
Organic Chemistry
11th Edition
ISBN: 9781118133576
Author: T. W. Graham Solomons, Craig Fryhle
Publisher: Wiley, John & Sons, Incorporated
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16, Problem 5Q
An industrial synthesis of benzaldehyde makes use of toluene and molecular chlorine as starting materials to produce 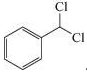 This compound is then converted to benzaldehyde. Suggest what steps are involved in the process.
This compound is then converted to benzaldehyde. Suggest what steps are involved in the process.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Please correct answer and don't used hand raiting don't used Ai solution
Please correct answer and don't used hand raiting
Please correct answer and don't used hand raiting
Chapter 16 Solutions
Organic Chemistry
Ch. 16 - PRACTICE PROBLEM 16.1 (a) Give IUPAC substitutive...Ch. 16 - Prob. 2PPCh. 16 - Prob. 3PPCh. 16 - Practice Problem 16.4
Provide the reagents and...Ch. 16 - Prob. 5PPCh. 16 - Prob. 6PPCh. 16 - Prob. 7PPCh. 16 - Prob. 8PPCh. 16 - Prob. 9PPCh. 16 - Prob. 10PP
Ch. 16 - Prob. 11PPCh. 16 - Practice Problem 16.12
What product would be...Ch. 16 - Prob. 13PPCh. 16 - Practice Problem 16.14
Dihydropyran reacts readily...Ch. 16 - Practice Problem 16.15 Show how you might use...Ch. 16 - Practice Problem 16.16 (a) Show how you might...Ch. 16 - Practice Problem 16.17
In addition to...Ch. 16 - Practice Problem 16.18
Triphenylphosphine can be...Ch. 16 - Prob. 19PPCh. 16 - PRACTICE PROBLEM 16.20
Give the structure of the...Ch. 16 - PRACTICE PROBLEM 16.21 What would be the major...Ch. 16 - Prob. 22PCh. 16 - 16.23 Write structural formulas for the products...Ch. 16 - Give structural formulas for the products formed...Ch. 16 - 16.25 What products would be obtained when...Ch. 16 - Predict the major organic product from each of the...Ch. 16 - 16.27 Predict the major product from each of the...Ch. 16 - 16.28 Predict the major product from each of the...Ch. 16 - Prob. 29PCh. 16 - 16.30 Write detailed mechanisms for each of the...Ch. 16 - Prob. 31PCh. 16 - Prob. 32PCh. 16 - Show how you would convert benzaldehyde into each...Ch. 16 - 16.34 Show how ethyl phenyl ketone could be...Ch. 16 - Show how benzaldehyde could be synthesized from...Ch. 16 - Give structures for compounds AE. Cyclohexanol...Ch. 16 - Prob. 37PCh. 16 - Prob. 38PCh. 16 - Prob. 39PCh. 16 - Prob. 40PCh. 16 - Prob. 41PCh. 16 - Prob. 42PCh. 16 - 16.43 The structure of the sex pheromone...Ch. 16 - Provide reagents that would accomplish each of the...Ch. 16 - Write a detailed mechanism for the following...Ch. 16 - Prob. 46PCh. 16 - Dutch elm disease is caused by a fungus...Ch. 16 - Prob. 48PCh. 16 - Compounds W and X are isomers; they have the...Ch. 16 - Compounds Y and Z are isomers with the molecular...Ch. 16 - Compound A (C9H18O) forms a phenylhydrazone, but...Ch. 16 - Compound B (C8H12O2) shows a strong carbonyl...Ch. 16 - (a) What would be the frequencies of the two...Ch. 16 - Prob. 54PCh. 16 - Prob. 1LGPCh. 16 - Which Wittig reagent could be used to synthesize ?...Ch. 16 - 16.2 Which compound is an acetal?
(d)More than...Ch. 16 - Which reaction sequence could be used to convert...Ch. 16 - 16.4 Complete the following syntheses. If more...Ch. 16 - 16.5 An industrial synthesis of benzaldehyde makes...Ch. 16 - Prob. 6Q
Additional Science Textbook Solutions
Find more solutions based on key concepts
During exponential growth, a population always (A) has a constant per capita population growth rate. (B) quickl...
Campbell Biology (11th Edition)
Match the following examples of mutagens. Column A Column B ___a. A mutagen that is incorporated into DNA in pl...
Microbiology: An Introduction
2. List the subdivisions of the thoracic and abdominopelvic cavities.
Human Anatomy & Physiology (2nd Edition)
83. The combustion of gasoline produces carbon dioxide and water. Assume gasoline to be pure octane (C8H18) and...
Introductory Chemistry (6th Edition)
PRACTICE 1.3 The melting point of table salt is 1474oF. What temperature is this on the Celsius and Kelvin scal...
Chemistry (7th Edition)
How are ribs classified?
Principles of Anatomy and Physiology
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- this is an organic chemistry question please answer accordindly!! please post the solution draw the figures on a paper please hand drawn and post, please answer EACH part till the end and dont just provide wordy explanations, please draw them on a paper and post clearly!! answer the full question with all details EACH PART CLEARLY please thanks!! im reposting this please solve all parts and draw it not just word explanations!!arrow_forwardA mixture of 0.412 M C12, 0.544 M F2, and 0.843 M CIF is enclosed in a vessel and heated to 2500 K. C12(g) + F2(g )2CIF(g) Kc = 20.0 at 2500 K Calculate the equilibrium concentration of each gas at 2500 K. [C12] = M [F2] = M [ CIF] =arrow_forwardShow reaction mechanism with explanation. don't give Ai generated solutionarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License