
Concept explainers
NMR spectra for four isomeric alkyl bromides with the formula
Assign structures to each of the alkyl bromides and assign the peaks in each spectrum.
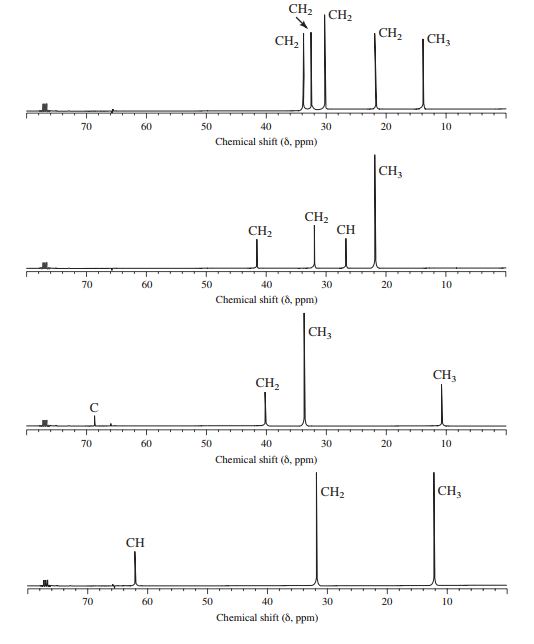
Interpretation:
Structures of each of the isomeric alkyl bromides with the formula
Concept introduction:
In
Carbon atoms having equivalent environment will have the same chemical shift.
In simple molecules, equivalent carbon atoms can be identified just by inspection.
The number of signals depends on the number of distinct carbon atoms in the structure of the compound.
If the structure is symmetrical, the number of signals in the
The electronegative atom/group decreases the shielding of the carbon atom to which it is attached.
In
An
The chemical shift for methyl carbon atoms ranges from
The chemical shift for methylene carbon atoms ranges from
The chemical shift for methine carbon atoms ranges from
The chemical shift for aromatic carbon atoms ranges from
The index of hydrogen deficiency is calculated as follows:
Here
Answer to Problem 43P
Solution:
a)
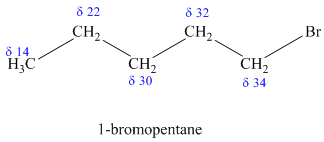
b)
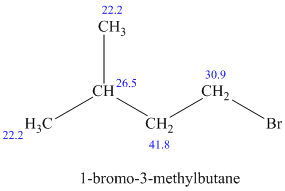
c)

d)

Explanation of Solution
a)
The molecular formula shows an index of hydrogen deficiency equal to zero. Thus, the compound contains ring or multiple bonds. All the carbon atoms must be
The signal at
Combining all this, the only possible structure for the isomer having the formula

b)
The molecular formula shows an index of hydrogen deficiency equal to zero. Thus, the compound contains ring or multiple bonds. All the carbon atoms must be
The signal at
The signal at
Combining all this, the only possible structure for the isomer having the formula
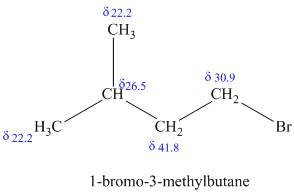
c)
The molecular formula shows an index of hydrogen deficiency equal to zero. Thus, the compound contains ring or multiple bonds. All the carbon atoms must be
The signal at
The signal at
The chemical shift values for the remaining two signals indicate two types of methyl groups. The signal at
Combining all this, the only possible structure for the isomer having the formula

d)
The molecular formula shows an index of hydrogen deficiency equal to zero. Thus, the compound contains ring or multiple bonds. All the carbon atoms must be
The signal at
The signal at
Combining all this, the only possible structure for the isomer having the formula

Want to see more full solutions like this?
Chapter 14 Solutions
ORGANIC CHEMISTRY-W/STUD.SOLN.MAN.
- Below are three MS spectra, Spectra 1, 2, and 3. Each of this mass spectrum corresponds to either Compound A (contains one Cl in the molecular formula), Compound B (one I in the molecular formula), and Compound C (one Br in the molecular formula). Identify which spectra corresponds to which compound. Provide explanations for your choices.arrow_forwardCompound CsH12 gives the following H-NMR. Draw the structure of the compound. Draw a box around the structure you want graded. 5.00 1.00 6.04 70 6.5 6.0 4.0 3.5 3.0 25 20 1.5 A student was adding bromine across the double bond of 2-butene to make 2,3-dibromobutane. After taking the NMR, the student discovered they didn't get the product expected. Based on the NMR, what product did they obtain? Draw a box around your answer. 1.00 2.01 |2.00 3.00arrow_forwardThe 1H and 13C NMR for a compound with the formula C5H12O are shown. Determine the structure of this compound. Assign the peaks: assign “a” to the most shielded hydrogen(s) in your structure, “b” to next-most shielded, etc; use capital letters for carbons).arrow_forward
- As a pioneer in Organic Chemistry you test out a potential new catalyst for the reaction of Br2 with the compound below. After isolating a major product, analysis reveals one Br has been added substituting one H. You take an 1H NMR (shown below) to help decide if an EAS, benzylic substitution or non-benzylic substitution has occurred. Draw the product. Then assign each proton in the product with a letter corresponding to the labels shown in the spectrum below.arrow_forward10 ppm A compound with the molecular formula CsH Br gives the above H-NMR spectrum. What is the structure of the compound? Br- Br Br Br Brarrow_forward4. How many signals would you observe in the ¹H-NMR spectra of the following molecules? NH OHarrow_forward
- Which of these choices best describes the interpretation of the following peak that may be recorded in a H NMR spectrum? 4.3 8 (1H, q). The underlined hydrogen atom is intended to be the one producing the peak that we are interpreting. O=C-CHCO2H O=C-CHCHX2 O=C-CHCH,X O=C-CHCH3 None of these interpretations describes this peak.arrow_forwardAn unknown compound has the formula C3H7NO2. Elucidate its structure by scrutinizing its 1H NMR spectra, shown. Specifically, label each different type of H atom in the final structure with its NMR chemical shift in ppm.arrow_forward3. If there was no proton decoupling, what would you expect the splitting to be for each numbered carbon? 3a. carbon 1 splitting 3b. carbon 2 splitting 3c. carbon 3 splitting 3d. carbon 4 splitting Structure And Functioarrow_forward
- Below is the ¹H NMR spectrum of a constitutional (structural) isomer of C9H100, with the integration given above each peak. In addition to other peaks not given here, the IR spectrum of the isomer showed a strong peak at 1700 cm-1 Determine which of the structures given below the spectrum matches the ¹H NMR spectrum. H₂C 2 8 A C 3 CH3 CH 3 to B D Ho PPM CH3 =CH₂ 2 3arrow_forwardWhich of these choices best describes the interpretation of the following peak that may be recorded in a 'H NMR spectrum? 2.5 8 (2H, d). The underlined hydrogen atom is intended to be the one producing the peak that we are interpreting. O=C-CH,CO2H O=C-CH2CHX2 O=C-CH2CH2X O=C-CH2CH3 None of these interpretations describes this peak.arrow_forward10- If 3-Bromo-1-phenyl-1-propene shows a complex NMR spectrum in which thevinylic proton at C2 is couples with both the C1 vinylic proton (J = 16 Hz) and the C3methylene protons (J = 8 Hz) Draw a tree diagram for the C2 proton signal.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole



