
Concept explainers
a)
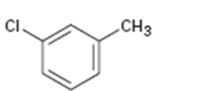
Interpretation:
Whether the compound given is ortho-, meta-, or para-disubstituted is to be stated.
Concept introduction:
Disubstituted benzenes are named using the prefixes ortho (o), meta (m) and para (p). An ortho-disubstituted benzene has its two substituent groups in a 1,2-relationship on the ring. A meta-disubstituted benzene has its two substituent groups in a 1,3-relationship on the ring. A para-disubstituted benzene has its two substituent groups in a 1,4-relationship on the ring.
To state:
Whether the compound given is ortho-, meta-, or para-disubstituted.
b)
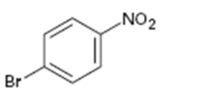
Interpretation:
Whether the compound given is ortho-, meta-, or para-disubstituted is to be stated.
Concept introduction:
Disubstituted benzenes are named using the prefixes ortho (o), meta (m) and para (p). An ortho-disubstituted benzene has its two substituent groups in a 1,2-relationship on the ring. A meta-disubstituted benzene has its two substituent groups in a 1,3-relationship on the ring. A para-disubstituted benzene has its two substituent groups in a 1,4-relationship on the ring.
To state:
Whether the compound given is ortho-, meta-, or para-disubstituted.
c)
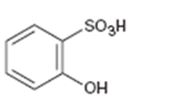
Interpretation:
Whether the compound given is ortho-, meta-, or para-disubstituted is to be stated.
Concept introduction:
Disubstituted benzenes are named using the prefixes ortho (o), meta (m) and para (p). An ortho-disubstituted benzene has its two substituent groups in a 1,2-relationship on the ring. A meta-disubstituted benzene has its two substituent groups in a 1,3-relationship on the ring. A para-disubstituted benzene has its two substituent groups in a 1,4-relationship on the ring.
To state:
Whether the compound given is ortho-, meta-, or para-disubstituted.
Trending nowThis is a popular solution!

Chapter 15 Solutions
Organic Chemistry
- Draw the constitutional isomer formed when the following alkenes are treated with each set of reagents: [1] H2O, H2SO4; or [2] BH3 followed by H2O2, −OH.arrow_forwardDraw the structure of all compounds that fit the following descriptions. twelve constitutional isomers having the molecular formula C6H12 andcontaining one ringarrow_forwardWhat bromo-substituted compound would be required to react with (CH2 = CH)2CuLi in order to form each of the following compounds?arrow_forward
- What are the relative positions of the substituents in the following compound. A. ipso B. ortho C. peri D. metaarrow_forwardOne of the principal components of lemongrass oil is limonene, C10H16. When limoneneis treated with excess hydrogen and a platinum catalyst, the product is an alkane offormula C10H20. What can you conclude about the structure of limonene?arrow_forwardIdentify the substituents and the systematic name of the compound.arrow_forward
- Use the molecular formula C8H15Br to illustrate thefollowing isomers:1.1.1 positional isomers 1.1.2 (-)-gauche en syn-periplanêre rotamere 1.1.3 E- en Z-alkene 1.1.4 enantiomere (gee ook beskrywers)arrow_forward1. Ethylene glycol, a major component of antifreeze, becomes especially poisonous when it is oxidized by the liver. Draw two oxidation products that could be derived from ethylene glycol. 2.When 2-methyl-1-butanol is dehydrated in an acid medi- um to an alkene, it yelds mainly 2-methyl-2-butene rather than 2-methyl-1-butene. This indicates that the dehydra- tion to an alkene is at least a two-step reaction. Suggest a mechanism to explain the reaction.arrow_forwardA 2-bromobutane react with methanol and form a enantiomeric pair of 2-methoxybutane. Draw the structures of the enntiomeric pairs of ethers.arrow_forward
- a. What alkane, with molecular formula C5H12, forms only one monochlorinated product when it is heated with Cl2? b. What alkane, with molecular formula C7H16, forms seven monochlorinated products (disregarding stereoisomers) when heated with Cl2?arrow_forwardDraw the constitutional isomer formed when the attached alkenes are treated with each set of reagents: [1] H2O, H2SO4; or [2] BH3 followed by H2O2, −OH.arrow_forwardAre the compounds in each pair constitutional isomers or are they not isomers of each other?arrow_forward
