
Concept explainers
Describe the
the splitting pattern for each signal, and the approximate chemical shift?

(a)
Interpretation:
The
Concept introduction:
The number of NMR signal in a compound is equal to the number of chemically non-equivalent protons present in that compound. In
Answer to Problem 14.17P
The
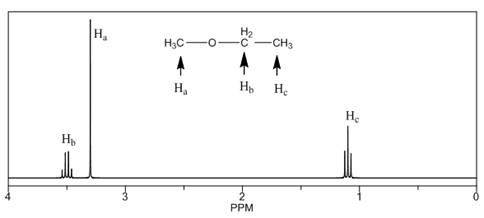
The number of NMR signals, the splitting pattern for each signal and the approximate chemical shift values are rightfully stated.
Explanation of Solution
The given compound is ethyl methyl ether
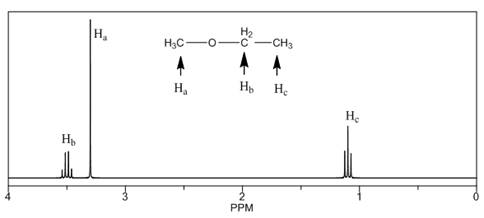
Figure 1
The proton
The
(b)
Interpretation:
The
Concept introduction:
The number of NMR signal in a compound is equal to the number of chemically non-equivalent protons present in that compound. In
Answer to Problem 14.17P
The
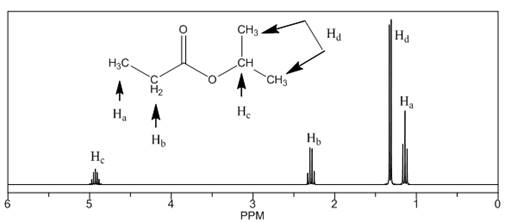
The number of NMR signals, the splitting pattern for each signal and the approximate chemical shift values are rightfully stated.
Explanation of Solution
The given compound is isopropylpropionate
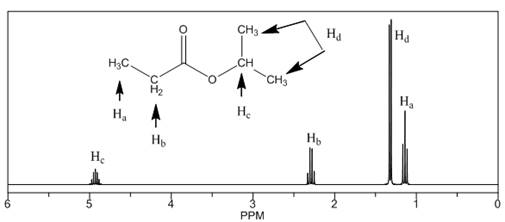
Figure 2
The proton
The
(c)
Interpretation:
The
Concept introduction:
The number of NMR signal in a compound is equal to the number of chemically non-equivalent protons present in that compound. In
Answer to Problem 14.17P
The
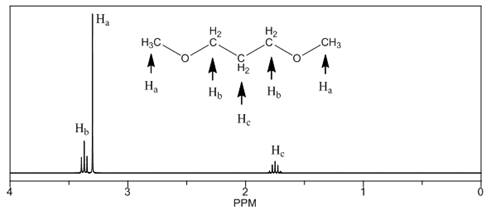
The number of NMR signals, the splitting pattern for each signal and the approximate chemical shift values are rightfully stated.
Explanation of Solution
The given compound is
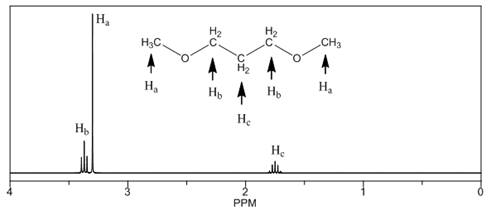
Figure 3
The proton
The
(d)
Interpretation:
The
Concept introduction:
The number of NMR signal in a compound is equal to the number of chemically non-equivalent protons present in that compound. In
Answer to Problem 14.17P
The
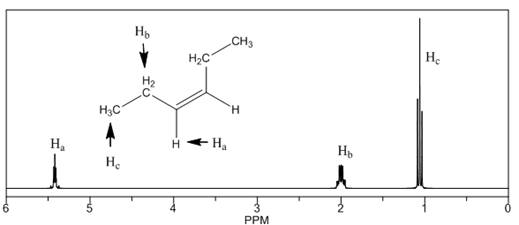
The number of NMR signals, the splitting pattern for each signal and the approximate chemical shift values are rightfully stated.
Explanation of Solution
The given compound is

Figure 4
The proton
The
Want to see more full solutions like this?
Chapter 14 Solutions
Organic Chemistry
- Indicate the correct option.a) Graphite conducts electricity, being an isotropic materialb) Graphite is not a conductor of electricityc) Both are falsearrow_forward(f) SO: Best Lewis Structure 3 e group geometry:_ shape/molecular geometry:, (g) CF2CF2 Best Lewis Structure polarity: e group arrangement:_ shape/molecular geometry: (h) (NH4)2SO4 Best Lewis Structure polarity: e group arrangement: shape/molecular geometry: polarity: Sketch (with angles): Sketch (with angles): Sketch (with angles):arrow_forward1. Problem Set 3b Chem 141 For each of the following compounds draw the BEST Lewis Structure then sketch the molecule (showing bond angles). Identify (i) electron group geometry (ii) shape around EACH central atom (iii) whether the molecule is polar or non-polar (iv) (a) SeF4 Best Lewis Structure e group arrangement:_ shape/molecular geometry: polarity: (b) AsOBr3 Best Lewis Structure e group arrangement:_ shape/molecular geometry: polarity: Sketch (with angles): Sketch (with angles):arrow_forward
- (c) SOCI Best Lewis Structure 2 e group arrangement: shape/molecular geometry:_ (d) PCls Best Lewis Structure polarity: e group geometry:_ shape/molecular geometry:_ (e) Ba(BrO2): Best Lewis Structure polarity: e group arrangement: shape/molecular geometry: polarity: Sketch (with angles): Sketch (with angles): Sketch (with angles):arrow_forwardDon't used Ai solutionarrow_forwardDon't used Ai solutionarrow_forward
- reaction scheme for C39H4202 Hydrogenation of Alkyne (Alkyne to Alkene) show reaction (drawing) pleasearrow_forwardGive detailed mechanism Solution with explanation needed. Don't give Ai generated solutionarrow_forwardShow work with explanation needed....don't give Ai generated solutionarrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning


