
Concept explainers
Draw structural formulas for (1) the alkyltriphenylphosphonium salt formed by treatment of each
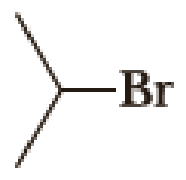
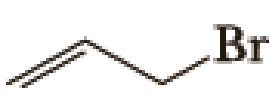
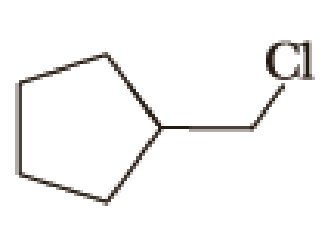
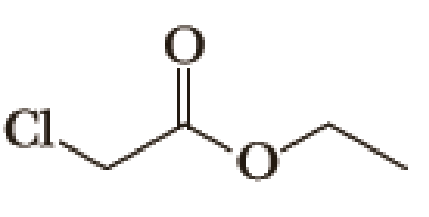

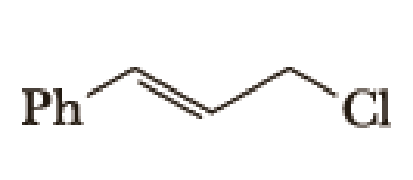
(a)
Interpretation:
The structural formulas for the materials formed in the each steps in the Wittig reaction of given reactant has to be drawn.
Concept introduction:
The functional group in the aldehydes and Ketones are carbonyl group.
Wittig reaction is a reaction in which a nucleophilic elimination occurs after nucleophilic addition reaction. In this reaction an aldehyde or ketone reacts with a triphenyl phosphonium ylide (Wittig reagent) to give an alkene and triphenylphosphine oxide.
The reaction can be represented as shown below,
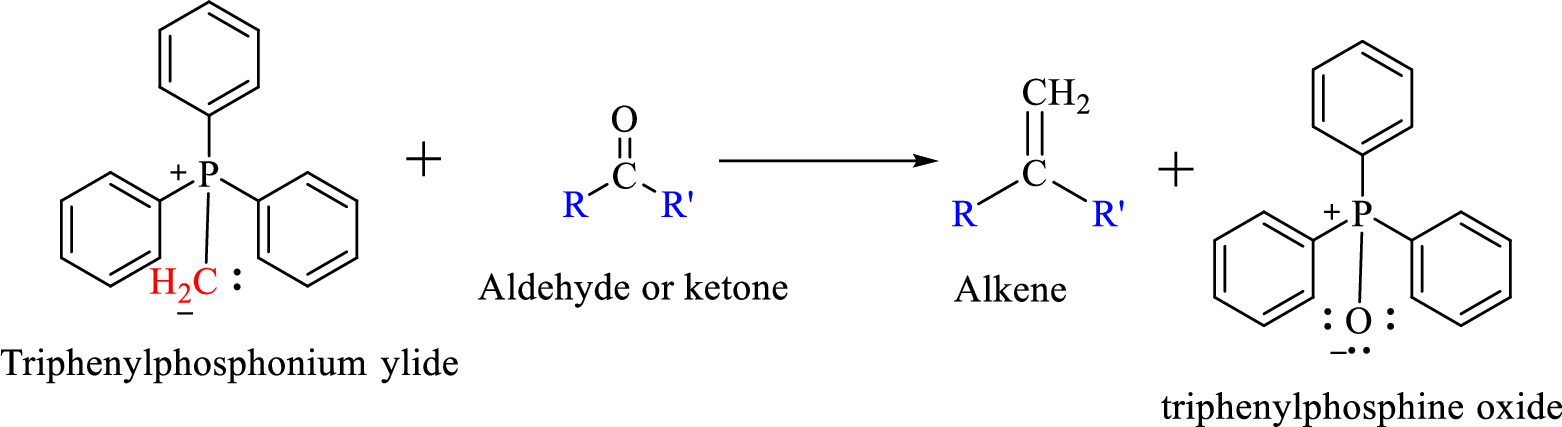
Mechanism:
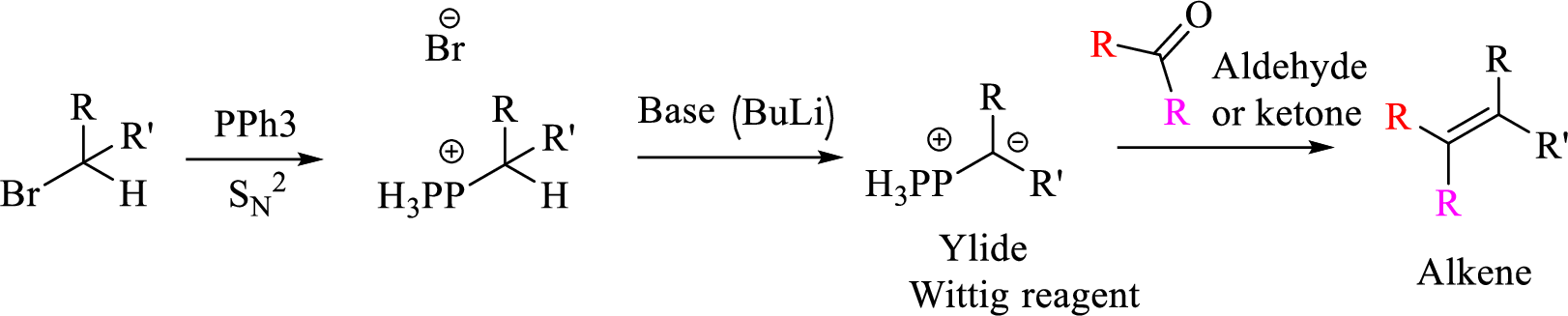
Explanation of Solution
Given halo compound is shown below, 1,2 and 3 in the questions are the important steps in Wittig reaction.
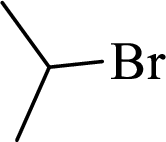
When treating this compound with triphenylphosphine corresponding alkyltriphenylphosphonium salt will form.
Thus, the alkyltriphenylphosphonium salt formed in the reaction (1) can be drawn as follows,
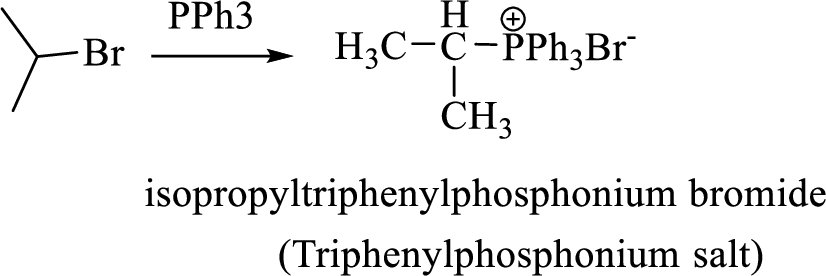
Phosphonium ylide formed by treatment of this phosphonium salt with butyl lithium (2) can be drawn as shown below,
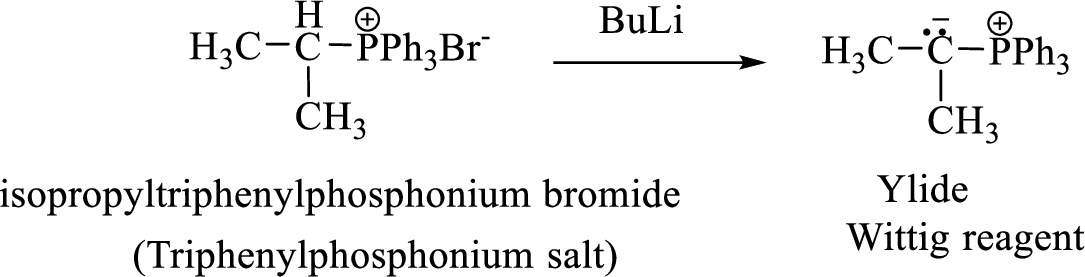
(3) The alkene formed by treatment of this phosphonium ylide with acetone is drawn below,
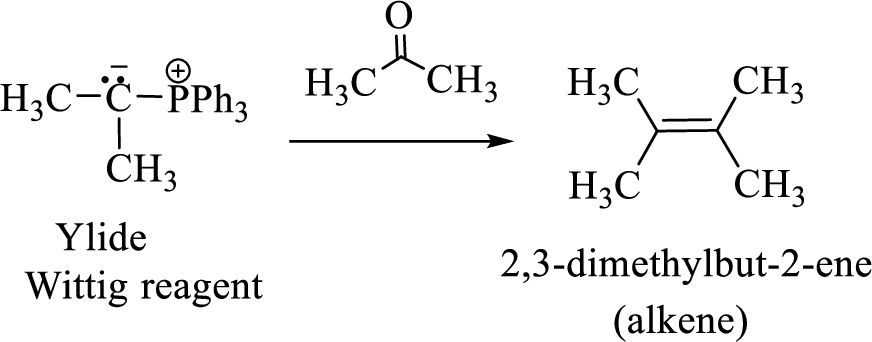
(b)
Interpretation:
The structural formulas for the materials formed in the each steps in the Wittig reaction of given reactant has to be drawn.
Concept introduction:
The functional group in the aldehydes and Ketones are carbonyl group.
Wittig reaction is a reaction in which a nucleophilic elimination occurs after nucleophilic addition reaction. In this reaction an aldehyde or ketone reacts with a triphenyl phosphonium ylide (Wittig reagent) to give an alkene and triphenylphosphine oxide.
The reaction can be represented as shown below,
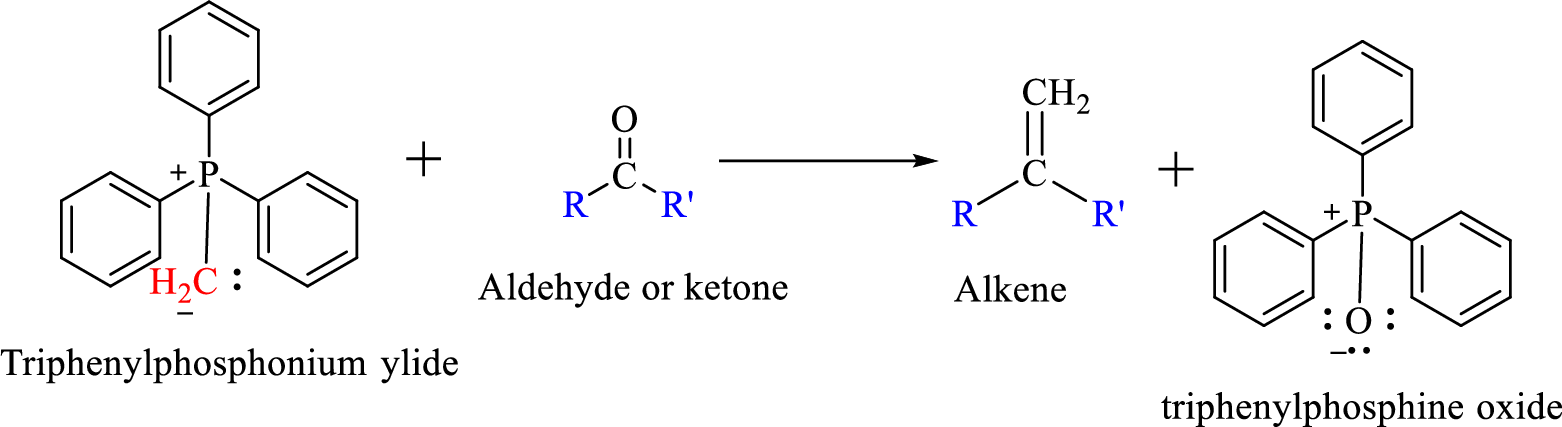
Mechanism:
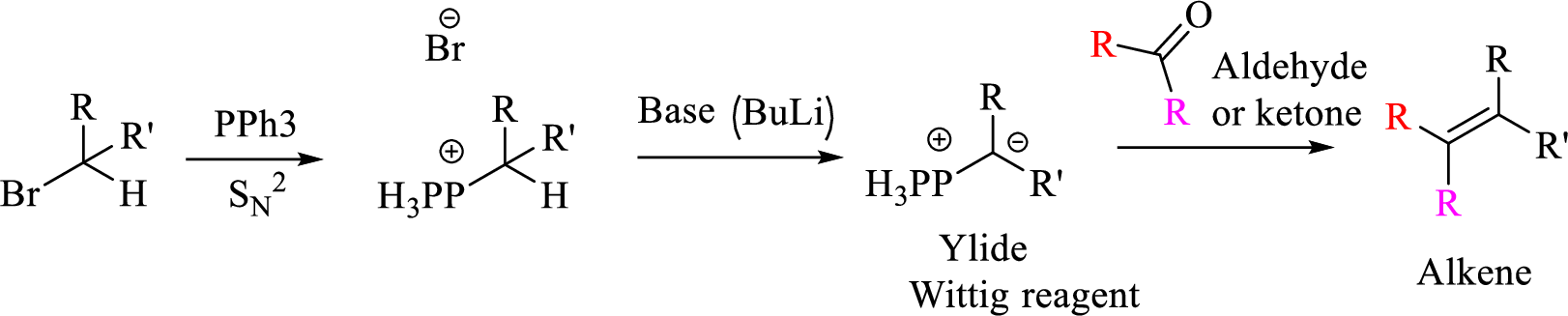
Explanation of Solution
Given halo compound is shown below, 1,2 and 3 in the questions are the important steps in Wittig reaction.
