
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 1.9, Problem CQ
Interpretation Introduction
Interpretation:
The reasonable contributing structure for the amide bond of the molecule has to be shown.
Concept introduction:
Resonance: The delocalization of electrons which is characterized as several structural changes.
Amide:
A carbon atom is double-bonded to an oxygen atom
If the carbonyl carbon is attached with nitrogen is called as amide.
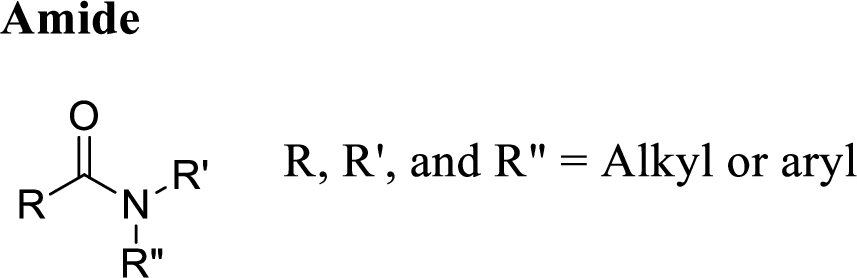
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Complete Table 3 (Remember that the values pertain to the Central atoms only. In this table, you will have more than one central atom so report more than one value, ie. 3 / 3)
Table 3 Molecules with Multiple Central Atoms
Molecule
C2H4
H2O2.
CH3OH.
CH3NH2
Number of Bond Pairs
/
/
/
/
Number of Lone Pairs
/
/
/
/
Number of Electron Domains
/
/
/
/
Molecular Geometry (Shape)
/
/
/
/
Bond Angle
/
/
/
/
Polar or Non-Polar
Nonpolar
Polar
Polar
Polar
2. Answer parts a-e using the skeletal structure of the molecule shown below.
NE
#
NC=N;
+
NÁ
a. Add all implied hydrogen atoms to the skeletal structure.
b. How many tetrahedral carbons are in the molecule? What hybridization do tetrahedral
carbon atoms have?
c. Consider the bond marked by *. Based on what you know about electronegativity trends,
which atom in the bond is more electron rich? Draw a dipole arrow pointing toward the
more electron rich atom.
d. Label each N atom with its hybridization (sp, sp², or sp³). Based on the hybridization that
you assigned, what atomic orbitals does each N atom possess? (Hint: Each atom should
always have 4 atomic orbitals.)
NA atomic orbitals NB atomic orbitals Ne atomic orbitals ND atomic orbitals
e. Consider the lone pair on each nitrogen atoms. What atomic or hybridized atomic orbital
holds each lone pair (sp, sp², sp³, s, or p)?
Lone pair on N₁ held by Lone pair on NB held by Lone pair on ND held by
5. NO₂-¹
Total # of Valence Electrons:
Lewis Structure (show all resonance structures if applicable)
3-D Model Sketch
Any polar bonds in the molecule? Yes No
Molecular Shape:
Molecular Polarity:
Any polar bonds in the molecule? Yes No
6. H30*¹ — use a yellow ball for oxygen (not red) when constructing this model
Total # of Valence Electrons:
3-D Model Sketch
Lewis Structure (show all resonance structures if applicable)
Molecular Shape:
Molecular Polarity:
Polar
Polar
Bond Angles
Non-Polar
Bond Angles
Non-Polar
Chapter 1 Solutions
Organic Chemistry
Ch. 1.1 - Prob. 1.1PCh. 1.2 - Prob. 1.2PCh. 1.2 - Judging from their relative positions in the...Ch. 1.2 - Classify each bond as nonpolar covalent or polar...Ch. 1.2 - Using the symbols and +, indicate the direction...Ch. 1.2 - Draw Lewis structures showing all valence...Ch. 1.2 - Draw Lewis structures for these ions and show...Ch. 1.3 - Draw Lewis structures and condensed structural...Ch. 1.3 - Prob. 1.9PCh. 1.3 - Prob. 1.10P
Ch. 1.3 - Prob. 1.11PCh. 1.3 - Prob. 1.12PCh. 1.4 - Predict all bond angles for these molecules. (a)...Ch. 1.5 - The geometry of carbon in diamond is tetrahedral,...Ch. 1.5 - Because of their spherical shape, C60 molecules...Ch. 1.5 - What best describes the CCC bond angles in C60? 1....Ch. 1.5 - Prob. 1.14PCh. 1.7 - Describe the bonding in these molecules in terms...Ch. 1.8 - Prob. 1.16PCh. 1.8 - Prob. 1.17PCh. 1.8 - Prob. 1.18PCh. 1.9 - Draw three contributing structures of the...Ch. 1.9 - What is the hybridization state of the circled...Ch. 1.9 - The molecule shown on the right in the example in...Ch. 1.9 - Prob. CQCh. 1.9 - The following structure is called imidazolium....Ch. 1 - Write the ground-state electron configuration for...Ch. 1 - Identify the atom that has each ground-state...Ch. 1 - Define valence shell and valence electron.Ch. 1 - How many electrons are in the valence shell of...Ch. 1 - Prob. 1.24PCh. 1 - Prob. 1.25PCh. 1 - Prob. 1.26PCh. 1 - Write Lewis structures for these compounds. Show...Ch. 1 - Write Lewis structures for these ions. Show all...Ch. 1 - Prob. 1.29PCh. 1 - Some of these structural formulas are incorrect...Ch. 1 - Following the rule that each atom of carbon,...Ch. 1 - Following are several Lewis structures showing all...Ch. 1 - Which statements are true about electronegativity?...Ch. 1 - Why does fluorine, the element in the upper right...Ch. 1 - Arrange the single covalent bonds within each set...Ch. 1 - Using the values of electronegativity given in...Ch. 1 - Prob. 1.37PCh. 1 - Use VSEPR to predict bond angles about each...Ch. 1 - Use VSEPR to predict bond angles about each atom...Ch. 1 - Use VSEPR to predict the geometry of these ions....Ch. 1 - Prob. 1.41PCh. 1 - Prob. 1.42PCh. 1 - What is the meaning of the term tertiary (3) when...Ch. 1 - What is the meaning of the term tertiary (3) when...Ch. 1 - Draw structural formulas for (a) The four primary...Ch. 1 - Draw structural formulas for the three tertiary...Ch. 1 - Prob. 1.47PCh. 1 - Identify the functional groups in each compound.Ch. 1 - Draw a three-dimensional representation for each...Ch. 1 - Tetrafluoroethylene, C2F4, is the starting...Ch. 1 - Which statements are true about resonance...Ch. 1 - Prob. 1.52PCh. 1 - Prob. 1.53PCh. 1 - Prob. 1.54PCh. 1 - Are the structures in each set valid contributing...Ch. 1 - State the orbital hybridization of each...Ch. 1 - Describe each highlighted bond in terms of the...Ch. 1 - Following is a structural formula of the...Ch. 1 - Draw a Lewis structure for methyl isocyanate,...Ch. 1 - What is the hybridization of the highlighted atoms...Ch. 1 - Using cartoon representations, draw a molecular...Ch. 1 - In what kind of orbitals do the lone-pair...Ch. 1 - Draw the delocalized molecular orbitals for the...Ch. 1 - Prob. 1.64APCh. 1 - Each compound contains both ions and covalent...Ch. 1 - Predict whether the carbon-metal bond in these...Ch. 1 - Prob. 1.67APCh. 1 - Phosphorus is immediately under nitrogen in the...Ch. 1 - Draw a Lewis structure for the azide ion, N3. (The...Ch. 1 - Cyanic acid, HOCN, and isocyanic acid, HNCO,...Ch. 1 - In Chapter 6, we study a group of organic cations...Ch. 1 - Many reactions involve a change in hybridization...Ch. 1 - Following is a structural formula of benzene,...Ch. 1 - Following are three contributing structures for...Ch. 1 - (a) Draw a Lewis structure for the ozone molecule,...Ch. 1 - The following two compounds are isomers; that is,...Ch. 1 - In future chapters, we will encounter...Ch. 1 - Prob. 1.78AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Fill out the table below. You may want to start by drawing all of the Lewis dot structures first. Consult with your instructor, and then complete Lewis Structures Lab Report Form A. Lewis Dot Structures, Hybridization and Geometries Austin Community College Chemistry r out the table below. You may want to start by drawing all of the Lewis dot structures first. Consult with your instructor, and then complete the rest of the table. Add formal charges, as necessary. Name: compound Date: Lewis Dot Structure hybridization electronic CH4 geometry molecular re polar or non-polar? geometry or a sure nase pres us t CH2F2 H20 Tem NH3 pi 3.arrow_forwardA model for H,O is shown in the chem3D window. H2O has bent geometry. This geometry is sometimes called angular or V-shaped geometry. ball & stick +| labels - Rotate the molecule until you have a feeling for its three-dimensional shape. How many atoms are bonded to the central atom? Consider the bond angle at the central atom. What is the approximate numerical value of this angle? degrees. You could view this geometry as being derived from a trigonal planar molecule by removing one atom, or as being derived from a tetrahedral molecule by removing two atoms. Which does the bond angle suggest? For practice, type in the name of the geometry of the molecule:arrow_forwardLet's Try (Evaluation) Match each compound whose Lewis structural formula is given below as one of the following functional groups (fil in the number) 1. Alcohol Ether 2. Aldehyde 3. 4. Ketone 5 Amine 6. Carboxylic Acid 7. Ester 8. Amidearrow_forward
- SC ! F1 Q @ 2 F2 W #3 Which structure has a formal charge of -1 on the carbon shown with a "C"? Note that the atom of interest in each case includes any hydrogens or lone pairs that are present. =C- (b) (c) (d) O a.. O b.. O c.. O d.. A Moving to another question will save this response. >> 20 F9 F10 F3 F4 E @ $ 4 R % F5 T MacBook Air F6 6 Y & 7 F7 U * 00 8 DII FB ( 9 ) 0 I P a F11arrow_forward3. The following shows all resonance structures for the following molecule. a. Draw in all implied lone pairs. b. Draw in curved arrows that show the flow of electrons, making sure the arrows show the precise starting point and destination of the electrons. Label each arrow as: lp →→→л (p=lone pair) c. d. Rank the resonance structures from most stable to least based on the number of formal charges and atoms that lack an octet of electrons. ol-of-o. B A D d-d-d-o E C F Garrow_forward1 For the molecule CH2F2 a. Draw the lewis dot structure. b. Determine the AEN for C-H and C-F and state whether the bonds are polar or nonpolar c. Draw the wedgie 3-D line drawing for CH2F2 and the dipole moment for the molecule, if present.arrow_forward
- TASK 1: COMPLETE ME! Complete what is required at by the following table. Copy table in a separate sheet of раper. Polar Molecule or Nonpolar Molecule Bond Central AEN Type of Bond Atom 1. Sulfur difluoride SF2 2. Methane CH4 3. Охуgen O2 4. Ammonia NH3 5. Hydrogen sulfide H2Sarrow_forwardQuestion 48 48. Which of the following statements are true? 1. 11. III. IV. The electrons in each molecule tend to orient themselves around the most electronegative element. Each molecular drawing follows the localized electron model. Both HF and CO₂ are linear molecules and therefore polar. The bond angles of NH3 are slightly less than 109.5° because the lone pair compresses the angles between the bonding pairs. a. I, III, IV b. I, II, IV c. I, II, III d. II, IV e. All of the above statements are correct. C B esc E Q @ 2 W # 3 E $ 4 * R % 5 T < 6 Y & 7 8 Uarrow_forwardQuestion 16.b of 25 Classify and describe the properties of the following nitrogen containing compound. Provide a systematic name for this structure. N,N,N- 2- N- eth tri hex bi di CH3 prop N.N- CH3 pent but meth al amide an 1-arrow_forward
- 4. Write the Lewis dot (electron dot) symbol for each covalent molecule. Remember the number of unpaired electrons in the Lewis dot symbol of the atom determines the number of bonds each atom makes. If your molecule has unpaired electrons you are not done. If your molecule has the wrong number of bonds for some atoms it is wrong. Try a different arrangement of atoms. Working left to right will help you. Show loan pairs. a. HCN b. CH3COOH c. H₂CCONHCH3 d. HCCH e. C6H12arrow_forwardClassify each molecule as polar or nonpolar. Electrons on the outer atoms are omitted for clarity. Polar Nonpolar Answer Bank F F. F F. F F-Xe-F F Farrow_forwardFor the formula CH,CF2, *draw the Lewis structure in your work* (which I will grade when you upload it). After drawing the complete Lewis structure, type in the shape, bond angle, etc. for each blank. What is the molecular shape or geometry for the carbon on the left (the C in CH2)? What is the molecular shape or geometry for the carbon on the right (the C in CF2)?| What is the bond angle for the carbon on the left (the C in CH)? Type in a plain number, but include a "<" sign in front of the number if applicable. What is the bond angle for the carbon on the right (the C in CF2)? Type in a plain number, but include a "<" sign in front of the number if applicable. What is the hybridization for the carbon on the left (the C in CH2)? Type this in without any superscript, for instance sp3 would be typed as "sp3". What is the hybridization for the carbon on the right (the C in CF2)? Type this in without any superscript, for instance sp3 would be typed as "sp3". Is the molecule overall polar…arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning