
Concept explainers
(a)
Interpretation:
The target molecule transformation should be draw and identified given the starting molecule of
Concept Introduction:
Oxidation Reaction: Generally alcohol can be oxidizing to aldehyde or
Reduction Reaction: This process in which any substance atoms, ion or molecule gains one or more electrons it is called reduction.
(a)
Answer to Problem 15.20UKC
The hydride added to the carbonyl carbon, the polar (C=O) carbon has a partial positive charge, then the reduction process has
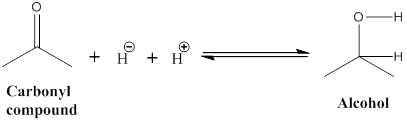
Explanation of Solution
Reduction reaction: The given carbonyl molecule is converted into alcohol (b) in this undergoes for reduction process was occurred. The hydride ion added to the carbonyl carbon, because carbonyl carbon is polar it as partial positive charges in carbon atom.
(b)
To determine give the reactions indicate which direction represents reduction and which represents oxidation.
Concept Introduction:
Carbonyl group: This group presence of a
Oxidation Reaction: Generally alcohol can be oxidizing to aldehyde or ketone and aldehyde can be further oxidized to carboxylic acids.
Reduction Reaction: This process in which any substance atoms, ion or molecule gains one or more electrons it is called reduction.
(b)
Answer to Problem 15.20UKC
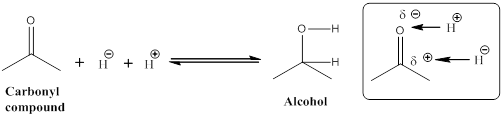
Given the arrow to the right represents reduction and the arrow to the left represents oxidation.
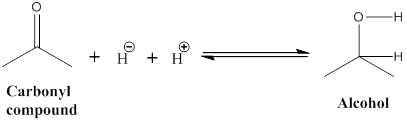
Explanation of Solution
Let us consider given reaction,

Given the arrow to the right represents reduction and the arrow to the left represents oxidation.
Want to see more full solutions like this?
Chapter 15 Solutions
EP FUND.OF GENERAL,ORG...-MOD.MASTERING
- In the preparation of aspirin, You can do the functional group test to ensure the completion of the reaction. (a) What is the name of reagent used? (b) What is your observation if any unreacted starting material is present? (c) What is the name of the functional group responsible for this reaction?arrow_forwardDraw the major organic product of the following reaction: 1. (CH3CH2)2NH, H+ (cat.) 2. о H 3. H3O+arrow_forwardOne biochemical strategy of many living organisms is the stepwise oxidation of organic compounds to CO₂ and H₂O and the conservation of a major part of the energy thus produced in the form of ATP. It is important to be able to recognize oxidation- reduction processes in metabolism. Reduction of an organic molecule results from the hydrogenation of a double bond (Eqn 1) or of a single bond with accompanying cleavage (Eqn 2). Conversely, oxidation results from dehydrogenation. In biochemical redox reactions, the coenzymes NAD and FAD dehydrogenate/hydrogenate organic molecules in the presence of the proper enzymes. CH₂ CH + H-H Acetaldehyde H reduction =(Q=F oxidation L CH₂-C +H+ +H-H Acetate reduction oxidation reduction oxidation CH₂-C CH₂- H H+ O-H reduction oxidation CH₂-C-H Ethanol CH, C-H+ o H Acetaldehyde (1)arrow_forward
- in which of the following alternatives is the greatest amount of energy released from the hydrogenation of:a) 1 mole propeneb) 1 mole of butadiene 1.3c) 1 mole pentyn-2d) 3 moles of ethenearrow_forwardWhich element is oxidized and which is reduced in the following reactions?arrow_forwardConsider the reaction: H O C—C—C—SCOA H₂C(CH₂)C= CO H H₂C-(CH₂) What kind of reaction is being performed here? b. What enzyme performs this reaction? OH H O O=C -C—C—C—SCOA H H c. What cofactors, if any, are required for this reaction?arrow_forward
- (i) During the Pyruvate Decarboxylase (PDC) catalysed reaction, a covalent coenzyme intermediate is formed. On the skeleton of the covalent coenzyme intermediate below draw the appropriate double bonds, charges, and electron pairs to illustrate the two resonance forms of this intermediate. Name each form in the boxes provided. HO S R' N -R HO S R' N -Rarrow_forwardWhich of the following statements is TRUE of enzyme catalysts? for heads up question 2 and 3 are false 1) To be effective, they must be present at the same concentration as their substrate. 2) They can increase the equilibrium constant for a given reaction by a thousand-fold or more. 3) They lower the free energy for conversion of substrate to product. 4) Their catalytic activity is dependent on pH. 5) They are generally equally active on D and L isomers of a given substrate.arrow_forwardDraw the product AND propose a reasonable, detailed stepwise mechanism, using curved arrow notation to show the flow of electrons, for the following reaction.arrow_forward
- Glycine hydrochloride (Cl− H3N+CH2COOH) is a diprotic acid that contains a carboxylic acid group and an ammonium group and is therefore called an amino acid. It is often used in biochemical buffers. Solve, (b) Write the chemical equations describing the dissociation of the first and second protons of Cl−H3N+CH2COOH.arrow_forwardIn the reaction below, the products have a higher free energy (G) than the reactants. What can you conclude about this reaction? AB + C A) It is endergonic with a negative delta G. 4 B) It is exergonic with a negative delta G. AC + B C) It is endergonic with a positive delta G. E) A and D D) It will proceed more quickly in the presence of a catalyst. F) B and D G) C and Darrow_forwardThe synthesis of products is limited by the amount of reactants.(a) If one mole each of CH4, NH3, H2S, and CO2 is added to 1 literof water (= 55.5 moles of H2O) in a flask, how many moles ofhydrogen, carbon, oxygen, nitrogen, and sulfur are in the flask?(b) Looking at the molecular formula in the table, how manymoles of each element would be needed to make 1.0 mole ofglycine? (c) What is the maximum number of moles of glycine thatcould be made in that flask, with the specified ingredients, if noother molecules were made? Explain. (d) If serine or methioninewere made individually, which element(s) would be used up firstfor each? How much of each product could be made?arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





