
Concept explainers
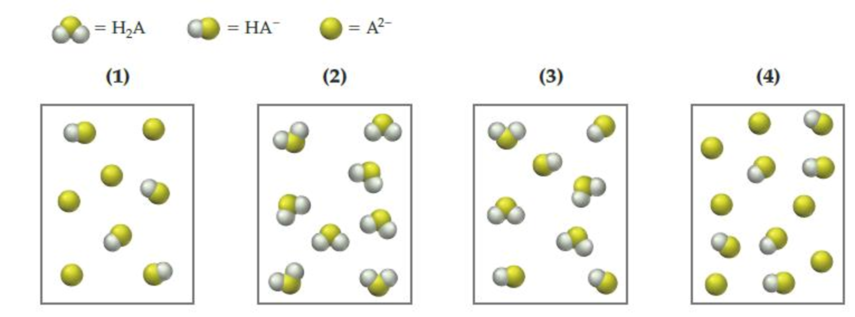
The following pictures represent solutions that contain one or more of the compounds H2A, NaHA, and Na2A, where H2A is a weak diprotic acid. (Na+ ions and solvent water molecules have been omitted for clarity.)
- (a) Which of the solutions are buffer solutions?
- (b) Which solution has the greatest buffer capacity?
(a)
Interpretation:
The buffer solutions should be given from the given representations.
Concept introduction:
Buffer solution:
An equal concentrations of conjugate acid-base pair in a solution is known as buffer solution and it will maintain the
Explanation of Solution
Reason for the correct option:
The given representations are,
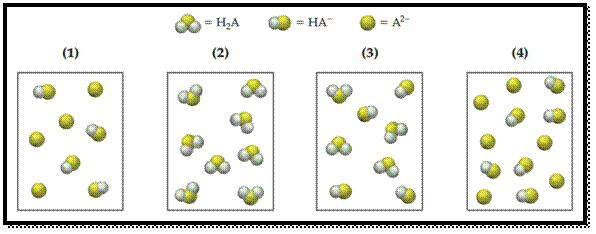
Figure 1
From the above given representations, pictures (1) and (4) are represents the buffer solutions because they are only have a conjugate acid-base pair.
Reason for the incorrect options:
Pictures (2) and (3) don’t have a conjugate acid-base pair so they are not a buffer solutions.
The pictures (1) and (4) are represents the buffer solutions.
(b)
Interpretation:
The greatest buffer capacity having buffer solutions should be given from the given representations.
Concept introduction:
Buffer solution:
An equal concentrations of conjugate acid-base pair in a solution is known as buffer solution and it will maintain the
The buffer capacity is directly proportional to the concentrations (number) of conjugate acid-base pair in a solution.
Explanation of Solution
Reason for the correct option:
The given representations are,

Figure 1
From the above given representations, picture (4) has more number of conjugate acid-base pairs than (1) so it has greatest buffer capacity.
Reason for the incorrect options:
Pictures (2) and (3) don’t have a conjugate acid-base pair so they are not a buffer solutions and Pictures (1) has the lower number of conjugate acid-base pair than (4) so it has a smallest buffer capacity than (4).
The pictures (4) has the highest buffer capacity.
Want to see more full solutions like this?
Chapter 15 Solutions
General Chemistry: Atoms First
- The buffer capacity indicates how much OH- or H+ ions a buffer can react with. What is the buffer capacity of the buffers in Problem 10?arrow_forwardA buffer solution is prepared by adding 5.50 g of ammonium chloride and 0.0188 mol of ammonia to enough water to make 155 mL of solution. (a) What is the pH of the buffer? (b) If enough water is added to double the volume, what is the pH of the solution?arrow_forwardA 20.8 mL sample of 0.279 M ethylamine, C2H5NH2, is titrated with 0.253 M hydrochloric acid. After adding 9.79 mL of hydrochloric acid, the pH isarrow_forward
- a. You want to prepare 1.00 L of a buffer solution with a pH of 3.5. You are given an 0.45 M solution of nitrous acid, HNO2, and an 0.68 M solution of NaOH. What volume of each of these solutions must be mixed in order to get the desired buffer solution? Use the simultaneous equations method to solve this problem. b)If the acid used in a. was nitric acid, HNO3, could the same buffer solution be made? Briefly explain. c) If the buffer solution prepared in a. also contained ~10–6M Ni2+ and ~10–6 M Pb2+ , could those metal ions be separated from each other by saturating the solution with H2S? Explain your answer by showing the necessary calculations.arrow_forwardSuppose that the solubility of Ca(OH)2 in water is 0.3g/0.1L. What is the pH of an aqeous saturated solution of Ca(OH)2?arrow_forwardFor a titration to be effective, the reaction must be rapid and the yield of the reaction must essentially be 100%. Is Kc > 1, < 1, or ≈ 1 for a titration reaction? Kc is the product of all reaction products (choose from: Multiplied by, added to, divided by, subtracted from) the product of the reactants, with all concentrations of reactants and products raised to their respective stoichiometric powers. As the titration proceeds, the amount of reactants (choose from: Increases, decreases, remains the same) and the amount of products (choose from: Increases, decreases, remains the same). Because the value of the numerator is (Choose from much greater than, much less than, approximately equal to) the value of the denominator, the value of Kc will be (Choose from much greater than, much less than, approximately equal to)1 for an effective titration reaction.arrow_forward
- What is the effect of dilution on the pH of a buffer and the buffer’s capacity?arrow_forwardThe following diagram represents a buffer composed ofequal concentrations of a weak acid, HA, and its conjugatebase, A-. The heights of the columns are proportionalto the concentrations of the components of the buffer.(a) Which of the three drawings, (1), (2), or (3), representsthe buffer after the addition of a strong acid? (b) Which ofthe three represents the buffer after the addition of a strongbase? (c) Which of the three represents a situation thatcannot arise from the addition of either an acid or a base?arrow_forwarda. You want to prepare 1.00 L of a buffer solution with a pH of 3.5. You are given an 0.45 M solution of nitrous acid, HNO2, and an 0.68 M solution of NaOH. What volume of each of these solutions must be mixed in order to get the desired buffer solution? Use the simultaneous equations method to solve this problem. b. If the buffer solution prepared in a. also contained ~10–6M Ni2+ and ~10–6 M Pb2+ , could those metal ions be separated from each other by saturating the solution with H2S? Explain your answer by showing the necessary calculations.arrow_forward
- A 29.5 mL sample of 0.258 M ethylamine, C2H5NH2, is titrated with 0.340 M hydrochloric acid.After adding 9.72 mL of hydrochloric acid, the pH isarrow_forwardA buffer solution contains 0.420 M NaHCO3 and 0.262 M Na2CO3. If 0.0303 moles of potassium hydroxide are added to 125 mL of this buffer, what is the pH of the resulting solution ?arrow_forwardA solution is formed by adding 6.00 grams of solid potassium acetate, KC₂H₃O₂, to 200.0 ml of 0.0240M calcium acetate, Ca(C₂H₃O₂)₂. Calculate the pH of the resulting solution. You may assume that the potassium acetate dissolve completely and that the change in volume is negligible with the addition of the solid.arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning