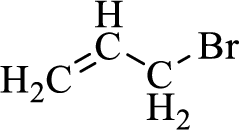
When treating this compound with triphenylphosphine corresponding alkyltriphenylphosphonium salt will form.
Thus, the alkyltriphenylphosphonium salt formed in the reaction (1) can be drawn as follows,
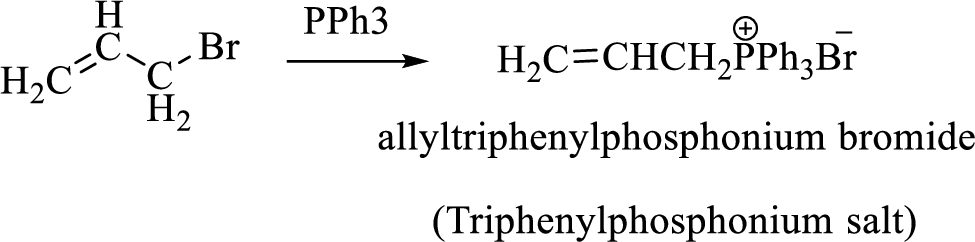
Phosphonium ylide formed by treatment of this phosphonium salt with butyl lithium (2) can be drawn as shown below,
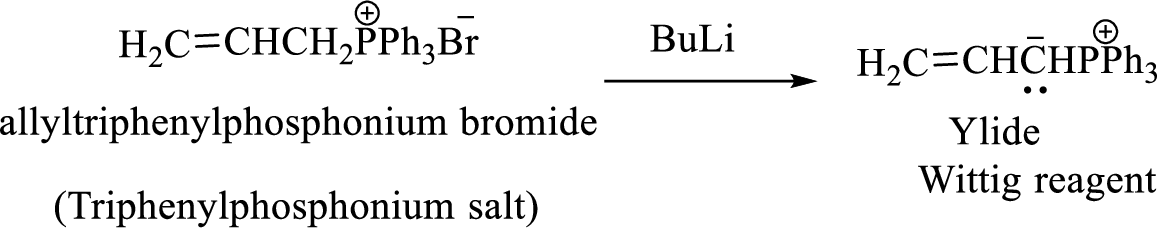
(3) The alkene formed by treatment of this phosphonium ylide with acetone is drawn below,
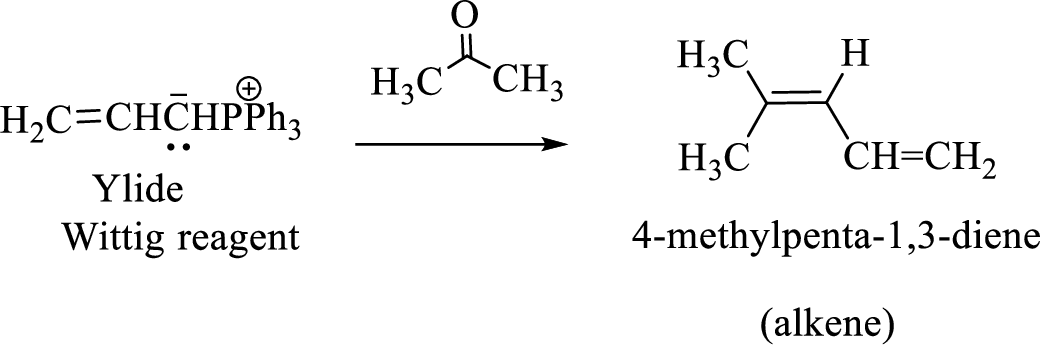
(c)
Interpretation:
The structural formulas for the materials formed in the each steps in the Wittig reaction of given reactant has to be drawn.
Concept introduction:
The functional group in the aldehydes and Ketones are carbonyl group.
Wittig reaction is a reaction in which a nucleophilic elimination occurs after nucleophilic addition reaction. In this reaction an aldehyde or ketone reacts with a triphenyl phosphonium ylide (Wittig reagent) to give an alkene and triphenylphosphine oxide.
The reaction can be represented as shown below,
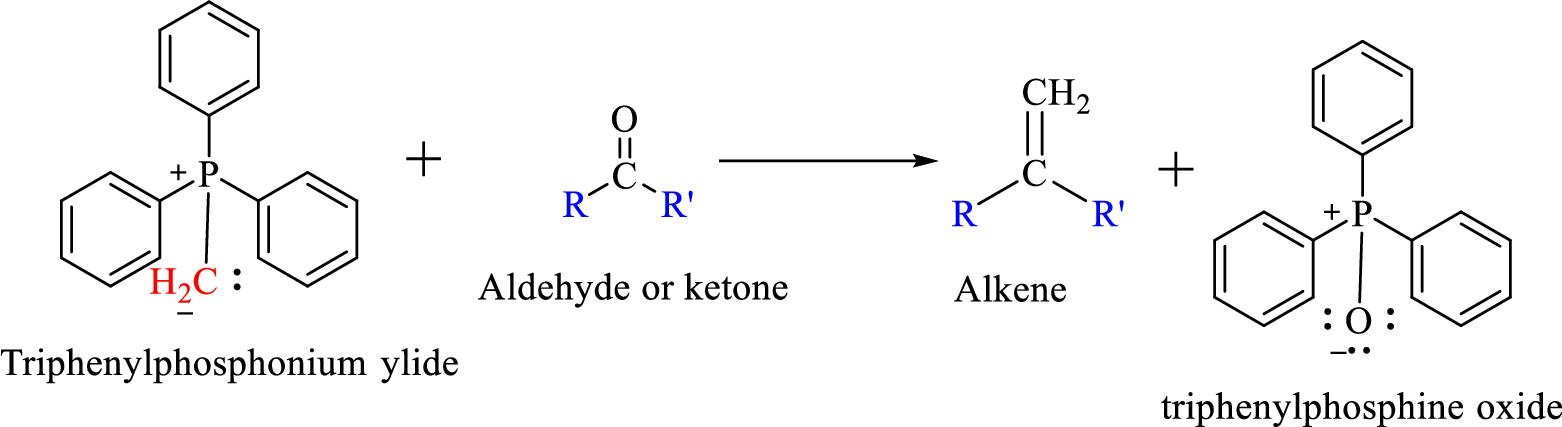
Mechanism:
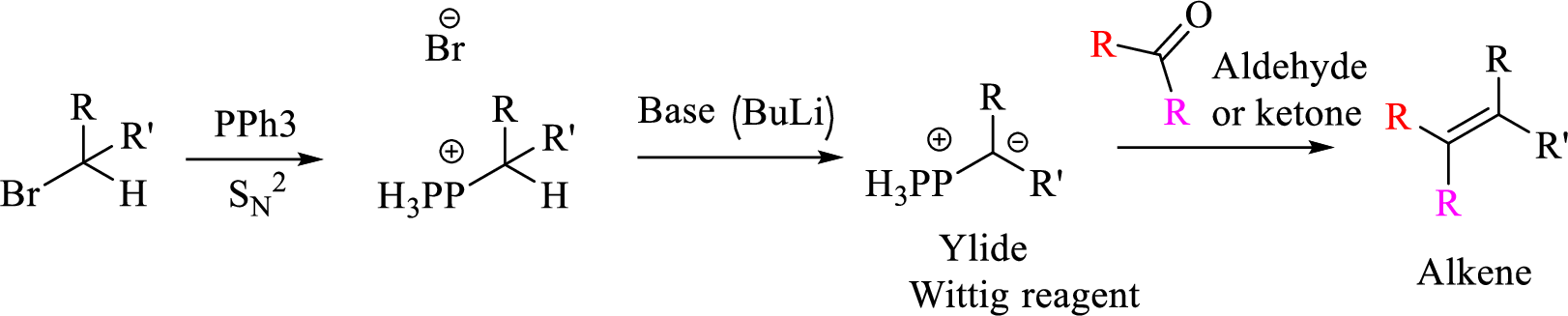
Explanation of Solution
Given halo compound is shown below, 1,2 and 3 in the questions are the important steps in Wittig reaction.
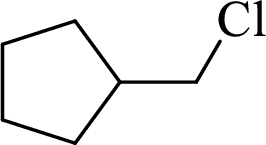
When treating this compound with triphenylphosphine corresponding alkyltriphenylphosphonium salt will form.
Thus, the alkyltriphenylphosphonium salt formed in the reaction (1) can be drawn as follows,
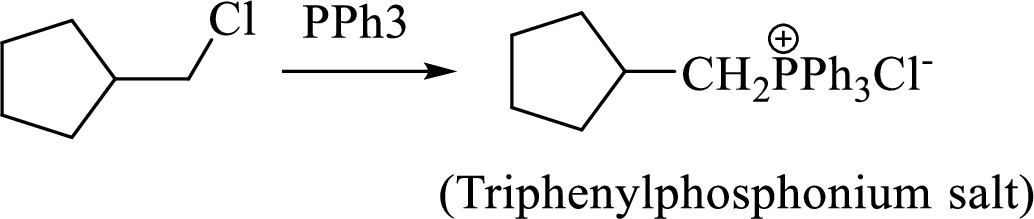
Phosphonium ylide formed by treatment of this phosphonium salt with butyl lithium (2) can be drawn as shown below,
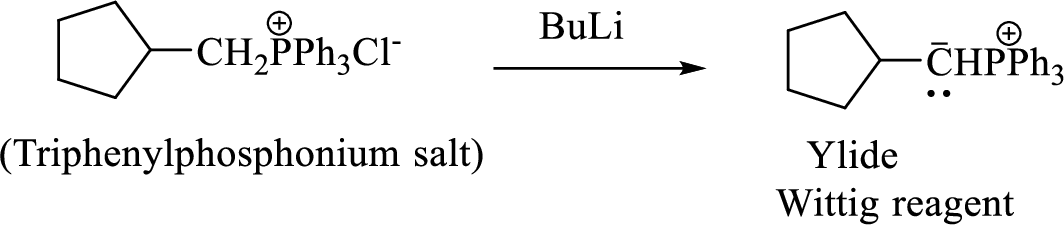
(3) The alkene formed by treatment of this phosphonium ylide with acetone is drawn below,
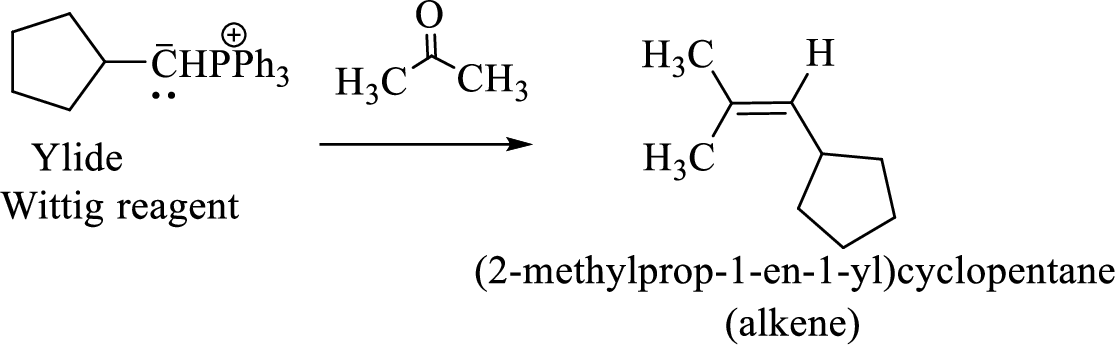
(d)
Interpretation:
The structural formulas for the materials formed in the each steps in the Wittig reaction of given reactant has to be drawn.
Concept introduction:
The functional group in the aldehydes and Ketones are carbonyl group.
Wittig reaction is a reaction in which a nucleophilic elimination occurs after nucleophilic addition reaction. In this reaction an aldehyde or ketone reacts with a triphenyl phosphonium ylide (Wittig reagent) to give an alkene and triphenylphosphine oxide.
The reaction can be represented as shown below,
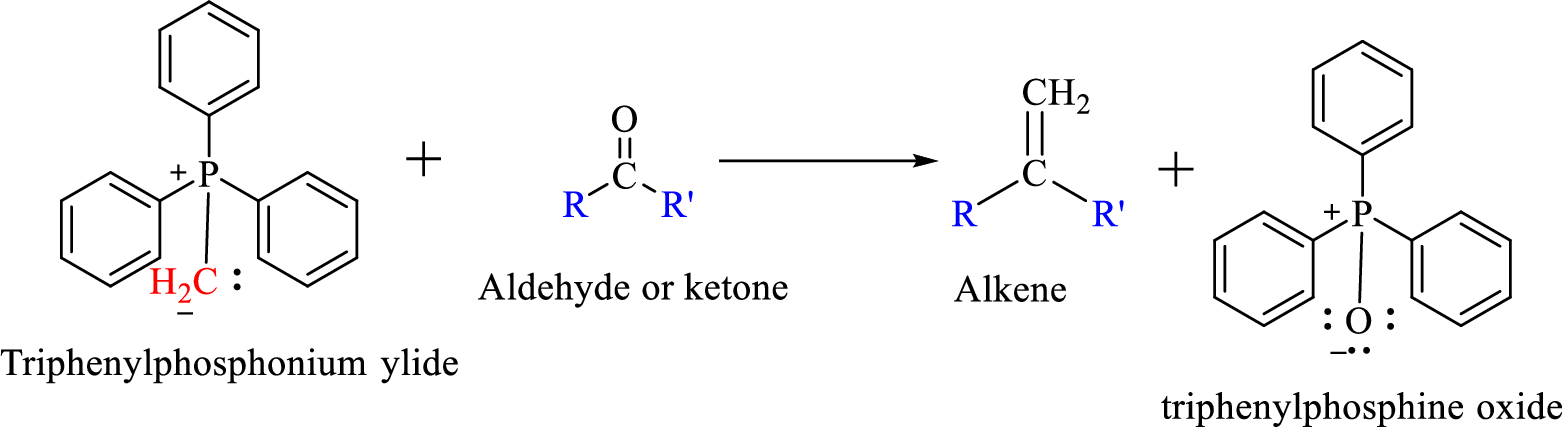
Mechanism:
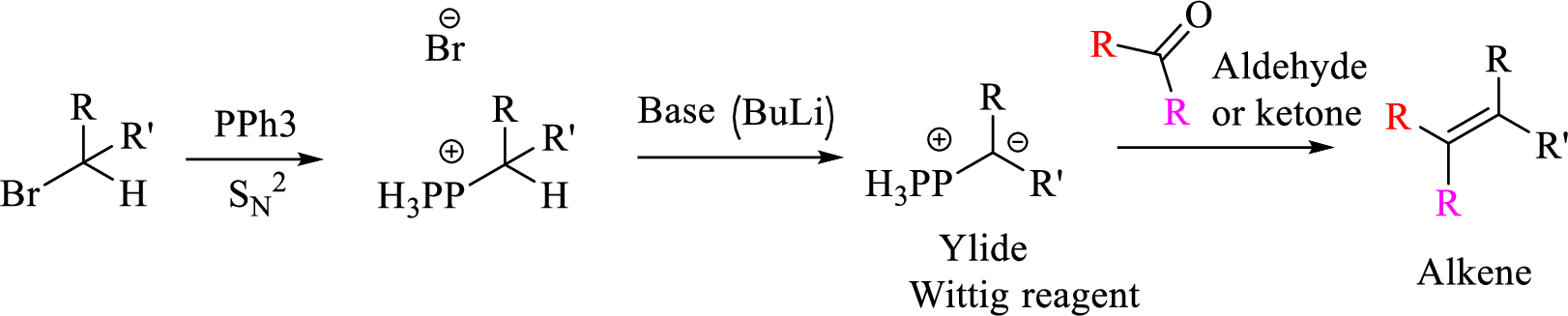
Explanation of Solution
Given halo compound is shown below, 1,2 and 3 in the questions are the important steps in Wittig reaction.
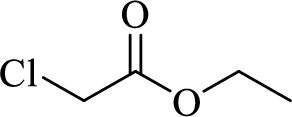
When treating this compound with triphenylphosphine corresponding alkyltriphenylphosphonium salt will form.
Thus, the alkyltriphenylphosphonium salt formed in the reaction (1) can be drawn as follows,
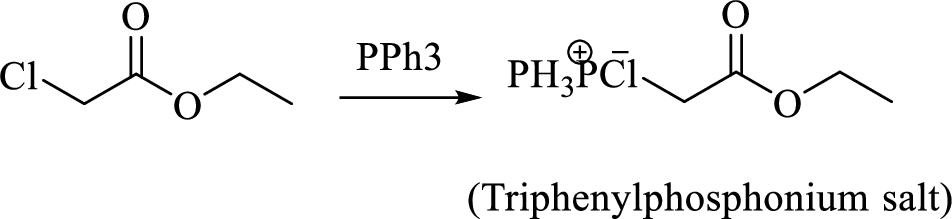
Phosphonium ylide formed by treatment of this phosphonium salt with butyl lithium (2) can be drawn as shown below,
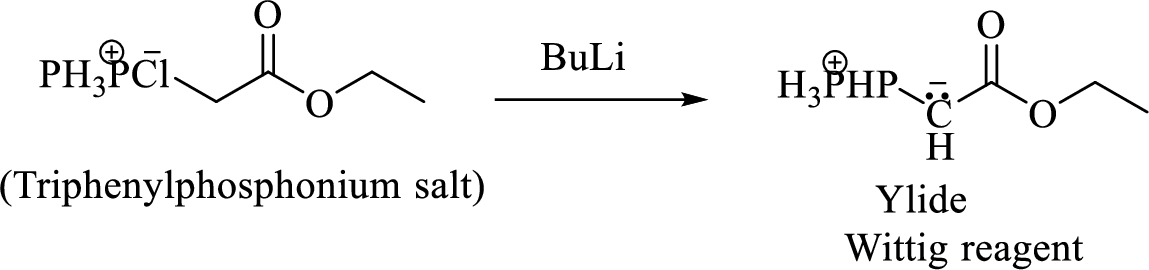
(3) The alkene formed by treatment of this phosphonium ylide with acetone is drawn below,
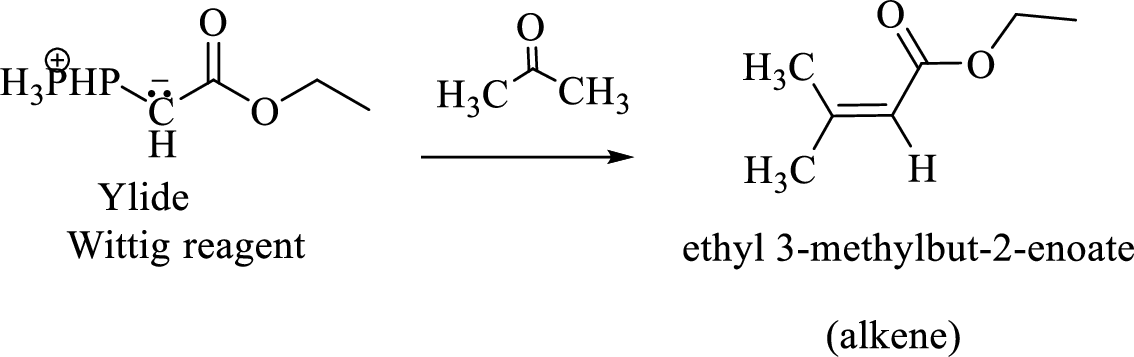
(e)
Interpretation:
The structural formulas for the materials formed in the each steps in the Wittig reaction of given reactant has to be drawn.
