
FUNDAMENTALS OF PHYSICS V.1
11th Edition
ISBN: 9781119573913
Author: Halliday
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 18, Problem 2Q
Figure 18-24 shows three linear temperature scales, with the freezing and boiling points of water indicated. Rank the three scales according to the size of one degree on them, greatest first.
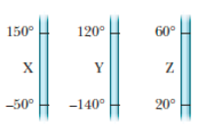
Figure 18-24 Question 2.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
A steel beam is used in the construction of a skyscraper. By what fraction ΔL/L0 does the length of the beam increase when the temperature changes from that on a cold winter day 4.2°C to that on a summer day 25.7 °C? Assume the linear expansion coefficient is 12x10-6 (C)-1.
o42 0 A 20.0 g copper ring at
0.000°C has an inner diameter of
D = 2.54000 cm. An aluminum
sphere at 100.0°C has a diameter
of d = 2.545 08 cm. The sphere is
put on top of the ring (Fig. 18-36),
and the two are allowed to come
to thermal equilibrium, with no
heat lost to the surroundings. The
sphere just passes through the
ring at the equilibrium tempera-
ture. What is the mass of the
Al
Cu
Figure 18-36 Problem 42.
sphere?
(a) Define a new temperature scale, say °N in which the boiling and freezing points of water are 1000°N and 100°N, respectively, and correlate this scale with the Fahrenheit and Celsius scales. (b) The °N reading on this scale is a certain number of t degrees on a corresponding absolute temperature scale. What is this absolute temperature at 0°N?
Ans. (a)lN = 9te + 100. (b) 2360°N abs, approx.
Chapter 18 Solutions
FUNDAMENTALS OF PHYSICS V.1
Ch. 18 - The initial length L, change in temperature T, and...Ch. 18 - Figure 18-24 shows three linear temperature...Ch. 18 - Materials A, B, and C are solids that are at their...Ch. 18 - A sample A of liquid water and a sample B of ice,...Ch. 18 - Question 4 continued: Graphs b through f of Fig....Ch. 18 - Figure 18-26 shows three different arrangements of...Ch. 18 - Figure 18-27 shows two closed cycles on p-V...Ch. 18 - For which cycle in Fig. 18-27, traversed...Ch. 18 - Three different materials of identical mass are...Ch. 18 - A solid cube of edge length r, a solid sphere of...
Ch. 18 - A hot object is dropped into a thermally insulated...Ch. 18 - Suppose the temperature of a gas is 373.15 K when...Ch. 18 - Two constant-volume gas thermometers are...Ch. 18 - A gas thermometer is constructed of two...Ch. 18 - a In 1964, the temperature in the Siberian village...Ch. 18 - At what temperature is the Fahrenheit scale...Ch. 18 - On a linear X temperature scale, water freezes at...Ch. 18 - ILW Suppose that on a linear temperature scale X,...Ch. 18 - At 20C, a brass cube has edge length 30 cm. What...Ch. 18 - ILW A circular hole in an aluminum plate is 2.725...Ch. 18 - An aluminum flagpole is 33 m high. By how much...Ch. 18 - Prob. 11PCh. 18 - An aluminum-alloy rod has a length of 10.000 cm at...Ch. 18 - SSM Find the change in volume of an aluminum...Ch. 18 - When the temperature of a copper coin is raised by...Ch. 18 - ILW A steel rod is 3.000 cm in diameter at 25.00C....Ch. 18 - When the temperature of a metal cylinder is raised...Ch. 18 - SSM WWW An aluminum cup of 100 cm3 capacity is...Ch. 18 - At 20C, a rod is exactly 20.05 cm long on a steel...Ch. 18 - GO A vertical glass tube of length L = 1.280 000 m...Ch. 18 - GO In a certain experiment, a small radioactive...Ch. 18 - SSM ILW As a result of a temperature rise of 32 C,...Ch. 18 - One way to keep the contents of a garage from...Ch. 18 - SSM A small electric immersion healer is used to...Ch. 18 - A certain substance has a mass per mole of 50.0...Ch. 18 - Prob. 25PCh. 18 - What muss of butter, which has a usable energy...Ch. 18 - SSM Calculate the minimum amount of energy, in...Ch. 18 - How much water remains unfrozen after 50.2 kJ is...Ch. 18 - In a solar water heater, energy from the Sun is...Ch. 18 - A 0.400 kg simple is placed in a cooling apparatus...Ch. 18 - ILW What mass of steam at 100C must be mixed with...Ch. 18 - The specific heat of a substance varies with...Ch. 18 - Nonmetric version: a How long does a 2.0 105...Ch. 18 - GO Samples A and B are at different initial...Ch. 18 - An insulated Thermos contains l30 cm3 of hot...Ch. 18 - A 150 g copper bowl contains 220 g of water, both...Ch. 18 - A person makes a quantity of iced tea by mixing...Ch. 18 - A 0.530 kg sample of liquid water and a sample of...Ch. 18 - GO Ethyl alcohol has a boiling point of 78.0C, a...Ch. 18 - GO Calculate the specific heat of a metal from the...Ch. 18 - SSM WWW a Two 50 g ice cubes are dropped into 200...Ch. 18 - GO A 20.0 g copper ring at 0.000C has an inner...Ch. 18 - In Fig. 18-37, a gas sample expands from V0 to...Ch. 18 - GO A thermodynamic system is taken from stale A to...Ch. 18 - SSM ILW A gas within a closed chamber undergoes...Ch. 18 - Suppose 200 J of work is done on a system and 70.0...Ch. 18 - Prob. 47PCh. 18 - GO As a gas is held within a closed chamber, it...Ch. 18 - GO Figure 18-42 represents a closed cycle for a...Ch. 18 - GO A lab sample of gas is taken through cycle abca...Ch. 18 - A sphere of radius 0.500 m, temperature 27.0C, and...Ch. 18 - The ceiling of a single-family dwelling in a cold...Ch. 18 - SSM Consider the slab shown in Fig. 18-18. Suppose...Ch. 18 - If you were to walk briefly in space without a...Ch. 18 - ILW A cylindrical copper rod of length 1.2 m and...Ch. 18 - The giant hornet Vespa mandarinia japonica preys...Ch. 18 - Prob. 57PCh. 18 - A solid cylinder of radius r1 = 2.5 cm, length h1...Ch. 18 - Prob. 59PCh. 18 - GO Figure 18-46 shows the cross section of a wall...Ch. 18 - SSM A 5.0 cm slap has formed on an outdoor tank of...Ch. 18 - Leidenfrost effect. A water drop will last about 1...Ch. 18 - GO Figure 18-49 shows in cross section a wall...Ch. 18 - Prob. 64PCh. 18 - Ice has formed on a shallow pond, and a shady...Ch. 18 - GO Evaporative cooling of beverages. A cold...Ch. 18 - In the extrusion of cold chocolate from a tube,...Ch. 18 - Prob. 68PCh. 18 - Figure 18-51 displays a closed cycle for a gas....Ch. 18 - In a certain solar house, energy from the Sun is...Ch. 18 - A 0.300 kg sample is placed in a cooling apparatus...Ch. 18 - The average rate at which energy is conducted...Ch. 18 - What is the volume increase of an aluminum cube...Ch. 18 - In a series of experiment, block B is to be placed...Ch. 18 - Figure 18-54 displays a dosed cycle for a gas....Ch. 18 - Three equal-length straight rods, of aluminum,...Ch. 18 - SSM The temperature of a 0.700 kg cube of ice is...Ch. 18 - GO Icicles. Liquid water coats an active growing...Ch. 18 - SSM A sample of gas expands from an initial...Ch. 18 - Figure 18-56a shows a cylinder containing gas and...Ch. 18 - SSM A sample of gas undergoes a transition from an...Ch. 18 - Prob. 82PCh. 18 - SSM The temperature of a Pyrex disk is changed...Ch. 18 - a Calculate the rate at which body heat is...Ch. 18 - SSM A 2.50 kg Jump of aluminum is heated to 92.0C...Ch. 18 - A glass window pane is exactly 20 cm by 30 cm at...Ch. 18 - A recruit can join the semi-secret 300 F club at...Ch. 18 - A steel rod at 25.0C is bolted at both ends and...Ch. 18 - An athlete needs to lose weight and decides to do...Ch. 18 - Soon after Earth was formed, heat released by the...Ch. 18 - Prob. 91PCh. 18 - A rectangular plate of glass initially has the...Ch. 18 - Suppose that you intercept 5.0 103 of the energy...Ch. 18 - A thermometer of mass 0.0550 kg and of specific...Ch. 18 - A sample of gas expands from V1 = 1.0 m3 and p1 =...Ch. 18 - Figure 18-59 shows a composite bar of length L =...Ch. 18 - On finding your stove out of order, you decide to...Ch. 18 - The p-V diagram in the Fig. 18-60 shows two paths...Ch. 18 - A cube of edge length 6.0 106 m, emissivity 0.75,...Ch. 18 - A flow calorimeter is a device used to measure the...Ch. 18 - An object of mass 6.00 kg falls through a height...Ch. 18 - The Pyrex glass mirror in a telescope has a...Ch. 18 - The area A of a rectangular plate is ab = 1.4 m2....Ch. 18 - Consider the liquid in a barometer whose...Ch. 18 - A pendulum clock with a pendulum made of brass is...Ch. 18 - Prob. 106PCh. 18 - Prob. 107PCh. 18 - A 1700 kg Buick moving at 83 km/h brakes to a...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Use the data at the back of this book to determine H for the combustion of a mole of glucose, C6H12O6+6O26CO2+6...
An Introduction to Thermal Physics
60. One billiard ball is shot east at 2.00 m/s. A second, identical billiard ball is shot west at 1.00 m/s. The...
College Physics: A Strategic Approach (4th Edition)
A spaceship (A) is moving at speed c/2 with to another spaceship (B). Observers in A and B set their clocks so ...
University Physics Volume 3
Youre driving at speed v0 when you spot a stationary moose on the road, a distance d ahead. Find an expression ...
Essential University Physics (3rd Edition)
Evaluate the following integrals:
(a)
(b)
(c)
(d)
Introduction to Electrodynamics
13.6 Find the magnitude and direction of the net gravitational force on mass A due to masses B and C in Fig. E1...
University Physics with Modern Physics (14th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- At 25.0 m below the surface of the sea, where the temperature is 5.00C, a diver exhales an air bubble having a volume of 1.00 cm3. If the surface temperature of the sea is 20.0C, what is the volume of the bubble just before it breaks the surface?arrow_forwardTwo concrete spans that form a bridge of length L are placed end to end so that no room is allowed for expansion (Fig. P16.63a). If a temperature increase of T occurs, what is the height y to which the spans rise when they buckle (Fig. P16.63b)?arrow_forwardWhen builders were constructing a sidewalk they forgot to include an expansion joint between two of the segments, L = 2.5 m at To = 20° C. Assume the opposite ends of each segment are fixed and the linear expansion coefficient is α = 11.9 × 10-6 °C-1. a) As the day heats to Tb the segments press against each other and begin to raise the junction a distance h forming a triangle. What is the height (in meters) at Tb=110° F? b) What should the gap have been to prevent them from touching in meters?arrow_forward
- *59 In Fig. 18-45a, two identical rec- tangular rods of metal are welded end Ti to end, with a temperature of T1 = 0°C on the left side and a temperature of T = 100°C on the right side. In 2.0 min, 10 J is conducted at a constant rate from the right side to the left side. How much time would be required to conduct 10 J if the rods were welded side to side as in Fig. 18-45b? T2 (a) т. (6) Figure 18-45 Problem 59.arrow_forward> An interval of 1 Celcius or Kelvin degree therefore is equal to 1 Fahrenheit or Rankine degrees, leading to the conversion factc Example: Consider the interval from 20 °F to 80 °F. 1. 1.8° F/1°C = 1, 1.8° R/1K = 1, 1°F/1°R = 1, 1°C/1K = 1 2. Calculate the equivalent temperatures in °C and the interval between them. Calculate directly the interval in °C.arrow_forwardA brass (a=19 x 10-6 °c- 1 ) ring of diameter 10.01 cm at 20.0°C is heated and slipped over an aluminum rod (a = 24 x 10-60c-1 ) of diameter 10.4 cm at 20.0°C. Assuming the average coefficients of linear expansion are constant, to what temperature (in c-1) must this combination be cooled to separate them?arrow_forward
- If two thermometers, one reading in oC and the other on K, are inserted in the same system, under the circumstances will they both have the same numerical reading? What will be the system’s temperature when the absolute thermometer reads twice the numerical reading of the Celsius thermometer?arrow_forwardFind the equilibrium temperature (T subscript f) of the systems in degrees Celsius.arrow_forwardAt temperatures of a few hundred kelvins the specificheat capacity of copper approximately follows theempirical formula c = α + βT + δT −2, whereα = 349 J/kg · K, β = 0.107 J/kg · K2, andδ = 4.58 × 105J · kg · K. How much heat is needed toraise the temperature of a 2.00-kg piece of copper from20 °C to 250 °C ?arrow_forward
- Approximately how long should it take 9.5 kg of ice at 0 °C to melt when it is placed in a carefully sealed Styrofoam ice chest of dimensions 25 cm × 35 cm x 55 cm whose walls are 1.1 cm thick? Assume that the conductivity of Styrofoam is double that of air and that the outside temperature is 35 °C. Express your answer using two significant figures. VE ΑΣΦ ? Sarrow_forwardIn a house the temperature at the surface of a window is 25 °C. The temperature outside at the window surface is 5.0 °C. Heat is lost through the window via conduction, and the heat lost per second *16. has a certain value. The temperature outside begins to fall, while the con- ditions inside the house remain the same. As a result, the heat lost per second increases. What is the temperature at the outside window surface when the heat lost per second doubles?arrow_forward(a) The number of kilocalories in food is determined by calorimetry techniques in which the food is burned and the amount of heat transfer is measured. How many kilocalories per gram are there in a 4.60 g peanut if the energy from burning it is transferred to 0.600 kg of water held in a 0.200 kg aluminum cup, causing a 43.6°C temperature increase? kcal/garrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning


Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning
Thermodynamics: Crash Course Physics #23; Author: Crash Course;https://www.youtube.com/watch?v=4i1MUWJoI0U;License: Standard YouTube License, CC-BY