
An ideal gas with γ = 1.67 starts at point A in Fig. 18.21, where its volume and pressure are 1.00 m3 and 250 kPa, respectively. It undergoes an adiabatic expansion that triples its volume, ending at B. It’s then heated at constant volume to C, and compressed isothermally back to A. Find (a) the pressure at B, (b) the pressure at C, and (c) the net work done on the gas.
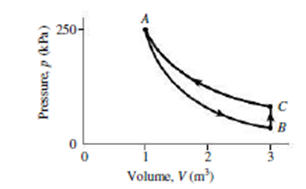
FIGURE 18.21 Problem 51
Want to see the full answer?
Check out a sample textbook solution
Chapter 18 Solutions
Essential University Physics (3rd Edition)
Additional Science Textbook Solutions
The Cosmic Perspective Fundamentals (2nd Edition)
College Physics: A Strategic Approach (3rd Edition)
Conceptual Physics (12th Edition)
Conceptual Physical Science (6th Edition)
Physics for Scientists and Engineers: A Strategic Approach with Modern Physics (4th Edition)
Lecture- Tutorials for Introductory Astronomy
- One mole of an ideal gas does 3 000 J of work on its surroundings as it expands isothermally to a final pressure of 1.00 atm and volume of 25.0 L. Determine (a) the initial volume and (b) the temperature of the gas.arrow_forwardFind the work done in the quasi-static processes shown below. The states are given as (p, V) values for the points in the PV plane: 1 (3 atm, 4 L), 2 (3 atm, 6 L), 3 (5 atm, 4 L), 4 (2 atm, 6 L), 5 (4 atm, 2 L), 6 (5 atm, 5 L) and 7 (2 atm, 5 L).arrow_forwardAt point A in a Carnot cycle, 2.34 mol of a monatomic ideal gas has a pressure of 1 4000 kPa, a volume of 10.0 L, and a temperature of 720 K. The gas expands isothermally to point B and then expands adiabatically to point C, where its volume is 24.0 L. An isothermal compression brings it to point D, where its volume is 15.0 L. An adiabatic process returns the gas to point A. (a) Determine all the unknown pressures, volumes, and temperatures as you f ill in the following table: (b) Find the energy added by heat, the work done by the engine, and the change in internal energy for each of the steps A B, B C, C D, and D A (c) Calculate the efficiency Wnet/|Qk|. (d) Show that the efficiency is equal to 1 - TC/TA, the Carnot efficiency.arrow_forward
- A car tile contains 0.0380 m3 of air at a pressure of 2.20105 Pa (about 32 psi). How much more internal energy does this gas have than the same volume has at zero gauge pressure (which is equivalent to normal atmospheric pressure)?arrow_forwardConsider these scenarios and state whether work is done by the system on the environment (SE) or by the environment on the system (ES): (a) opening a carbonated beverage; (b) filling a flat tire; (c) a sealed empty gas can expands on a hot day, bowing out the walls.arrow_forwardAn ideal gas initially at 300 K undergoes an isobaric expansion at 2.50 kPa. If the volume increases from 1.00 m3 to 3.00 m3 and 12.5 kJ is transferred to the gas by heat, what are (a) the change in its internal energy and (b) its final temperature?arrow_forward
- Five moles of monatomic ideal gas have initial pressure 2.50 × 10³ Pa and initial volume 2.10 m³. While undergoing an adiabatic expansion, the gas does 1480 J of work. What is the final pressure of the gas after the expansion?arrow_forwardConsider a 1 mole of an ideal gas at 25.00 oC. This gas is allowed to expand reversibly and isothermally from a volume of 7.95 liters to 117.61 liters. Calculate the work done by the gas in joules. Answer in 6 significant figures.arrow_forwardA chemical reaction transfers 6250 J of thermal energy into 12.0 moles of an ideal gas while the system expands by 2.00 x 10-2 m3 at a constant pressure of 1.25 x 10o5 Pa. HINT (a) To find the change in internal energy, first calculate the work done on the gas and then apply the first law of thermodynamics. (b) For an ideal gas at constant pressure, PAV = NRAT. Click the hint button again to remove this hint. (a) Find the change in the internal energy (in J). (b) Calculate the change in temperature of the gas (in K).arrow_forward
 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
 Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning



