
Organic Chemistry
7th Edition
ISBN: 9780321803221
Author: Paula Y. Bruice
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17, Problem 67P
How many signals would the product of the following reaction show in
- a. its 1H NMR spectrum?
- b. its 13C NMR spectrum?
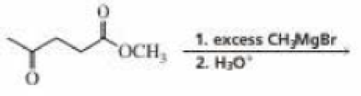
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
5.
What would be different in the 1H NMR spectra of a mono and disubstituted azulene?How many signals would you expect in the 13C NMR of a mono and disubstituted azulene?
How can you distinguish the following compounds using:a. their infrared spectra? b. their 1H NMR spectra?
1. Why does H2 not give an IR spectrum?
2. Explain why primary amines and unsubstituted amides have two NH stretching absorptions.
3. Why do anhydrides show two carbonyl peaks?
4. HCl is known to give addition reactions to carbon-carbon double bonds. Why is this behavior not observed in this reaction?
5. Predict the structure of the product expected from addition of molecular bromine to maleic acid.
Chapter 17 Solutions
Organic Chemistry
Ch. 17.1 - Prob. 1PCh. 17.1 - Give two names for each of the following:Ch. 17.1 - Name the following:Ch. 17.2 - Prob. 4PCh. 17.4 - What products are formed when the following...Ch. 17.4 - We saw on the previous page that...Ch. 17.4 - a. How many stereoisomers are obtained from the...Ch. 17.4 - Prob. 9PCh. 17.4 - Write the mechanism for the reaction of acetyl...Ch. 17.4 - Prob. 11P
Ch. 17.4 - Show how the following compounds can be...Ch. 17.5 - Prob. 13PCh. 17.5 - Prob. 14PCh. 17.6 - In the mechanism for cyanohydrin formation, why is...Ch. 17.6 - Prob. 16PCh. 17.6 - Prob. 17PCh. 17.6 - Show two ways to convert an alkyl halide into a...Ch. 17.7 - Prob. 20PCh. 17.7 - Prob. 21PCh. 17.7 - Prob. 22PCh. 17.7 - Prob. 23PCh. 17.8 - Prob. 24PCh. 17.9 - What reducing agents should be used to obtain the...Ch. 17.9 - Prob. 26PCh. 17.9 - Prob. 27PCh. 17.10 - Prob. 28PCh. 17.10 - Prob. 29PCh. 17.10 - Prob. 30PCh. 17.10 - The pKa of protonated acetone is about 7.5. and...Ch. 17.10 - Prob. 32PCh. 17.10 - Prob. 33PCh. 17.10 - Prob. 34PCh. 17.10 - Excess ammonia must be used when a primary amine...Ch. 17.10 - The compounds commonly known as amino acids are...Ch. 17.11 - Hydration of an aldehyde is also catalyzed by...Ch. 17.11 - Which ketone forms the most hydrate in an aqueous...Ch. 17.11 - When trichloroacetaldehyde is dissolved in water,...Ch. 17.12 - Which of the following are a. hermiacetals? b....Ch. 17.12 - Prob. 41PCh. 17.12 - Explain why an acetal can be isolated but most...Ch. 17.13 - Prob. 43PCh. 17.13 - Prob. 44PCh. 17.13 - What products would be formed from the proceedings...Ch. 17.13 - a. In a six-step synthesis, what is the yield of...Ch. 17.13 - Show how each of the following compounds could be...Ch. 17.15 - Prob. 48PCh. 17.17 - Prob. 50PCh. 17.18 - Prob. 51PCh. 17.19 - Prob. 52PCh. 17 - Draw the structure for each of the following: a....Ch. 17 - Prob. 54PCh. 17 - Prob. 55PCh. 17 - a. Show the reagents required to form the primary...Ch. 17 - Prob. 57PCh. 17 - Using cyclohexanone as the starting material,...Ch. 17 - Prob. 59PCh. 17 - 60. Show how each of the following compounds could...Ch. 17 - Fill in the boxes:Ch. 17 - Prob. 62PCh. 17 - Identify A through O:Ch. 17 - Prob. 64PCh. 17 - Prob. 65PCh. 17 - Prob. 66PCh. 17 - How many signals would the product of the...Ch. 17 - Prob. 68PCh. 17 - Prob. 69PCh. 17 - Prob. 70PCh. 17 - Prob. 71PCh. 17 - Prob. 72PCh. 17 - Prob. 73PCh. 17 - Prob. 74PCh. 17 - Prob. 75PCh. 17 - Prob. 76PCh. 17 - Prob. 77PCh. 17 - A compound gives the following IR spectrum. Upon...Ch. 17 - a. Propose a mechanism for the following reaction:...Ch. 17 - Prob. 80PCh. 17 - Prob. 81PCh. 17 - Prob. 82PCh. 17 - Prob. 83PCh. 17 - Prob. 84PCh. 17 - In the presence of an acid catalyst, acetaldehyde...Ch. 17 - Prob. 86PCh. 17 - Prob. 87PCh. 17 - Prob. 88PCh. 17 - A compound reacts with methylmagnesium bromide...Ch. 17 - Show how each of the following compounds can be...Ch. 17 - Prob. 91PCh. 17 - Prob. 92PCh. 17 - The pKa values of the carboxylic acid groups of...Ch. 17 - The Baylis-Hillman reaction is a DABCO...Ch. 17 - Prob. 95PCh. 17 - Prob. 96P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A B. C. H. CH Br HC-C-CH CHy CH3-C-CH H-C-CH,CH, CH CH, Which of the following compounds give four major peaks in their 1H NMR spectra? OA and B. Band C. A and C. O All of themarrow_forwardUsing proton NMR spectra, how could a chemist distinguish between the following two compounds? H. H. СОСН Ph COCH; Ph H. Compound A Compound B The alkene protons in compound A display a larger coupling constant. Compound A and compound B show a different number of signals in the proton spectrum. The alpha alkene proton would be more shifted than the beta alkene proton in compound B Proton NMR spectroscopy cannot be used to distinguish between these compounds. The alkene protons in compound B display a larger coupling constant.arrow_forward5. How many signals would you expect each of the following molecules to have in its 'H NMR spectrum? Label the structure (a, b, etc...) to correlate protons with the signals in the ¹H NMR spectrums. In which regions of the spectrum would you expect to see each signal? How many hydrogen atoms will each signal integrate for? H or OMe HO Vanillin 4-hydroxy-3-methoxybenzaldehyde HO OH OMe Vanillyl Alcohol 4-hydroxy-3-methoxybenzyl alcoholarrow_forward
- Construct a structure of the given 1H NMR data signals.A. C5H12O 0.91 δ (3H, triplet) 1.19 δ (6H, singlet) 1.50 δ (2H, quartet) 2.24 δ (1H, singlet) B. C4H10O 0.90 δ (6H, doublet) 1.76 δ (1H, multiplet) 3.38 δ (2H, doublet) 3.92 δ (1H, singlet) C. C5H10O 1.09 δ (6H, doublet) 2.12 δ (3H, singlet) 2.58 δ (1H, septet)arrow_forward3. Nuclear Magnetic Resonance Spectroscopy a. Draw the 'H NMR spectrum for the following compound. CIarrow_forward2. The following synthesis was planned. The 'H NMR spectrum of the final product was run and is presented below. Was the synthesis successful? If not, what is the actual product and why? sos H2N' 10 로 1H 2H 2H 8 1. NaNO2 HCI (aq.) 2H 2H 6 2. H 1H 1H PPM 1H OH 1H HO 2arrow_forward
- QUESTION 3 Briefly explain how you might distinguish between the following substances by comparing their 1H NMR spectra. a. and Compound I Compound II b. Propanal acetonearrow_forward3.How many sets of equivalent protons are there in hexane? a. 2 b. 3 c. 6 d. 7 4. What is the splitting of the signal in the 'H NMR spectrum for the methyl protons of 1-bromo- 2-methylpropane? a. singlet b. doublet c. triplet d. nonet 5. Which C,H,Br compound gives a doublet at approximately 3.3 ppm in the 'H NMR spectrum? 1 b. 2 a. ⠀ c. 3 d. 4 Br H₂C-O-CH₂-CH₂-C-H a. i b. ii c. iii d. iv 2 iv Br 6. Which of the protons in the following molecule appear furthest downfield in the 'H NMR spectrum? Br Brarrow_forward1. How many proton signals would be seen in the 'H NMR for the following compounds? 6pt Br Br а. d. f. #3 aiarrow_forward
- Which compound gives the following 1H NMR spectrum? A. aldehyde B. allyl ether C. ketone D. anisolearrow_forwardWhat product did you obtain from the competitive nitration of methyl benzoate? a. Which particular IR absorptions led you to this conclusion? b. How can the NMR signals in the aromatic region be used to confirm the identity of your product?arrow_forwardd. Which compound would be expected to show the following NMR spectrum? 6.0 4.0 2.0 4.8 4.6 4.4 14 1 30 4.2 40 16 28 26 24 22 20 16 14 12 1 ppm one a. b. Br с. HO.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY