
Concept explainers
Interpretation:
The structural features of the
Concept introduction:
A carbonyl group has a carbon atom that is connected to an oxygen atom through a double bond and single bonded to two other carbon atoms respectively.
•
• The alcohols contain hydroxyl
• The carboxylic acids contain carboxylic
Answer to Problem 15.1E
The structure of carboxylic acid functional group is shown below.
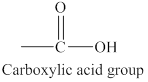
The structure of carboxylic acid functional group is different from alcohols, aldehydes or ketones because it possesses both carbonyl group and hydroxyl group. On the other hand, alcohols has only hydroxyl group and aldehydes or ketones contain only carbonyl group in their structures.
Explanation of Solution
The carboxylic acid functional group has a carbon atom bonded to an oxygen atom by a double bond and bonded to a hydroxyl group
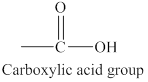
Figure 1
Aldehydes and ketones contain carbonyl functional group in their parent chain. The structural formula of an aldehyde is
The structure of carboxylic acid functional group is different from both aldehydes and ketones because it has hydroxyl group in addition to the carbonyl group.
The alcohols contain only hydroxyl
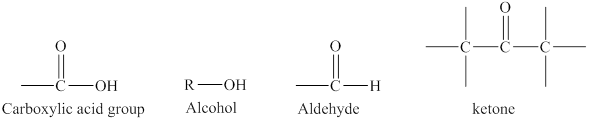 Figure 2
Figure 2
The carboxylic acid functional group structure is shown in Figure 1.
The difference in the structure of carboxylic acid from the structures of an alcohol, an aldehyde or a ketone is rightfully stated.
Want to see more full solutions like this?
Chapter 15 Solutions
Chemistry For Today: General, Organic, And Biochemistry, Loose-leaf Version
- On a microscopic level, how does the reaction of bromine with a saturated hydrocarbon differ from its reaction with an unsaturated hydrocarbon? How are they similar?arrow_forwardExplain why carboxylic acids are much stronger acids than alcohols.arrow_forwardHow does the structure of an alcohol differ from an ether? Describe how an aldehyde differs in structure from a ketone. Thiols are compounds which resemble alcohols, except that the oxygen atom is replaced by a sulfur atom. Draw the analogous thiol for the four carbon alcohol in Table 1. Describe the structural difference between carboxylic acids and esters. Are ethers polar molecules? Would you expect ethers to have higher or lower boiling points than alkanes (circle one)? Explain. Pentane (an alkane) has a boiling point of 36 °C. Does the data agree with your prediction? explain why this could be the casearrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





