
Concept explainers
Draw the organic products formed in each reaction.
a. ![]() d.
d. ![]() g.
g. 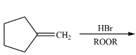
b.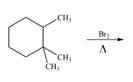 e.
e. 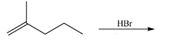 h.
h.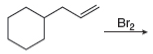
c. ![]() f.
f. 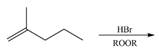 i.
i.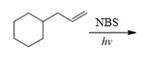
(a)
Interpretation: The organic products formed by the given reaction are to be drawn.
Concept introduction: Alkenes and alkynes show addition reaction due to presence of one and two pi-bonds, respectively. Alkanes show substitution reaction due to absence of pi-bond.
Answer to Problem 15.48P
The organic products formed by the given reaction are shown below.
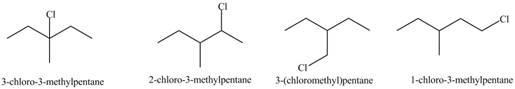
Explanation of Solution
Alkanes undergo chlorination when they are treated with
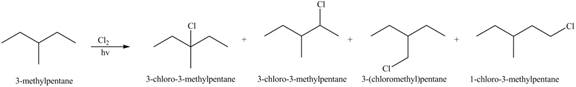
Figure 1
The organic products formed by the given reaction are shown in Figure 1.
(b)
Interpretation: The organic products formed by the given reaction are to be drawn.
Concept introduction: Alkenes and alkynes show addition reaction due to presence of one and two pi-bonds, respectively. Alkanes show substitution reaction due to absence of pi-bond.
Answer to Problem 15.48P
The organic products formed by the given reaction are shown below.
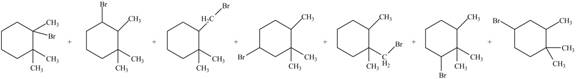
Explanation of Solution
Alkanes undergo bromination by free radical mechanism when they are treated with
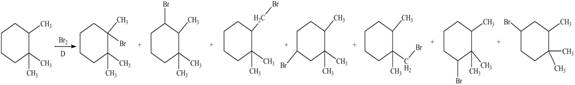
Figure 2
The organic products formed by the given reaction are shown in Figure 2.
(c)
Interpretation: The organic products formed by the given reaction are to be drawn.
Concept introduction: Alkenes and alkynes show addition reaction due to presence of one and two pi-bonds, respectively. Alkanes show substitution reaction due to absence of pi-bond.
Answer to Problem 15.48P
The organic products formed by the given reaction are shown below.
Explanation of Solution
Alkanes undergo bromination by free radical mechanism when they are treated with
Figure 3
The organic products formed by the given reaction are shown in Figure 3.
(d)
Interpretation: The organic products formed by the given reaction are to be drawn.
Concept introduction: Alkenes and alkynes show addition reaction due to presence of one and two pi-bonds, respectively. Alkanes show substitution reaction due to absence of pi-bond.
Answer to Problem 15.48P
The organic products formed by the given reaction are shown below.
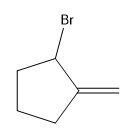
Explanation of Solution
Alkenes undergo bromination by free radical mechanism when they are treated with NBS in the presence of light. Bromination takes place at allylic carbon atom. One product is formed by the given bromination reaction as shown in Figure 3.
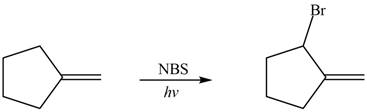
Figure 4
The organic products formed by the given reaction are shown in Figure 4.
(e)
Interpretation: The organic products formed by the given reaction are to be drawn.
Concept introduction: Alkenes and alkynes show addition reaction due to presence of one and two pi-bonds, respectively. Alkanes show substitution reaction due to absence of pi-bond.
Answer to Problem 15.48P
The organic products formed by the given reaction are shown below.
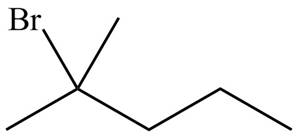
Figure 5
Explanation of Solution
Alkenes undergo addition reaction when they are treated with
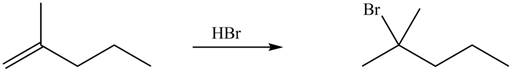
Figure 5
The organic products formed by the given reaction are shown in Figure 5.
(f)
Interpretation: The organic products formed by the given reaction are to be drawn.
Concept introduction: Alkenes and alkynes show addition reaction due to presence of one and two pi-bonds, respectively. Alkanes show substitution reaction due to absence of pi-bond.
Answer to Problem 15.48P
The organic products formed by the given reaction are shown below.
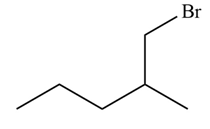
Explanation of Solution
Alkenes undergo addition reaction when they are treated with
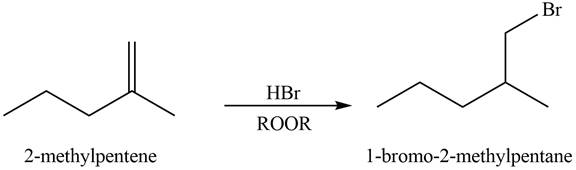
Figure 6
The organic products formed by the given reaction are shown in Figure 6.
(g)
Interpretation: The organic products formed by the given reaction are to be drawn.
Concept introduction: Alkenes and alkynes show addition reaction due to presence of one and two pi-bonds, respectively. Alkanes show substitution reaction due to absence of pi-bond.
Answer to Problem 15.48P
The organic products formed by the given reaction are shown below.
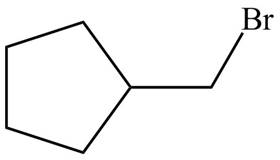
Explanation of Solution
Alkenes undergo addition reaction when they are treated with
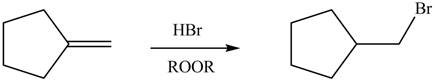
Figure 7
The organic products formed by the given reaction are shown in Figure 7.
(h)
Interpretation: The organic products formed by the given reaction are to be drawn.
Concept introduction: Alkenes and alkynes show addition reaction due to presence of one and two pi-bonds, respectively. Alkanes show substitution reaction due to absence of pi-bond.
Answer to Problem 15.48P
The organic products formed by the given reaction are shown below.
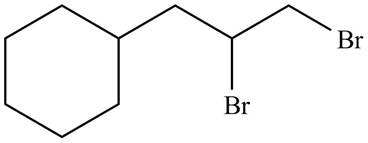
Explanation of Solution
Alkenes undergo addition reaction when they are treated with
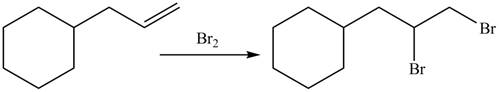
Figure 8
The organic products formed by the given reaction are shown in Figure 8.
(i)
Interpretation: The organic products formed by the given reaction are to be drawn.
Concept introduction: Alkenes and alkynes show addition reaction due to presence of one and two pi-bonds, respectively. Alkanes show substitution reaction due to absence of pi-bond.
Answer to Problem 15.48P
The organic products formed by the given reaction are shown below.
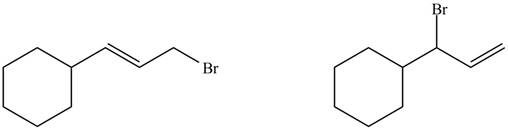
Explanation of Solution
Alkenes undergo bromination by free radical mechanism when they are treated with NBS in the presence of light. Bromination takes place at allylic carbon atom. Two different products are formed by the given bromination reaction as shown in Figure 9.
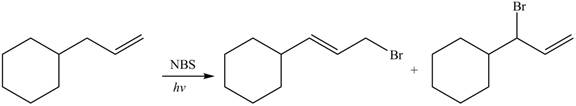
Figure 9
The organic products formed by the given reaction are shown in Figure 9.
Want to see more full solutions like this?
Chapter 15 Solutions
Organic Chemistry-Package(Custom)
- Label each compound as aromatic, antiaromatic, or not aromatic. Assume all completely conjugated rings are planar. Å a. b. C. d.arrow_forwardSynthesize each compound from cyclohexanol using any other organic or inorganic compounds. CH,OH a. g. (Each cyclohexane ring must come from cyclohexanol.) COOH b. d. h. сно CHs (Each cyclohexane ring must come from cyclohexanol.)arrow_forward4.36 Give the IUPAC name for each compound. a. C. e. b. d. f.arrow_forward
- Select the carbonyl compound which can form only a single enol via tautomerization. A. B. H C. D.arrow_forwardDraw a stepwise mechanism for the attached reaction that forms ether D. D can be converted to the antidepressant fluoxetine (trade name Prozac) in a single steparrow_forward4.36 Give the IUPAC name for each compound. a. b. C. e. g. i. mর d. f. h.arrow_forward
- Explain why each compound is aromatic, anti aromatic or non aromatic? Explain C. D. A. B.arrow_forwardDraw a stepwise mechanism for the following reaction that forms ether D. D can be converted to the antidepressant uoxetine (trade name Prozac) in a single step.arrow_forwardDraw a stepwise mechanism for the following reaction that forms ether D. D can be converted to the antidepressant fluoxetine (trade name Prozac) in a single step.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





