
Concept explainers
(a)
Interpretation:
The given radical is to be classified as
Concept introduction:
A free radical is an atom or ion with unpaired electron. They are reactive intermediates formed by the homolysis of covalent bond. Free radicals are classified as
Answer to Problem 15.1P
The given free radical is classified as
Explanation of Solution
The given species is,
![]()
Figure 1
The given free radical is attached to two carbon atoms. Thus, it is classified as
The given free radical is classified as
(b)
Interpretation:
The given radical is to be classified as
Concept introduction:
A free radical is an atom or ion with unpaired electron. They are reactive intermediates formed by the homolysis of covalent bond. Free radicals are classified as
Answer to Problem 15.1P
The given free radical is classified as
Explanation of Solution
The given species is,
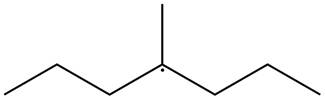
Figure 2
The given free radical is attached to three carbon atoms. Thus, it is classified as
The given free radical is classified as
(c)
Interpretation:
The given radical is to be classified as
Concept introduction:
A free radical is an atom or ion with unpaired electrons. They are reactive intermediates formed by the homolysis of covalent bond. Free radicals are classified as
Answer to Problem 15.1P
The given free radical is classified as
Explanation of Solution
The given species is,
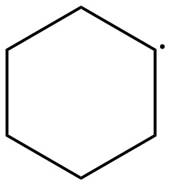
Figure 3
The given free radical is attached to two carbon atoms. Thus, it is classified as
The given free radical is classified as
(d)
Interpretation:
The given radical is to be classified as
Concept introduction:
A free radical is an atom or ion with unpaired electrons. They are reactive intermediates formed by the homolysis of covalent bond. Free radicals are classified as
Answer to Problem 15.1P
The given free radical is classified as
Explanation of Solution
The given species is,
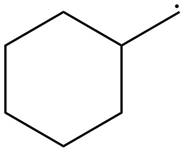
Figure 4
The given free radical is attached to one carbon atom. Thus, it is classified as
The given free radical is classified as
Want to see more full solutions like this?
Chapter 15 Solutions
Organic Chemistry-Package(Custom)
- SO,H HNO3 (1 eq.) H₂SO4 0 °C HNO, (1 eq.) H₂SO4 0 °Carrow_forwardQueation 1arrow_forwardDraw the curved arrows and the products formed in the acid–base reaction of HBr and NH3. Determine the direction of equilibrium. Step 1: What happens in an acid–base reaction? Step 2: Draw the products of the acid–base reaction. Step 3: Draw the curved arrow mechanism of the acid–base reaction. Step 4: Determine the direction of equilibriumarrow_forward
- Which of the following reactions are correct? Explain the answer. + H₂O + H₂O H₂SO4 H OH H OHarrow_forwardDraw the missing starting material. Reagent 1 is benzene and AlCl3. Reagent B is Zn(Hg) and HCl.arrow_forwardDraw a reaction energy diagram that corresponds to the following reaction. conc. H,SO4 OHarrow_forward
- Please match the products to each reaction. O Drag and drop options on the right- hand side to reorder and match with items on the left. Reordering may cause items on the right-hand side to swap positions. 1. Product A 2. Product B 3. Product D Product C H CHO 1) a) H20 NH3 H 2) b) 2 mol CH3OH, H20 OH H 3) c) H20 HO, H 4) d) но. H20 HO. II 4.arrow_forward3. In each blank, put the major product of the reaction. CH3 кон Br KOH OH H+ OH H+arrow_forwardвх H KOH (catalytic) EtOH 78 °Carrow_forward
- Determine the major product of the reaction shown in the box د b C A B C NaBH CH₂OH Select an answer and submit. For keyboard navigation, use the up/down arrow keys to select an answer. d D OH OH B ON OH C b Darrow_forwardDraw (HC(CH3)2)2. Need answer fast i'll rate uparrow_forwardWhich set of answers is organized in orderfrom highest to lowest priority?A. –CH2NH2 > –CN > –COOH > –NH2B. –COOH > –CN > –CH2NH2 > –NH2C. –NH2 > –CN > –CH2NH2 > –COOHD. –NH2 > –COOH > –CN > –CH2NH2E. –CN > –NH2 > –CH2NH2 > –COOHarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





