
Concept explainers
Interpretation:
The new nucleus after the emission of a positron needs to be determined.
Concept introduction:
A proton is turned into a neutron and a positron (with a positive charge) leaves the nucleus.
Positron emission happens when a nucleus emits a positron
Answer to Problem 51UTC
Solution:
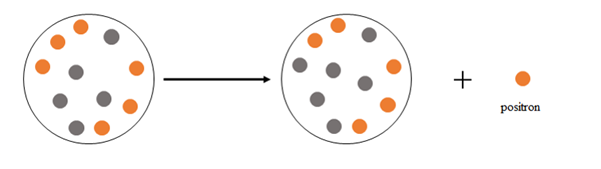
Explanation of Solution
Positron emission happens when a nucleus emits a positron
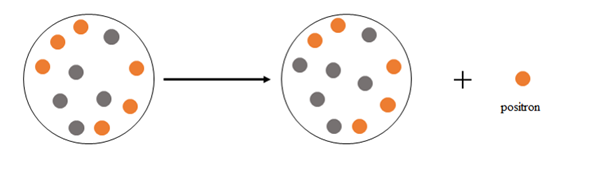
At the beginning there are 6 protons and 5 neutrons, after the emission there are 5 protons and 6 neutrons.
Thus, positron emission happens when a nucleus emits a positron
Want to see more full solutions like this?
Chapter 16 Solutions
Pearson eText Basic Chemistry -- Instant Access (Pearson+)
- The table below is a summary of different modes of nuclear decay. Fill in the changes in atomic number (Z), number of neutrons (N), and mass number (A) in each case. Use “+” sign for increase, “–“ sign for decrease, and “0” for no change. Provide the symbol for each elementary particle involved in the decay process. (12 points) Decay Mode Particle Symbol Change in: Change in: Change in: Z N A Alpha decay 4/2α Beta decay 0/-1 β Positron decay 0/+1 β Electron capture 0/-1 earrow_forwardPart 2: Balancing Nuclear Reactions (5.2) 238 92U. 23Th + He 90 1. Identify the reactants and products in the above reaction. 2. For each element, what do the top (superscripted) and the bottom (subscripted) numbers represent respectively? 3. How many protons and neutrons are found in the uranium isotope used above? 4. How many protons and neutrons are found in the thorium isotope used above? 5. What is the difference in, a. the number of protons between U and Th? b. the number of neutrons between U and Th? c. the mass number between U and Th? 6. How do you explain these differences with respect to the nuclear reaction above?arrow_forward10.2. Write the equations for the following examples of nuclear decay: a) α emission by Bi 83 193 c) ß+ by 34Se 68 b) ß− by 12Mg 27 d) electron capture by 32Ge 71arrow_forward
- Part 1: Writing Radioactive Decay Reactions (5.2) lodine-131 (131I) isotope undergoes beta decay. 1. Write the symbol for the beta particle released by iodine-131 (show the respective superscript and subscript values). 2. In the nuclear equation for beta decay of iodine-131, does the beta particle appear on the reactant or product side of the arrow? Why?arrow_forwardMacmillan Learning Iridium-192 is one radioisotope used in brachytherapy, in which a radioactive source is placed inside a patient's body to treat cancer. Brachytherapy allows the use of a higher than normal dose to be placed near the tumor while lowering the risk of damage to healthy tissue. Iridium-192 is often used in the head or breast. Use the radioactive decay curve of iridium-192 to answer the three questions. Sample remaining (9) 100 90 70 50- 10 20- 0 time: 10 20 30 10 mass remaining: half-life: 3 70 If the initial sample is 8.00 g. what mass of the original iridium-192 remains after 55 days? Estimate the half-life of the radioisotope. 80 90 100 110 120 130 140 150 160 170 180 190 Time Idays) How many days would it take for two-thirds of the sample to decay? B days daysarrow_forwarda 10.5. Sodium-24 (t½= 15h) is used to study blood circulation. If a patient is injected with aqueous solution of 2ªNaCl whose activity is 2.5 x 10° d/s, how much of the activity present in the patient's body and excreted fluids after 4.0 days? le of 35S a B- emitter, has an 30tiarrow_forward
- 6.7 A neutral K meson decays in flight via KO n*x. If the negative pion is produced at rest, calculate the kinetic energy of the positive pion. [Mass of K° is 498 MeV/c2; that of n is 140 MeV/c?]arrow_forward3.3. Write the nuclear equation for the nuclear reaction in which a nuclei undergoes ß- decay and changes to cesium-133.3.5. Sodium-24 (t1/2= 15h) is used to study blood circulation. If a patient is injected with an aqueous solution of 24NaCl whose activity is 2.5x109d/s, how much of the activity is present in the patient’s body and excreted fluids after 4.0 days?arrow_forwardSW Science 10 Unit 5 Radioactive Decay Worksheet Name:asmin laward th Student #: 5.2.1 Radioactive Decay - Half-Life 1. Complete the following table by shading in the appropriate number of squares or calculating the amount of parent material left or decay product formed. Amount % Decay Product FORMED % Parent Radioactive Mass (g) Fraction of Parent Material Material Lefe Material LEFT original sample 100g 100% 0% after 1 half-life 50g 50% after 2 half-lives 25g 12.5% after 3 half-lives 93.75% after 4 half-lives 2. Use the graph on the following page to plot the results of the table in question #1. NOTE: All Radioactive Decay graphs will have a similar shape.arrow_forward
- 7.2.5 moles of Ni-56 initially exist. Find the number of moles of Co-56 and Fe-56 after 20 days. (Required nuclide information should be retrieved from https://atom.kaeri.re.kr/nuchart/?zlv=0.)arrow_forwardWatch help video Element X decays radioactively with a half life of 8 minutes. If there are 530 grams of Element X, how long, to the nearest tenth ofa minute, would it take the element to decay to 7 grams? y = a(.5)%arrow_forward(a) Chlorine has two common isotopes, one of nucleon number 35 and one of nucleon number 37. The proton number is 17. Complete the table: nucleon number 35 nucleon number 37 number of protons number of neutrons number of electrons (b) Some isotopes are radioactive. State two practical uses of radioisotopes. For each example outline how it is used. 1. 2.arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

