
Concept explainers
(a)
Interpretation: To determine whether oxaloacetate is associated with (1) transamination, (2) oxidative deamination, or (3) the urea cycle.
Concept introduction: Transamination reaction is a biochemical reaction that involves the transfer of an amino group. In transamination reaction exchange of an amino group from an
A biochemical reaction in which an
A urea cycle is a cyclic biochemical pathway that involves the production of urea using ammonium ions and aspartate molecules as nitrogen sources. The reactants in the formation of carbamoyl phosphate are ammonium ion, water, and carbon dioxide.
(a)
Answer to Problem 15.73EP
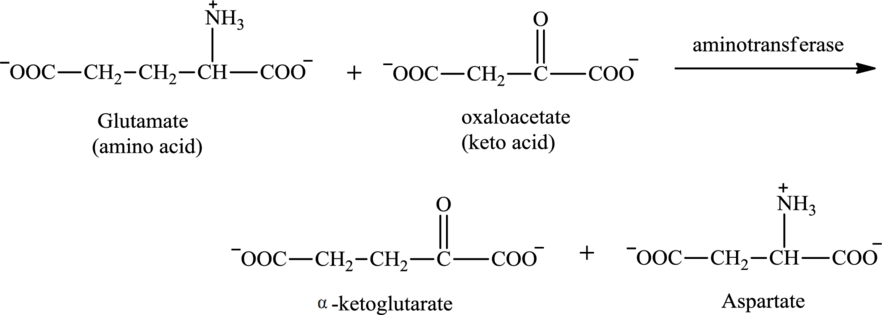
Oxaloacetate is associated with the transamination reaction.
Explanation of Solution
The structure of oxaloacetate is:
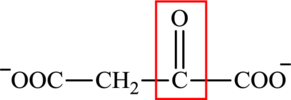
Oxaloacetate contains both a carbonyl group and a carboxyl
(b)
Interpretation: To determine whether arginine is associated with (1) transamination, (2) oxidative deamination, or (3) the urea cycle.
Concept introduction: Transamination reaction is a biochemical reaction that involves the transfer of an amino group. In transamination reaction exchange of an amino group from an
A biochemical reaction in which an
A urea cycle is a cyclic biochemical pathway that involves the production of urea using ammonium ions and aspartate molecules as nitrogen sources. The reactants in the formation of carbamoyl phosphate are ammonium ion, water, and carbon dioxide.
(b)
Answer to Problem 15.73EP
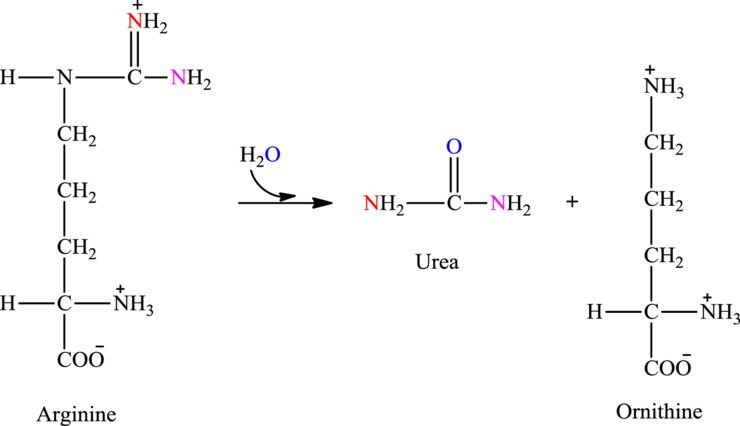
Arginine is associated with the urea cycle.
Explanation of Solution
In step 3 of the urea cycle, catalyst argininosuccinate lyase catalyzes the cleavage of argininosuccinate into arginine and fumarate. Arginine formed step 3 reacts with water and undergo hydrolysis in step 4.
Hydrolysis of arginine produce urea and also regenerates the starting fuel ornithine.
(c)
Interpretation: To determine whether
Concept introduction: Transamination reaction is a biochemical reaction that involves the transfer of an amino group. In transamination reaction exchange of an amino group from an
A biochemical reaction in which an
A urea cycle is a cyclic biochemical pathway that involves the production of urea using ammonium ions and aspartate molecules as nitrogen sources. The reactants in the formation of carbamoyl phosphate are ammonium ion, water, and carbon dioxide.