Concept introduction:
The functional group in the aldehydes and Ketones are carbonyl group.
Wittig reaction is a reaction in which a nucleophilic elimination occurs after nucleophilic addition reaction. In this reaction an aldehyde or ketone reacts with a triphenyl phosphonium ylide (Wittig reagent) to give an alkene and triphenylphosphine oxide.
The reaction can be represented as shown below,
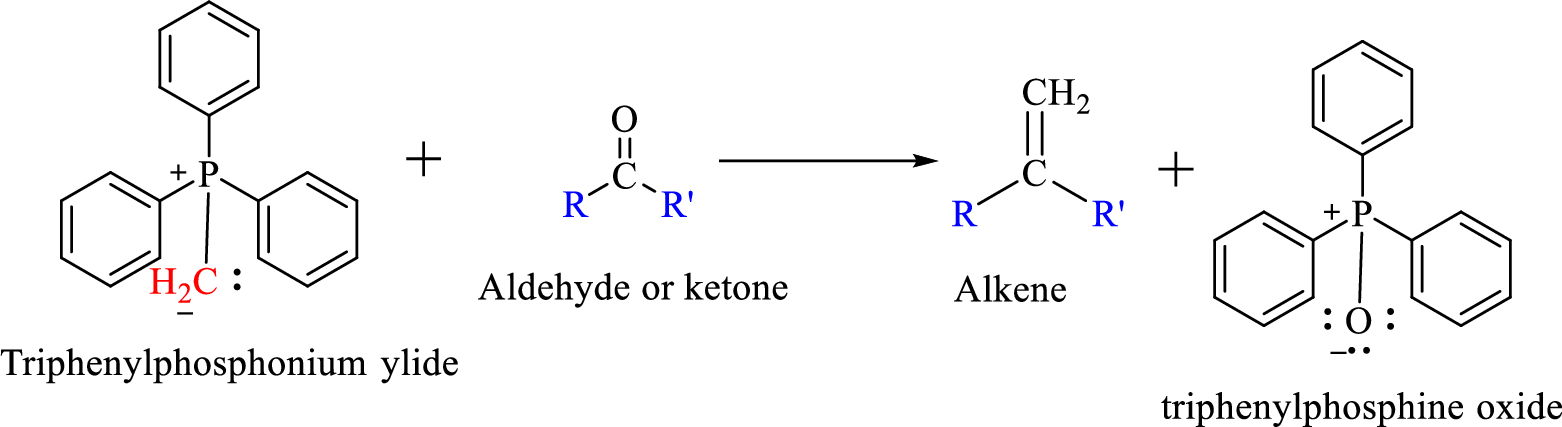
Mechanism:
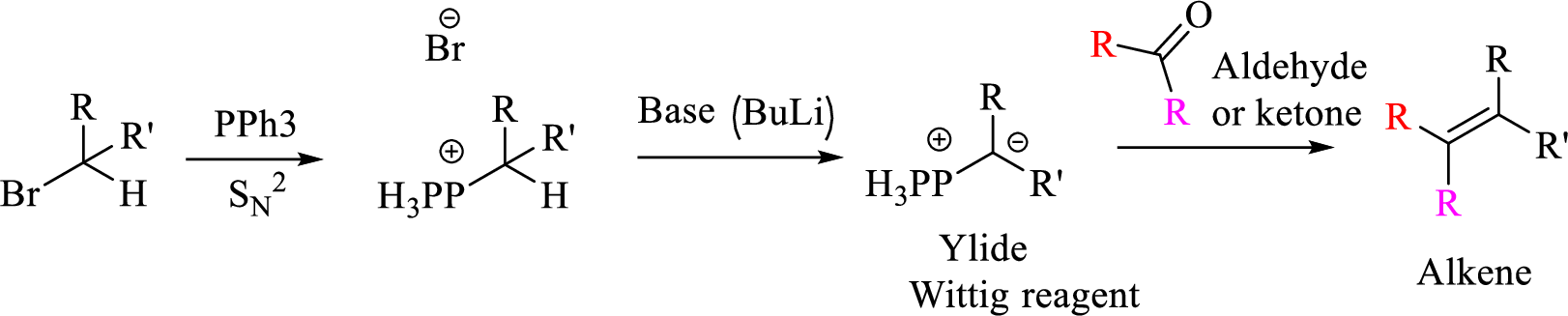
Explanation of Solution
Given halo compound is shown below, 1,2 and 3 in the questions are the important steps in Wittig reaction.

When treating this compound with triphenylphosphine corresponding alkyltriphenylphosphonium salt will form.
Thus, the alkyltriphenylphosphonium salt formed in the reaction (1) can be drawn as follows,
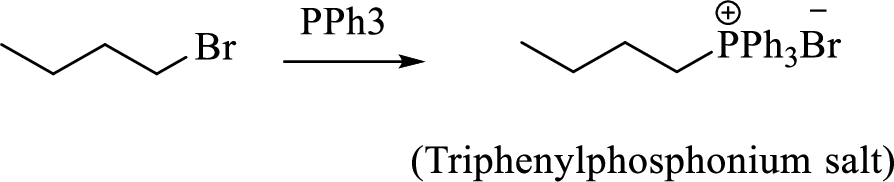
Phosphonium ylide formed by treatment of this phosphonium salt with butyl lithium (2) can be drawn as shown below,
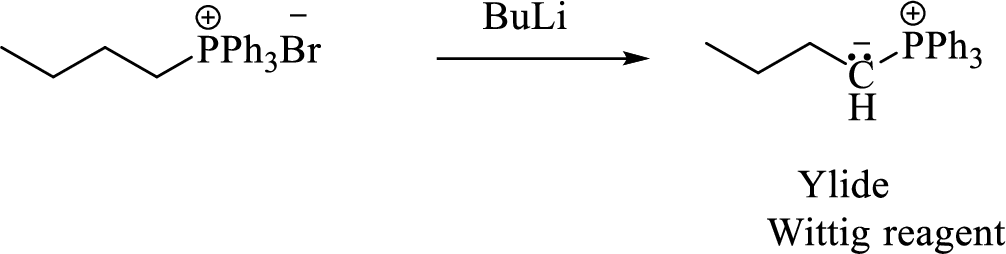
(3) The alkene formed by treatment of this phosphonium ylide with acetone is drawn below,
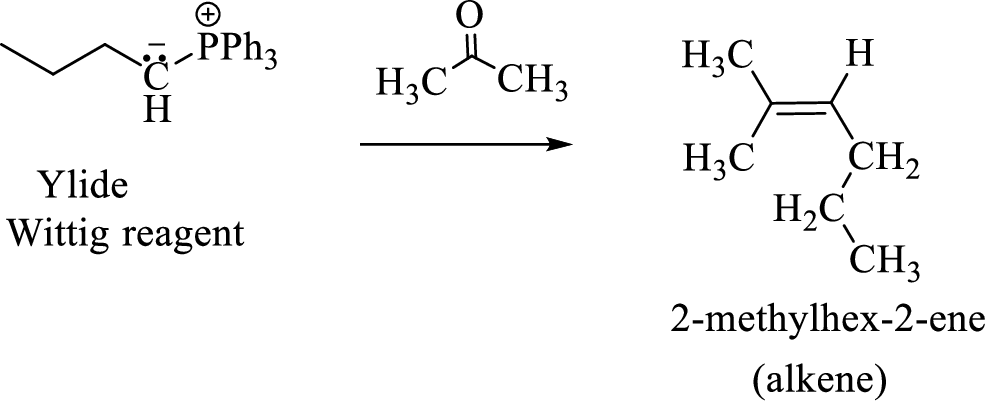
(f)
Interpretation:
The structural formulas for the materials formed in the each steps in the Wittig reaction of given reactant has to be drawn.
Concept introduction:
The functional group in the aldehydes and Ketones are carbonyl group.
Wittig reaction is a reaction in which a nucleophilic elimination occurs after nucleophilic addition reaction. In this reaction an aldehyde or ketone reacts with a triphenyl phosphonium ylide (Wittig reagent) to give an alkene and triphenylphosphine oxide.
The reaction can be represented as shown below,
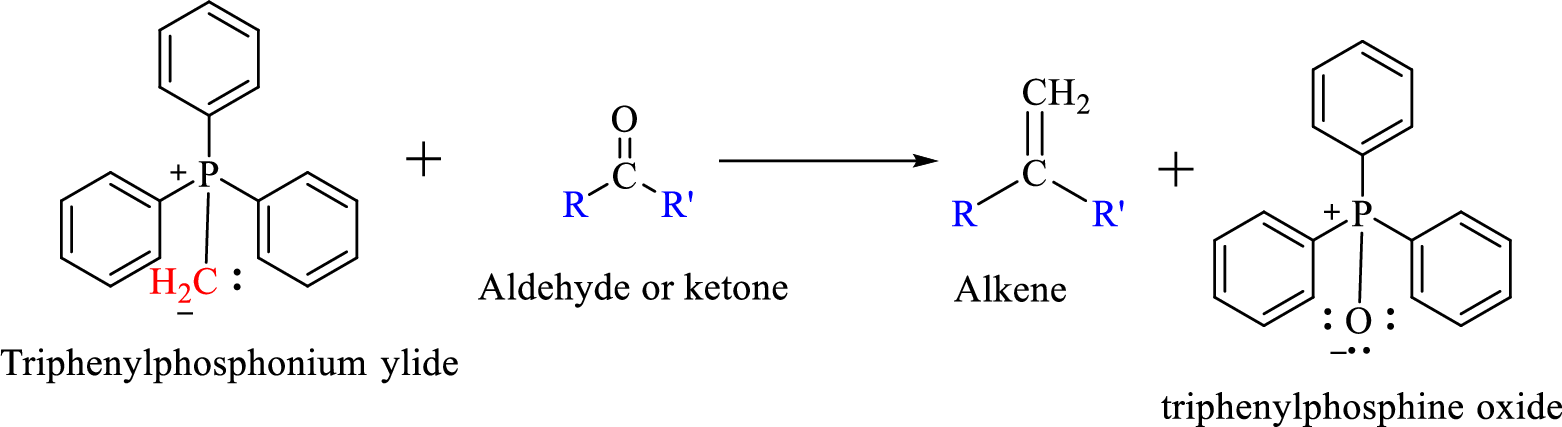
Mechanism:
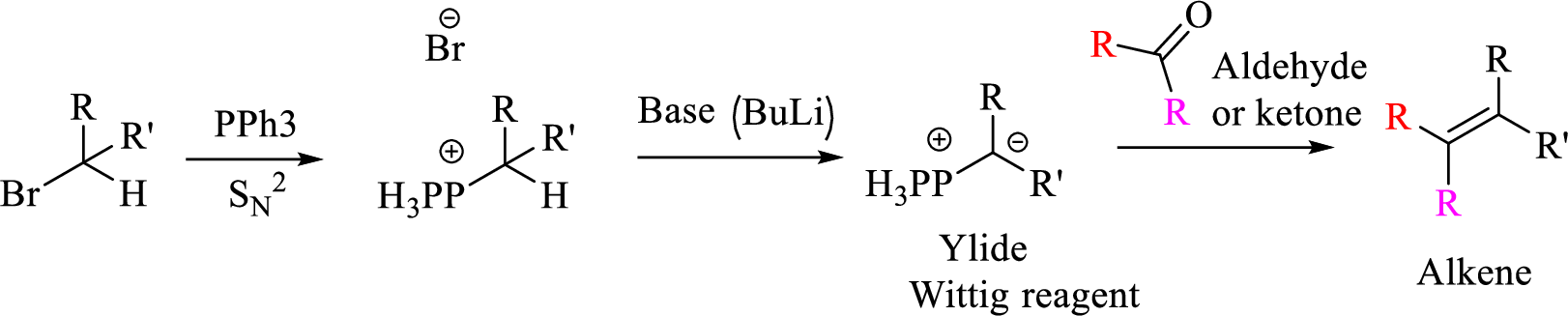
Explanation of Solution
Given halo compound is shown below, 1,2 and 3 in the questions are the important steps in Wittig reaction.

When treating this compound with triphenylphosphine corresponding alkyltriphenylphosphonium salt will form.
Thus, the alkyltriphenylphosphonium salt formed in the reaction (1) can be drawn as follows,
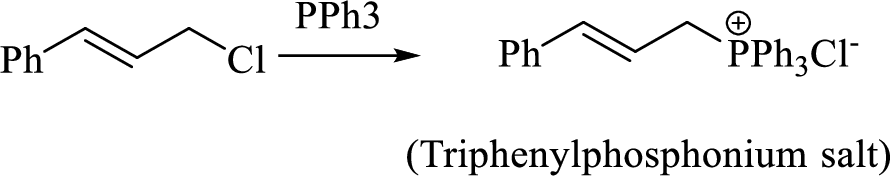
Phosphonium ylide formed by treatment of this phosphonium salt with butyl lithium (2) can be drawn as shown below,
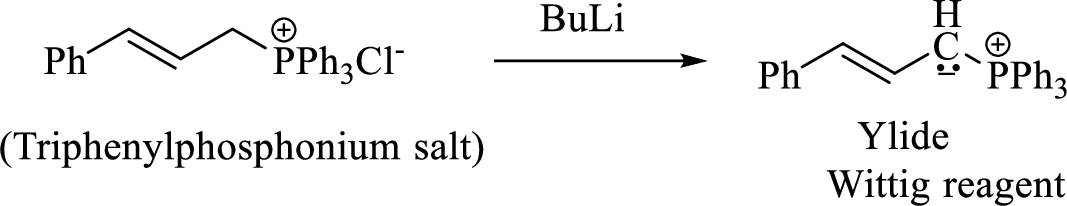
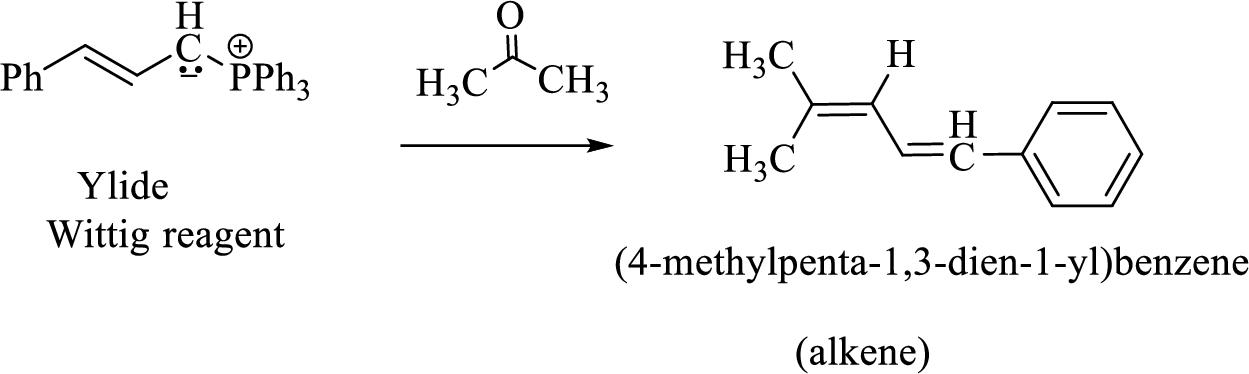
(3) The alkene formed by treatment of this phosphonium ylide with acetone is drawn below,
Want to see more full solutions like this?
Chapter 16 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Chemistry: Structure and Properties (2nd Edition)
Thermodynamics, Statistical Thermodynamics, & Kinetics
Essential Organic Chemistry (3rd Edition)
Chemistry: The Molecular Nature of Matter
Chemistry: A Molecular Approach (4th Edition)
- Describe how 3-methyl-1-phenyl-3-pentanol can be prepared from benzene. You can use any inorganic reagents and solvents, and any organic reagents provided they contain no more than two carbons.arrow_forwardAcetylene reacts with sodium amide in the presence of propyl halide produces aldehyde produces ketones It produces 2-pentanearrow_forwardShow how to synthesize carboxylic acids from oxidation of alcohols and aldehydes,carboxylation of Grignard reagents, oxidative cleavage of alkenes and alkynes,hydrolysis of nitriles, and oxidation of alkylbenzenesarrow_forward
- Draw the principal organic product for the reaction of 1-bromopentane with lithium in diethyl ether, followed by formaldehyde in diethyl ether, and then followed by dilute acid.arrow_forwardThe ketone 2-heptanone has been identified as contributing to the odor of a number of dairy products, including condensed milk and cheddar cheese. Describe the synthesis of 2-heptanone from acetylene and any necessary organic and inorganic reagents.arrow_forward11) Synthesize 2-methyl-3-hydroxycyclohexone from cyclohexone, methyl iodide, and inorganic precursors.arrow_forward
- Ethyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l). The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 8.50 gof butanoic acid and excess ethanol? Express your answer in grams to three significant figures.arrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) Given 8.50 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100%yield? Express your answer in grams to three significant figures.arrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) a) Given 7.70 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100% yield? b) A chemist ran the reaction and obtained 5.25 g of ethyl butyrate. What was the percent yield? c) The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 7.70 g of butanoic acid and excess ethanol?arrow_forward
- Draw the structure of each product from the reaction of benzene with 2-chloro-1-methylcyclohexane using AlCl 3 as the catalyst and Identify the major product.arrow_forwardDraw the structural formula of the enol formed in each alkyne hydration reaction; then draw the structural formula of the carbonyl compound with which each enol is in equilibriumarrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring. It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l)CH3CH2CH2CO2H(l)+CH2CH3OH(l)⟶H+CH3CH2CH2CO2CH2CH3(l)+H2O(l) A chemist ran the reaction and obtained 5.40 g of ethyl butyrate. What was the percent yield, The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 7.45g of butanoic acid and excess ethanol?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
