the bonding framework of a molecule must remain intact. In the given structure of cyclobutadiene, it has alternating double and single bonds, but in the representation, the position of the four carbon atoms is different. Thus, it is the incorrect way of writing a resonance structure.
In the two representations, the positions of the four carbons atoms are different.
Want to see more full solutions like this?
Chapter 2 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Chemistry: A Molecular Approach (4th Edition)
Biochemistry: Concepts and Connections (2nd Edition)
Organic Chemistry (8th Edition)
Anatomy & Physiology (6th Edition)
Human Physiology: An Integrated Approach (8th Edition)
Microbiology: An Introduction
- Don't used Ai solutionarrow_forwardPlease correct answer and don't used hand raitingarrow_forward1. For each of the following, predict the products of the reaction by writing a balance net ionic equation for each. If no reaction is expected, then write NO REACTION. (a) AgNO3 (aq) is mixed with Na2CO3 (aq). (b) An aqueous solution of ammonium sulfate is added to an aqueous solution of calcium chloride. (c) RbI (aq) is added to Pb(NO3)2 (aq). (d) NaCl (s) is added to AgNO3 (aq).arrow_forward
- 4. Determine the amount in grams of AgCl (s) formed when 2.580 g AgNO3(s) is added to 45.00 mL of a 0.1250 M CrCl3 (aq) (The other product is aqueous chromium (III) nitrate) 5. Determine the amount (in grams) of Cobalt (II) phosphate formed when an aqueous solution of 30.0 ml of 0.450 M Sodium Phosphate is mixed with 20.0 mL of 0.500 M aqueous solution of cobalt (II) nitrate. (The other product is aqueous sodium nitrate)arrow_forward7. Consider the following reaction that describes the dissolution of copper metal in nitric acid: Cu (s) + 4 HNO3 (aq) → Cu(NO3)2 (aq) + 2 H₂O (1) + 2 NO2 (g) How many mL of 3.50 M HNO3 (aq) are required to dissolve 20.00 g Cu?arrow_forwardPlease correct answer and don't used hand raitingarrow_forward
- Don't used Ai solutionarrow_forward3. An unknown element, X, combines with chlorine to give a substance with the formula XC14. A chlorine analysis of the substance indicates that it contains 83.47% chlorine by mass. What element is X and what is the formula of this compound? (Hint: to identify an element or compound, identify its molar mass. Remember that Molar Mass = (grams A)/(moles A). Solve for each individually and then divide them to find molar mass.)arrow_forward1. When hydrogen sulfide (H2S, MM = 34.08 g/mol) gas is bubbled into a solution of sodium hydroxide (NaOH, 40.00 g/mol), sodium sulfide (Na2S, 78.04 g/mol) and water (18.02 g/mol) are produced according to the balanced chemical equation shown below? H2S 2 NaOH --> Na2S 2 H₂O (a) Assuming the reaction goes to completion, how many grams of sodium sulfide are formed if 2.50g of hydrogen sulfide is bubbled into a solution containing 1.85g of NaOH? (20 pts) (b) Which reactant and how much of it remains after the reaction has been completed? (15 pts) (c) If only 0.400g of sodium sulfide was recovered, what is the percent yield of this reaction (5 pts)arrow_forward
- The organic compound MTBE (methyltertiarybutylether) is used as a fuel additive that allows gasoline to burn more cleanly thus leading to a reduction in pollution. Recently, however, MTBE has been found in the drinking water of a number of communities. As a result several states are phasing out the use of MTBE as a fuel additive. A combustion experiment using 10.00 g of MTBE was found to produce 24.97g of CO2 and 12.26 g of H2O. (a) What is the empirical formula of MTBE assuming it contains C, H, and O only? (b) The molar mass of MTBE was experimentally determined to be 88.1 g/mol. Using this information what is the molecular formula of MTBEarrow_forwardPart 4: Provide a detailed retrosynthetic analysis and a plausible forward synthesis the following molecule. храдо ofarrow_forward3A: Starting with benzocyclobutene, synthesize the naphthalene derivative below.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
