
Concept explainers
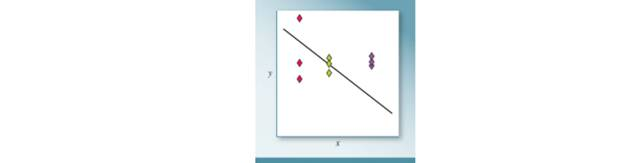
The true dependence of y on x is represented by the line. Three students measured y as a function of x and plotted their data on the graph. Which set of data has the best accuracy and which has the best precision, respectively?
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Chemistry
- The accepted (true) density of aluminum is 2.70 g/cm3. If a student, after performing an experiment, found the value to be 2.45 g/cm3, calculate the percent error for this value. Group of answer choicesarrow_forwardPerform the calculation and record the answer with the correct number of significant figures. (34.123 + 1.50) (98.7654 8.100) 0.40 Incorrectarrow_forwardAn electric current of 155.0 mA flows for 42.0 minutes. Calculate the amount of electric charge transported.. Be sure your answer has the correct unit symbol and 3 significant digits. 0 x10 μ 0 9 Xarrow_forward
- Find the difference between the value of x2 and the sum of x1 plus x3.; let x4 be equivalent to the sum of x1 and x3 and let x5 be the difference between x2 and x5. This calculation scheme is shown below. Make notes about any similarities or differences between the values in your notes. x4 = x1 + x3 x5 = x2 – x4 Data Analyis This section will include all data collected during the lab. Thermochemical Data Tinitial (°C) Tfinal (°C) ΔT (°C) moles NaOH qreaction (kJ) ΔHrxn Reaction 1 25.0 30.3 +5.3 0.025 -1.11 -44.4 Reaction 2 25.0 37.0 +12.0 0.025 -2.51 -100.4 Reaction 3 25.0 31.7 +6.7 0.025 -1.40 -56.1 Reaction 1: NaOH(s) → Na+(aq) + OH-(aq) + x1 kJ 1g /39.977g/mol = 0.025 moles Moles NaOH = 0.025 qsolution = (4.184 J/g °C) (50.0g) (30.3°C -25.0°C) = -1108.76 J/ 1000 qreaction (kJ) = -1.11 kJ ΔH = -1.11 kJ/ 0.025 moles ΔHrxn = -44.4 kJ/mol…arrow_forwardBob and Jill are measuring the height of Mr. Chenet (Mr Chenet is 1.8593 meters tall). Bob gets a measurement of 1.6329, while Jill gets a measurement of 1.8. This means that Bob is more accurate, while Jill is more precise. True or False?arrow_forwardA student determines the density of a metal to be 8.55g/cm3. If the theoretical value is 8.91g/cm3, what is the percent error? Group of answer choices 4.0% 1.2% 3.6% 4.2%arrow_forward
- 1. Perform the following calculations and give the answer rounded to the correct number of significant figures: [(3.35 x 1.06) + (21 1.773)] The answer isarrow_forwardGive an example from which the difference of Accuracy and Precision is described and explain why it is soarrow_forwardChemistry 130 Experiment 5-Dota Anolysis for Accuracy and Precision 3. A second group of students performed the experiment to determine the percentage of sugar in Bazooka chewing gum. The student's results were 5.5%, 66.5%, 66.2%, and 66,7%. Should the students take the average of all four measurements? Why or why not? Will including the measurement affect the accuracy, precision, or both - why? Instructor Initials 4. A third group of students performed the experiment to determine the percentage of sugar in Bazooka chewing gum. The results were 62.5%, 68.5%, and 62.5%, and 67.7%. The mean is 65.3%. The average deviation is 2.8%. (a) Calculate the RAD in ppt. (show equation). Based on the RAD, how precise where the students? Explain. (b) Assuming the theoretical value of sugar in Bazooka chewing gum is 66.7%, calculate the student's percent error (show equation). (c) Compare the precision and the accuracy. How would you describe the data? Explain.arrow_forward
- What is the uncertainty in the measured value 12.37040 after it is rounded to the following number of significant figures? The uncertainty of 2 is what?arrow_forwardThe theoretical density of metal is 2.70g/ cm cube. The experimental density a student found in the lab is 2.68g/ cm cube. What is the percent errorarrow_forwardChoose the letter of the correct answer. 1) Three students weigh a standard mass of 3.00 g on the same balance. Each students obtains a reading of exactly 1.85 g for the mass of the standard. What do these results imply? a) The balance used is accurate but nit precise b) The balance used is precise but not accurate c) The balance used is both accurate and precise d) The balance used is neither accurate nor precise 2) Aria and Jessi determined the mass of an object on two different balances. The accepted mass of the object was 2.355 g. Vivz obtained the following values: 2.531 g, 2.537 g, 2.535 g, and 2.533 g. Kenny obtained the values of 2.350 g, 2.404 g, 2.296 g, and 2.370 g. Which statement best describes the results that were obtained by Vivz and Kenny? a) Aria obtained the better precision. b) Aria obtained the better accuracy. c) Jessi obtained the better accuracy. d) Jessi obtained the better precision.arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning





