
Concept explainers
A
Figure 16.6. What is the structure of this diol?
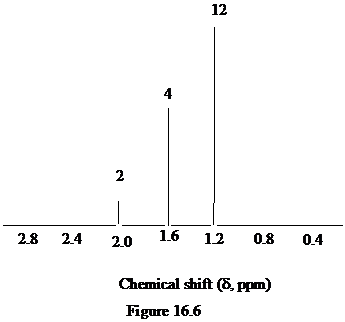
Interpretation:
The structure for the diol having molecular formula
Concept introduction:
The vicinal diols on treatment with periodic acid
The oxygen atoms do not show any effect on the index of hydrogen deficiency.
The index of hydrogen deficiency (IHD) represents the presence of number of multiple bonds or a ring in a molecule which can be calculated from the following formula,
Here,
The
The chemical shift for the protons at carbon bonded to electron withdrawing group increases due to de-shielding effect.
Answer to Problem 36P
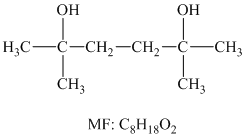
Solution: The structure for the diol having molecular formula
Explanation of Solution
As it is mentioned that the diol having molecular formula
As the oxygen atoms have no effect on index of hydrogen deficiency, the molecular formula of the given compound can be written as
Thus, the index of hydrogen deficiency in the given molecule can be calculated as follows:
This shows that the molecule does not have a ring or multiple bond. It is saturated compound.
The approximate chemical shift values from the given
a)
b)
c)
The arrangement of atoms in a molecule form the given chemical shift values are as follows:
The chemical shift at
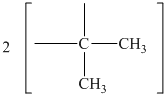
The peak at
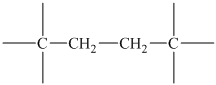
The peak at
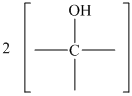
Hence, the structure of diol having molecular formula
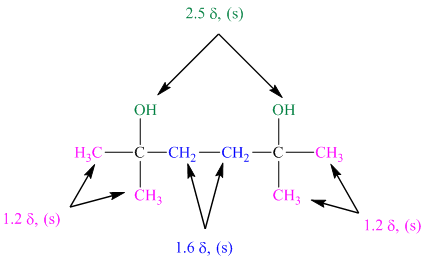
The structure of diol having molecular formula
Want to see more full solutions like this?
Chapter 16 Solutions
Organic Chemistry - Standalone book
- Propose a structural formula for each compound consistent with its 1H-NMR and 13C-NMR spectra. (a) C5H10O2 (b) C7H14O2 (c) C6 H12O2 (d) C7H12O4 (e) C4H7ClO2 (f) C4H6O2arrow_forwardwhat is the structure of o-methylphenoxide anion and what is its pKa?arrow_forwardA. explain each step with words on how to get the structure. Step 1: Calculate the Index of Hydrogen Deficiency (IHD). Step 2: Use IR to determine obvious functional groups. Step 3: Use 1H-NMR to determine structural details. Step 4: Confirm the final structure using 13C-NMR.arrow_forward
- Match the ff reagents: a. Br₂ in CH₂Cl₂ b.CrO₃ , H₂SO₄, acetone c. concentrated HCl with ZnCl₂ d. aqueous FeCl₃ e. aqueous NaHCO₃ f. ammoniacal AgNO₃ 1. 4-bromophenol from benzene 2. hexane from 1-hexene 3. 1-hexanol from hexanal 4. hexane from hexanoic acid 5. 2-hexanol from 2-methyl-2-hexanol 6. 1-hexene from 1-hexynearrow_forward1. Why does H2 not give an IR spectrum? 2. Explain why primary amines and unsubstituted amides have two NH stretching absorptions. 3. Why do anhydrides show two carbonyl peaks? 4. HCl is known to give addition reactions to carbon-carbon double bonds. Why is this behavior not observed in this reaction? 5. Predict the structure of the product expected from addition of molecular bromine to maleic acid.arrow_forward2-cyanophenol (pka 7.0) is even more acidic than the 4-cyanophenol isomer. Propose an explanation of why it is so.arrow_forward
- E. explain each step with words on how to get the structure. Step 1: Calculate the Index of Hydrogen Deficiency (IHD). Step 2: Use IR to determine obvious functional groups. Step 3: Use 1H-NMR to determine structural details. Step 4: Confirm the final structure using 13C-NMR.arrow_forwardGiven the following reagents: a. Br₂ in CH₂Cl₂ b.CrO₃ , H₂SO₄, acetone c. concentrated HCl with ZnCl₂ d. aqueous FeCl₃ e. aqueous NaHCO₃ f. ammoniacal AgNO₃ which will match and quickly differentiate the compounds in the ff: 1. 4-bromophenol from benzene 2. hexane from 1-hexene 3. 1-hexanol from hexanal 4. hexane from hexanoic acid 5. 2-hexanol from 2-methyl-2-hexanol 6. 1-hexene from 1-hexynearrow_forwardCarboxylic acids are acidic enough to dissolve in both 10% NaOH and NaHCO3. Phenols, ArOH, are weak acids and most phenols will not dissolve in 10% NaHCO3. How would you separate a mixture of a carboxylic acid (RCOOH), an amine (RNH2), a phenol (ArOH) and a hydrocarbon (RH) using chemically active extraction? Draw a flow chart to illustrate your separation. Show each species in the aqueous and organic layers.arrow_forward
- 2- Show (draw) or print or bring the 1H-NMR spectra and 13C-NMR and IR spectra formethyl ester.arrow_forwardA compound shows only one peak at 0.9 ppm on its 1H-NMR. Its IR shows only a strong peak right below 3,000 cm-1. Its molar mass is 72. Elemental analysis indicated 83.3% carbon, 16.7% hydrogen. Which compound is it? Pentane 2-Methyl-butan-1-ol 2,2-Dimethylpropane CHO-CH2-CHO Cyclobutanaminearrow_forward
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