(c)
Answer to Problem 15.73EP
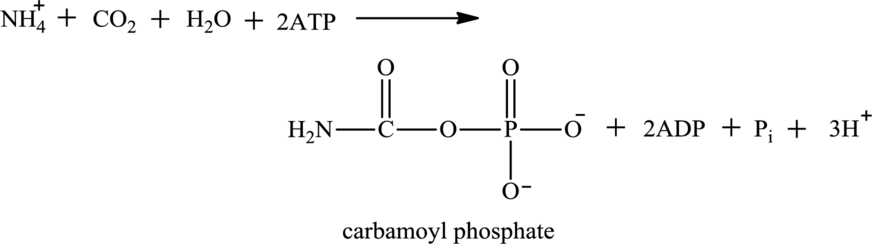
Water
Explanation of Solution
Water is one of the reactants in oxidative deamination reaction. For example, glutamate is an
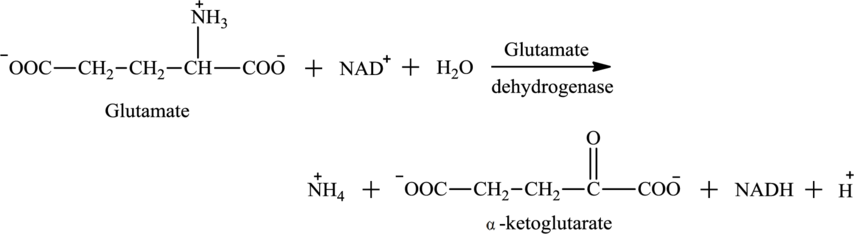
Water is also associated with the urea cycle. Ammonium ion produced from oxidative deamination reaction, carbon dioxide, water, and two ATP molecules reacts to form carbamoyl phosphate. Carbamoyl phosphate is fuel for the urea cycle. The
(d)
Interpretation: To determine whether ATP is associated with (1) transamination, (2) oxidative deamination, or (3) the urea cycle.
Concept introduction: Transamination reaction is a biochemical reaction that involves the transfer of an amino group. In transamination reaction exchange of an amino group from an
A biochemical reaction in which an
A urea cycle is a cyclic biochemical pathway that involves the production of urea using ammonium ions and aspartate molecules as nitrogen sources. The reactants in the formation of carbamoyl phosphate are ammonium ion, water, and carbon dioxide.
(d)
Answer to Problem 15.73EP
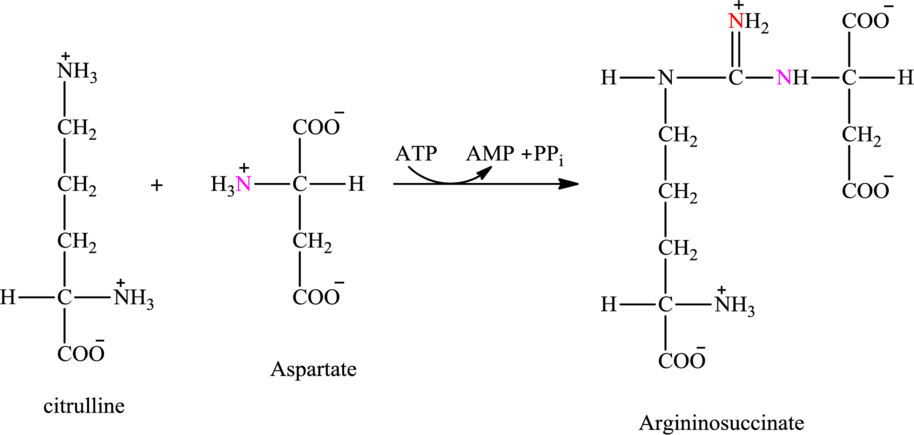
ATP is associated with the urea cycle.
Explanation of Solution
Adenosine triphosphate (ATP) is a molecule that is defined as the energy currency of life and provides energy to carry out the
Step 2 of the urea cycle involves a condensation reaction of citrulline with aspartate to produce argininosuccinate. The reaction is carried out with the expenditure of ATP molecule.
Want to see more full solutions like this?
Chapter 15 Solutions
Organic And Biological Chemistry
- The main difference between HDL and LDL is that only LDL: O Transports lipids from the liver to the tissue O Transports lipids from the tissue back to the liver O Has a high lipid content relative to protein O Both a and c O Both b and carrow_forwardWhich of these reactions take place in both anaerobic and aerobic respiration? A. glycolysis B. citric acid cycle C. electron-transport chain D. acetyl-CoA formation E. all of the abovearrow_forwardwhat is the role of taurine in lipids digestionarrow_forward
- . Which vitamin or vitamins has ( have) each of the following functions? a. Is involved in prostaglandin synthesis b. Is a coenzymes precursor c. Is involved in calcium deposition in bonearrow_forwardQUESTION 41 In the deficiency referred to in question 40, a deficiency of the form of develops which makes the erythrocyte susceptible to oxidative destruction oxidized, oxaloacetate O oxidized, glutathione reduced, oxaloacetate reduced, glutathionearrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co




